 More than one chromatographical method needs to be utilized to assess and characterized a protein biochemical due to its heterogeneity.
More than one chromatographical method needs to be utilized to assess and characterized a protein biochemical due to its heterogeneity.
Some of the methods utilized include size exclusion chromatography for the quantitation of dimers as well as aggregates. Ion exchange is another approach to assess charge variants. These methods utilize non-denaturing conditions as well as aqueous eluents.
To characterize a protein molecule fully the primary amino acid sequence and the post translational modifications on the sequence needs to be understood. Normally reverse phase HPLC is the method of choice in these cases as denaturing conditions are required.
Experiment
Reverse Phase Agilent ZORBAX Rapid Resolution High Definition (RRHD) 300SB-C18 columns (1.8 µm, 2.1X50mm p/n 857750-902) were used. These have an advantage of improved packing process and ability to withstand pressures up to 1200 bar when used with the Agilent 1290 Infinity LC.
The sample was insulin, oxidized insulin chain A and chain B obtained from bovine pancreas. The sample concentration was 1mg/ml.
Results
The results revealed:-
- Better speed – the system was able to separate the test mixture rapidly and distinguished insulin from contaminants in five minutes. Multiple gradients did not slow the process down. The rapid equilibration meant that the system could be suitable for use with a wide range of organic samples. Flow rate could be adjusted to make the separation even faster without changing the efficiency.
- Good reproducibility – the system took 200 consecutive injections of samples and proved its reproducibility of results. The peak shape, retention time as well as efficiency remained same after 200 insulin injections without cleaning the system columns in between.
- Heat degraded insulin separation – heat degrading the sample insulin resulted in formation of heat degradation products that were rapidly resolved in the system column from the existing monomer insulin concentration.
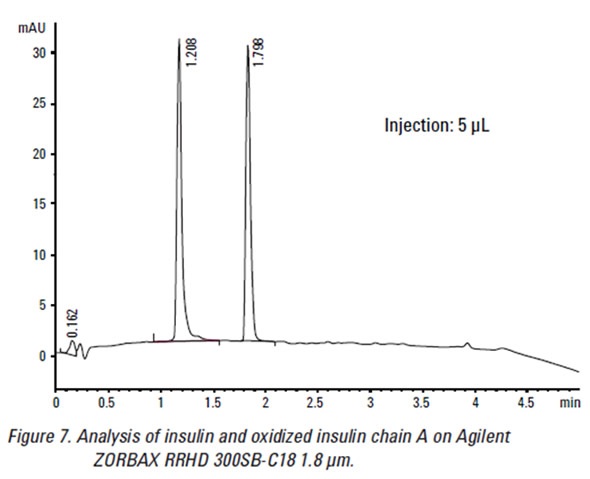
- Insulin isoform separation – the column also separated the insulin isoforms like oxidized insulin chain A. The separation remained robust in efficacy despite changing gradient systems. Shallower gradients, however, were found to be preferable in analyzing oxidized insulin chain A to allow the molecule to stay longer in the column for analysis.
Summary
The results showed that using Agilent ZORBAX Rapid Resolution High Definition (RRHD) 300SB-C18 1.8 µm columns resulted in faster and reliable separation of the isoforms and purified versions of insulin from the sample.
The system allows for high pressure UHPLC and the Stablebond 300 Å pore sized particles could remain unbroken even in acidic conditions of the system.
The system is well-suited to quality control checks of the structures of primary proteins.
Further information
About Agilent Technologies

Agilent Technologies Inc. (NYSE: A) is the world's premier measurement company and a technology leader in chemical analysis, life sciences, diagnostics, electronics and communications. The company's 20,500 employees serve customers in more than 100 countries. Agilent offers the broadest range of innovative measurement solutions in the industry. The company's four businesses - Chemical Analysis, Life Sciences, Diagnostics and Genomics, and Electronic Measurement - provide customers with products and services that make a real difference in the lives of people everywhere. And at Agilent Research Laboratories, we conduct research that anticipates customer needs and produces breakthroughs that power growth.. Information about Agilent is available at https://www.agilent.com/.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.