In this interview, News-Medical talks to Sylvia Xie, product scientist at Sino Biological, about the ongoing emergence of transmembrane proteins as targets for therapeutic antibodies.
What is the current landscape of antibodies targeting membrane proteins?
As of June 2022, 162 antibodies had been approved for a wide array of diseases in the world, including cancers, immune-related diseases, infectious diseases, and hematological diseases1. Over half target membrane proteins, primarily proteins with large extracellular domains and simple transmembrane domains such as tyrosine kinase receptor proteins. Only two target complex transmembrane proteins like G-protein-coupled receptors (GPCRs)2.
What are the established and emerging membrane protein targets for antibody therapies?
Established antibody targets include transmembrane proteins EGFR, VEGFR2, FGFR2, HER2, and complex membrane proteins CCR4 and CGRPR. Monoclonal and bispecific antibodies targeting complex membrane proteins such as GPRC5D, Claudins (Claudin 6 and Claudin 18), SSTR2, and CXCR4 have shown successful results in CAR-T cell therapy at the preclinical and clinical stages.
Why are membrane proteins important in drug development?
Membrane proteins account for approximately 20% of the human proteome and play crucial roles in biological functions such as protein ligation, recognition, transport, anchoring, and transduction. Mutations in protein folding or assembly are major causes of human diseases including cancers and cardiovascular diseases3. Membrane proteins comprise approximately 60% of possible drug targets and are a constant focus in pharmaceutical and biological research. Therefore, the development of drugs targeting membrane proteins has significant therapeutic value across a wide range of diseases.
What are the main challenges associated with complex membrane protein expression and what are the solutions?
Complex membrane proteins are important therapeutic antibody targets, but they are still underutilized due to production difficulties. Three key challenges limit their use in drug development and experimental research: 1) low expression; 2) unstable purification; 3) difficulty maintaining natural conformation.
Membrane proteins naturally exist in low abundance and recombinant overexpression of membrane proteins commonly results in aggregation and low yield correlated with low functionality. When separated from a surrounding phospholipid environment, membrane proteins may become insoluble and display a loss of activity or function4. Their poor water solubility also makes them difficult for in vivo applications as secreted proteins need to be in aqueous environments for diffusion or circulation. Purification is difficult due to the low abundance of membrane proteins and detergents are usually required to stabilize them in aqueous environments. Furthermore, an antibody must recognize the specific epitope of a membrane protein for desired binding to occur. Since membrane proteins are complex and can exhibit multiple conformational states, expression in the proper form is crucial for antibody-antigen binding affinity2.
To address these challenges, various strategies have been developed, including the use of detergents, polymers, ligands, or lipids to enhance protein stability and extraction4. Detergent choice is important and usually requires a series of tests; some strong detergents are very efficient at isolating the protein at the expense of reduced stability and loss of conformational epitopes5. Proteins can be difficult to detect if the antibodies are unable to recognize the epitopes. The inclusion of epitope tags can increase detectability, but they can potentially disrupt protein folding and function4.
What techniques can be used to express these difficult-to-produce transmembrane proteins?
Traditionally, membrane protein expression consists of the recombinant expression followed by isolation using detergents. One solution to purify membrane proteins is the use of detergents. They are amphipathic molecules with hydrophilic heads and hydrophobic tails which aggregate and form micelles that can trap hydrophobic molecules, extracting them from the membrane (Figure 1a).
Another option is the nanodisc technique which takes advantage of transmembrane proteins retaining their characteristics when bound to phospholipids. Encapsulation of the membrane-embedded protein in membrane scaffold protein (MSP) nanodiscs requires prior solubilization with detergents4 (Figure 1b). Using styrene-maleic acid (SMA) copolymer, membrane proteins can be extracted directly from prokaryotic and eukaryotic expression systems in the absence of detergents, preserving the protein structure and function (Figure 1c). Compared to MSP nanodiscs, SMA nanodiscs also have the advantage of preserving proteins' nature by maintaining the surrounding native lipids without introducing any heterologous proteins. This enables the study of protein structure and function in native-like environments4.
A third option is to use virus-like particles (VLPs) which are nanoscale particles formed by the self-assembly of viral capsid proteins, typically 100-300 nm in diameter. VLPs lack viral genetic material and cannot replicate autonomously, but retain the ability to penetrate cells which makes them suitable for a variety of applications6. On their surfaces, VLPs are capable of displaying membrane proteins in their natural and stable conformation (Figure 1d).
