Introduction
Chromatin immunoprecipitation (ChIP) is used to study the interactions between proteins and DNA. This process can be categorized into two methods – native (N-ChIP) and cross-linked (X-ChIP). Native chromatin prepared by enzymatic digestion is used by N-ChIP, which can be employed to study histone modifications and transcription factors that are tightly bound to DNA.
Chemicals are used to cross link the DNA and protein components of chromatin in X-ChIP, which is suitable for histone modifications and more loosely bound, or low abundant, transcription factor protein targets. The two assays can be carried out at gene/locus specific sites via genome wide analysis and qPCR through downstream sequencing (X or N-ChIP-seq).
Chromatrap® supplies high-quality X-ChIP-seq and N-ChIP-seq kits. In this article, the enrichment of the histone H3K4me3 modification signal across the whole genome is compared, using the chromatin prepared by the two techniques (native and cross-link).
To demonstrate the efficiency and sensitivity of the Chromatrap® spin column technology for native and cross-linked ChIP-seq, H3K4me3 was considered in this analysis. ‘Open’ transcription start sites and gene activation are found associated with the histone mark H3K4me3. The histone exists in euchromatin throughout the genome and is a rich epigenomic target.
Method
Chromatin preparation
The human endometrial epithelial cancer cell line Hec50 (Holinka et al., 1996) was used for preparing chromatin for X-ChIP and N-ChIP, according to the Chromatrap® protocols. 1 x 106 cells were briefly grown to 80% confluency for the N-ChIP, and these cells were scraped in ice cold PBS and collected through centrifugation.
Hypotonic Buffer was used to lyse the cells, and the separated nuclei were exposed to micrococcal nuclease digestion to create chromatin fragments between 100 and 500 bp long. Then, the samples were dialyzed to eliminate undesirable impurities and processed for ChIP. In the case of X-ChIP, 1 x 106 cells were grown to 80% confluency and fixed in 1% formaldehyde for a period of 10 minutes.
Glycine was used to quench the fixation reaction, and the cells were collected in ice-cold PBS. Once the cells were spun down and the supernatant was discarded, they were re-suspended and lysed in Hypotonic Buffer. Centrifugation was performed to collect the released nuclei, which were lysed in Lysis Buffer and the chromatin was sheared by sonication to achieve 100-500 bp length fragments.
Chromatin IP
Slurries were prepared at a 5:2 chromatin: antibody (H3K4me3 – Cat no.70010) ratio (μg) for immunoprecipitation. Slurries were incubated for one hour at 4 °C prior to immunoprecipitation with the Chromatrap® spin columns following the appropraite Native or X-linked protocol. At the same time, inputs consisting of 5 μg chromatin were prepared which were used for downstream analyzes but not subjected to ChIP enrichment.
After elution, the X-ChIP samples were reverse cross-linked for two hours, before subjecting them to Proteinase K digestion. Proteinase K digestion was immediately done on the N-ChIP samples. Before library preparation, all samples were purified using kit supplied Chromatrap® DNA purification columns and reagents.
Library preparation
The NEBNext® UltraTM (NEB) library preparation kit was used for preparing libraries, which were quantified through the KAPA Biosystems (Roche) quantification kit for NGS. To confirm sample size distributions between 200 and 300 bp, as shown in Figure 1, samples were processed on an Agilent Bioanalyzer (Agilent Technologies).
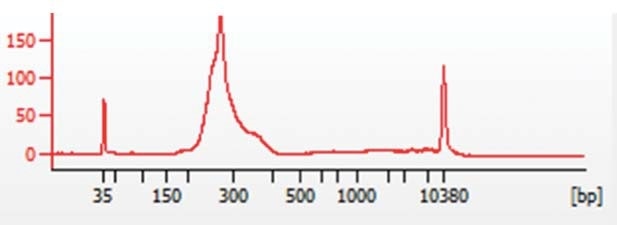
Figure 1. Representative Bioanalyzer trace showing chromatin sample size distribution between 200 and 300bp.
For additional information and help with designing the ChIP-seq experiment, readers can refer to Chromatrap®’s Top 10 tips for library preparation brochure.
Chromatrap® ChIP-seq analysis
The Illumina® MiSeq was used to perform paired end (2x75 bp reads) high throughput sequencing, and FASTQC was employed to analyze the quality of the sequencing data. The ENCODE UCSC genome browser database was used to map the sequences and Bowtie2 was used for alignments.
MACS2 together with Chromatrap® ChIP-seq data analysis software was used to analyze peak enrichment data, with input for N and X-linked chromatin samples as background/baseline. To visualize H3K4me3 enrichment peaks, integrative genomics viewer online software (IgV1.4) was used for the X-ChIP and N-ChIP samples.
Results
Both N and X-linked ChIP provided high-quality sequence scores (FASTQC software). For all samples, duplication rates were less than 5%, and Q30 scores over 30 (Q30 scores correlate to a 1 in 1000 probability of a base being called incorrectly) were noted. Figure 2 A-C respectively shows the FastQC scores and the duplication levels acquired for H3K4me3 for X-ChIP and N-ChIP samples.