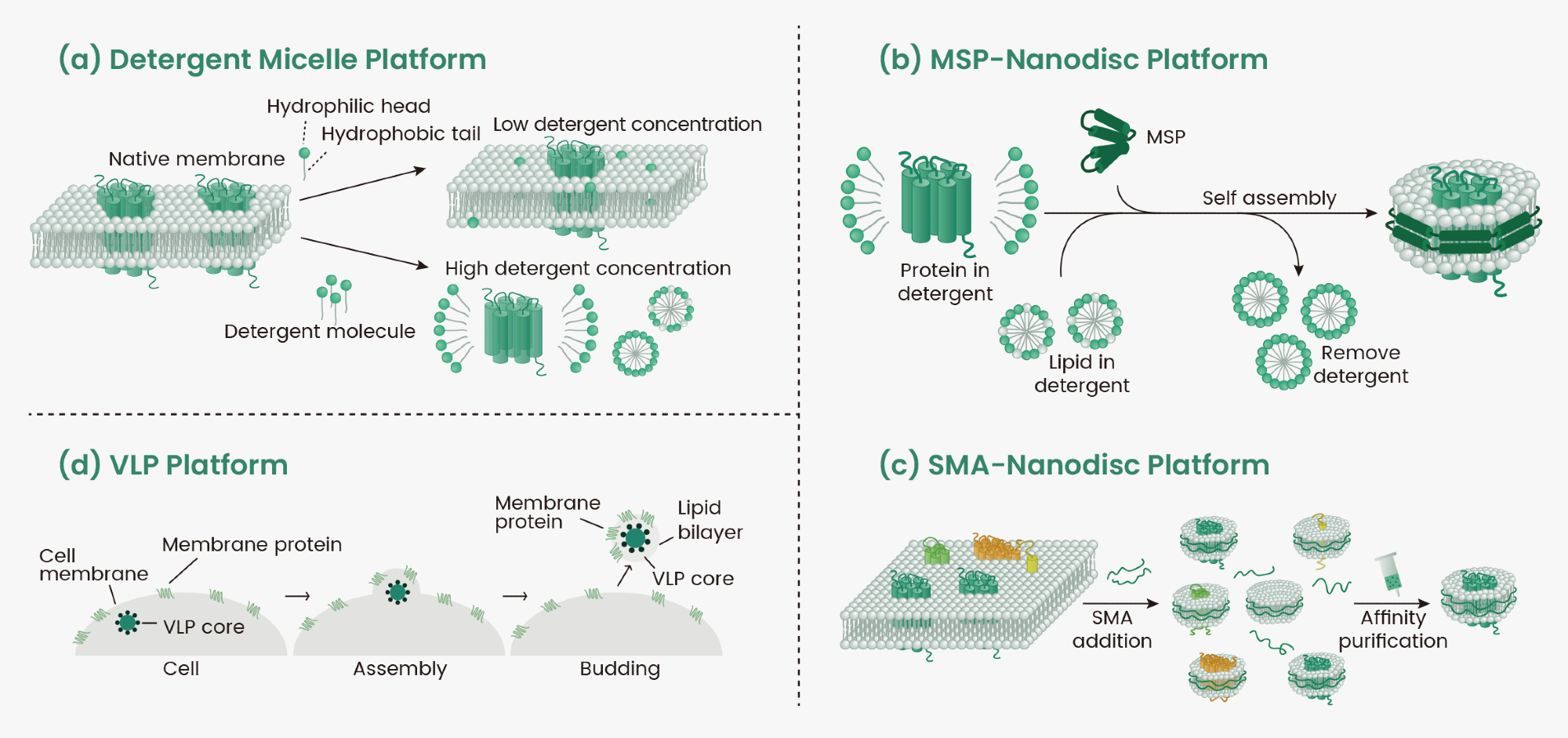
Figure 1. Schematic diagram of detergent, nanodisc, and VLP platforms. Image Credit: Sino Biological Inc.
How does Sino Biological support the development of membrane proteins?
Sino Biological is a leader in manufacturing high-quality multi-pass transmembrane protein products. Our three HEK293 expression-based platforms, VLP, detergent micelle, and SMA-nanodisc platforms, generate premium raw materials for early-stage drug development. A number of multi-pass transmembrane proteins (e.g., 4-pass: Claudin-6, Claudin-9, Claudin-18.1, and Claudin-18.2; 7-pass: GPRC5D, CXCR4, and SSTR2) are available in stock to readily support related research. Additionally, Sino Biological offers custom multi-pass transmembrane protein services based on the three platforms. The service workflow consists of the following steps: 1) gene synthesis and codon optimization; 2) vector construction; 3) pilot-scale protein expression and purification based on the selected platform; 4) large-scale expression and purification.