A. Sequence quality and duplication
|
Sample
|
Q30
|
Duplication rates
|
|
N-ChIP H3K4me3
|
>30
|
4.13%
|
|
X-ChIP H3K4me3
|
>30
|
2.58%
|
B. Sequence duplication levels for N-ChIP and X-ChIP respectively
.jpg)
.jpg)
C. Q30 scores for N-ChIP and X-ChIP samples respectively
.jpg)
.jpg)
Figure 2. FastQC sequence quality and duplication for both N-ChIP and X-ChIP sequenced samples.
Chromatrap® ChIP-seq Data Analysis software was employed to analyze Native and X-linked sequenced samples for peak enrichment. 65000 peaks, with strong enrichment were observed for N-ChIP samples; peaks displayed 3->30 fold enrichment over the input. For X-ChIP, 39000 peaks with strong enrichment were obtained. Due to the potential of formaldehyde cross linking to mask protein epitopes, which make them less accessible to the antibody, a greater number of peaks is expected from N-ChIP.
For both Native and X-linked chromatin samples, low resolution genome wide IGV visualized peak enrichment showed similar H3K4me3 peaks. Figures 3A and 3B show the peak maps, indicating similar levels of H3K4me3 enrichment across the genome for the N-ChIP and X-ChIP, respectively. This shows that Chromatrap® immunoprecipitation is reproducible and comparable between N-ChIP and X-ChIP-seq on a genome wide scale.
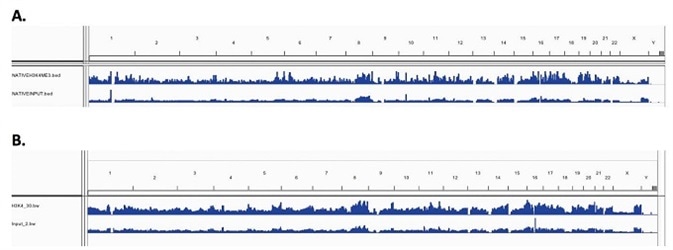
Figure 3. Enrichment peak maps of H3K4me3 across the whole genome (A) N-ChIP (B) X-ChIP
Uniquely identified genes
The identified peaks were mapped to their closest gene loci (+/-5000 bp distance to the TSS to find out which peaks relate to each gene), and the uniquely identified genes were compared between N and X-linked ChIP samples so as to determine the similarities in the H3K4me3 genome wide landscape. In the N-ChIP samples, 20315 uniquely mapped genes were associated with H3K4me3, while in the X-linked ChIP samples, 19508 unique gene loci were associated with H3K4me3, highlighting a 90% resemblance between samples, as shown in Figure 4.
.jpg)
Figure 4. H3K4me3 uniquely identified genes
When analyzing specific gene loci, additional validation of the homology between X-ChIP-seq and N-ChIP-seq is seen. Figure 5 shows that in N and X-linked ChIP samples, the ubiquitously expressed DNA binding protein ZNF566 showed comparable H3k4me3 peak enrichment above background.
.jpg)
Figure 5. H3K4me3 enrichment onto ZNF566 for both N and X-linked Chromatrap ChIP-seq
Conclusion
In this article, Chromatrap® demonstrated robust and comparable enrichment of H3K4me3 from cross-linked and native chromatin. It is highlighted in the study that both native and cross-linked chromatin preparation techniques can be used with Chromatrap® extraction and capture technology.
These methods, by providing excellent data, ensure excellent H3K4me3 enrichment and high throughput sequencing. Using the two assay technologies, high-quality FASTQC metrics and considerable genome wide peak enrichment levels are achieved. Chromatrap® has a wide range of ChIP assays, which provide users with high flexibility and choice for different chromatin preparation methods as well as enrichment for downstream next generation sequencing.
About Chromatrap®
 Chromatrap® is a product of Porvair Sciences, a wholly owned subsidiary of Porvair plc.
Chromatrap® is a product of Porvair Sciences, a wholly owned subsidiary of Porvair plc.
They are one of the largest manufacturers of Ultra-Clean microplates, 96 well well filtration plates and Microplate handling equipment for life science and synthetic chemistry. With offices and Class VIII clean room manufacturing located in the UK, combined with a world-wide network of distributors and dedicated distribution hub in the USA, we pride ourselves on their continuous innovation, research and flexibility to meet customer demands. We offer OEM production and contract manufacturing through their North Wales facility.
Their porous polymeric material, BioVyon™, whose chemical functionalisation can endow it with internal surface properties individually configured to capture and separate target species out of difficult mixtures, has opened up many possibilities in the field of BioSciences where molecules of interest such as DNA, RNA, proteins etc can be selectively pulled out of complex mixtures of biological origin. The materials have proven to be a remarkably good substrate for accepting novel chemistries such as the organically bound Protein A and Protein G in Chromatrap®.
Using their 25 years experience of microplate manufacturing, Porvair Sciences has now developed a high-throughput bead-free ChIP assay based on their filtration plates containing their Chromatrap chemistry. Chromatrap-96 enables large scale epigenetic screening to become a reality in many laboratories and eliminates many of the long and laborious steps previously undertaken in such work.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.