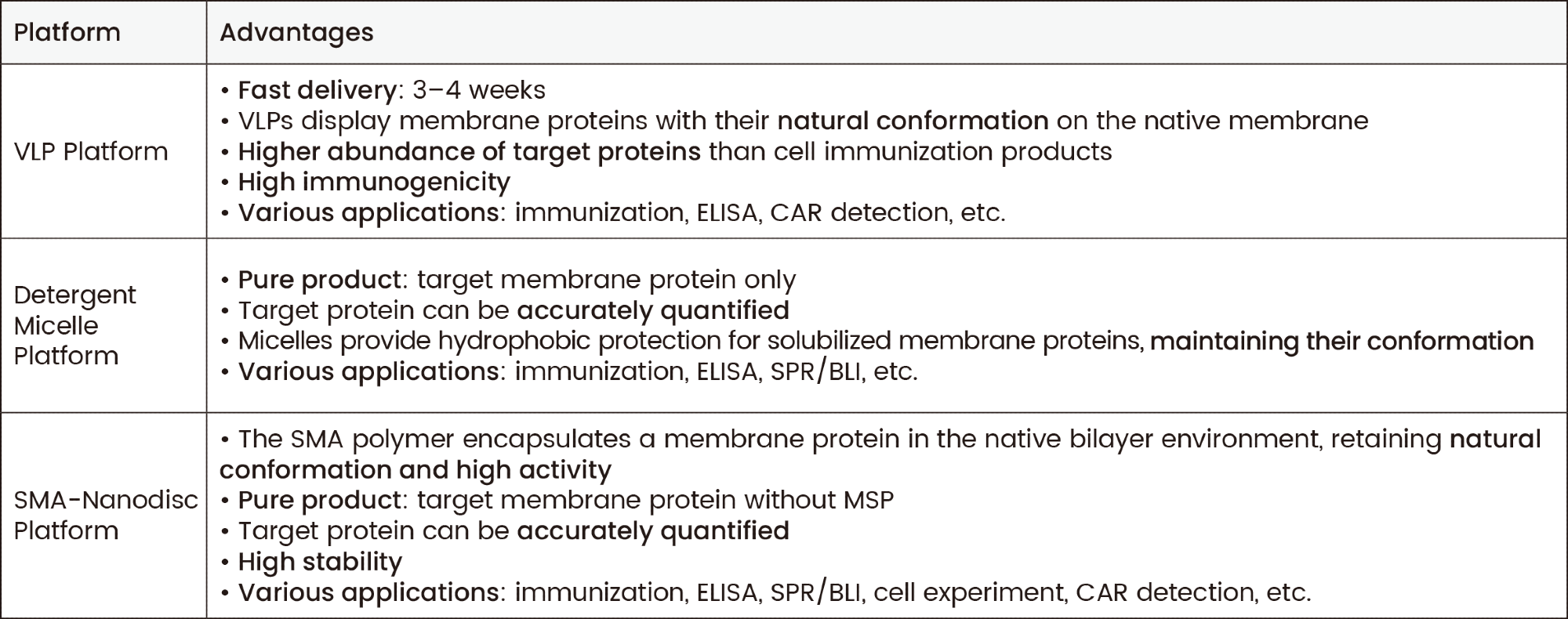
Each transmembrane protein platform has its unique features. The advantages of VLP, detergent micelle, and SMA-nanodisc platforms are summarized in Table 1.
Table 1. Advantages of VLP, detergent micelle, and SMA-nanodisc platforms. Sino Biological
About the interviewee
Sylvia Xie is a Product Scientist at Sino Biological, a global leader in recombinant technology, where she manages the product catalog and supports product development. Prior to joining Sino Biological, Sylvia received her MS degree from the University of Houston (TX, USA) in Biomedical Engineering, where her research focused on targeted drug therapy in autoimmune diseases.
References and further reading
- https://doi.org/10.1093/abt/tbac021
- https://doi.org/10.1016/j.eng.2020.11.013
- https://doi.org/10.1093/bib/bbaa132
- https://doi.org/10.1007/s40259-018-0289-y
- https://doi.org/10.1038/s41598-020-73285-9
- https://doi.org/10.1186/s12951-021-00806-7
About Sino Biological Inc.
Sino Biological is an international reagent supplier and service provider. The company specializes in recombinant protein production and antibody development. All of Sino Biological's products are independently developed and produced, including recombinant proteins, antibodies and cDNA clones. Sino Biological is the researchers' one-stop technical services shop for the advanced technology platforms they need to make advancements. In addition, Sino Biological offers pharmaceutical companies and biotechnology firms pre-clinical production technology services for hundreds of monoclonal antibody drug candidates.
Sino Biological's core business
Sino Biological is committed to providing high-quality recombinant protein and antibody reagents and to being a one-stop technical services shop for life science researchers around the world. All of our products are independently developed and produced. In addition, we offer pharmaceutical companies and biotechnology firms pre-clinical production technology services for hundreds of monoclonal antibody drug candidates. Our product quality control indicators meet rigorous requirements for clinical use samples. It takes only a few weeks for us to produce 1 to 30 grams of purified monoclonal antibody from gene sequencing.