Lysozyme is abundant in secretions such as saliva, human milk, tears and mucus, and is also known as N-acetylmuramide glycanhydrolase or muramidase.
It is an antimicrobial enzyme that is generated by animals and creates part of the innate immune system that plays a key part in the prevention of bacterial infections.
Lysozyme acts as a mild antiseptic as it is able to destroy the cell walls of certain bacteria and is a frequently utilized enzyme for lysing Gram-positive bacteria. For instance, E. coli can be lysed by utilizing lysozyme to free the contents of the periplasmic space.
The main structure of lysozyme is a single polypeptide that contains 129 amino acids. Lysozyme is folded into a compact, globular structure with a long cleft on the protein surface in physiological conditions. It was the first enzyme structure to be solved using X-ray diffraction.
The low cost and fairly simple structure make it a popular model in a lot of biological research that is currently underway. Lysozyme denatures at high temperatures, and the molecular weight of lysozyme is 14.4 KDa, which is small.
The scattering intensity of lysozyme is a challenge for dynamic light scattering (DLS) measurement as it is very weak.
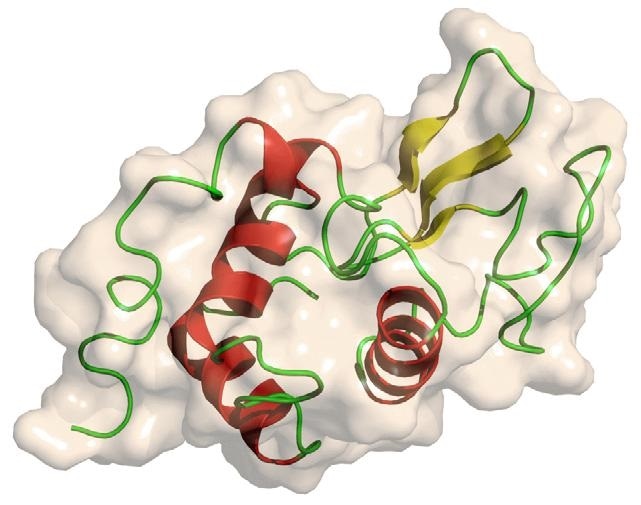
Three-dimensional Structural Lysozyme According to Protein Data Bank. Image Credit: Bettersize Instruments
Theory and instrumentation
DLS quantifies the intensity fluctuations of the sample due to Brownian motions of particles. The diffusion coefficient D is gathered and related to the particle size, i.e., the hydrodynamic diameter DH, by the Stokes-Einstein equation:
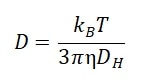
Where kB is the Boltzmann constant, η is the dispersant viscosity and T is the temperature. The molecular weight may be calculated by the empirical Mark-Houwink equation after gathering the diffusion coefficient:
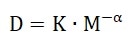
Where K and α are constants that relate to the molecular density to some extent.
In this article, the lysozyme sample was characterized using the BeNano 90 Zeta (Bettersize Instruments), which utilizes a 10 mW laser with a wavelength of 633 nm.
Single mode optical fibers are also employed for signal transmission to maximize the signal-noise ratio in the BeNano 90 Zeta; high speed correlators are used such that the fast decay of correlation functions for small particles can be sufficiently calculated.
Experiment
Lysozyme samples with varying concentrations were prepared in a PBS buffer solution that had a pH of 7. Table 1 shows the sample information.
Table 1. Information of lysozyme sample. Source: Bettersize Instruments
| No. |
Concentration |
Note |
| 1 |
10 mg/mL |
|
| 2 |
5 mg/mL |
|
| 3 |
30 mg/mL |
Protein denaturation study |
Using a built-in temperature control system of the BeNano 90 Zeta, the measurement temperature was set to be 25°C ± 0.1°C. The presence of impurities like dust would have a huge effect on the measurement results, as the scattering intensity was very weak and the lysozyme molecules were extremely small.
The samples were filtered by a 220 nm filter before the measurement for those reasons. Each sample was measured a minimum of three times to ensure repeatability of the results.
Results and discussion
The scattered light signals of the samples were utilized to gather correlation functions. Correlation functions of small particles decayed very quickly because of the rapid Brownian motion, as shown in Figures 1-4.
.jpg)
Figure 1. Correlation functions of 10 mg/mL lysozyme solution. Image Credit: Bettersize Instruments
Figure 2. Size distributions of 10 mg/mL lysozyme solution. Image Credit: Bettersize Instruments
.jpg)
Figure 3. Correlation functions of 5 mg/mL lysozyme solution. Image Credit: Bettersize Instruments
.jpg)
Figure 4. Particle size distributions of 5 mg/mL lysozyme solution. Image Credit: Bettersize Instruments
The excellent stability and sensitivity of the BeNano 90 Zeta were demonstrated by the signal-noise ratios of correlation functions being high enough for good repeatability.
A powerful calculation capability of the BeNano 90 Zeta allows the small particles to have sufficient signals for the correlation function to supply reliable results within a very short time.
The results that were gathered from the various measurements are listed below. The particle size of lysozyme was measured to be between 3 and 4 nm, as shown in Table 2. Using the Mark-Houwink equation, using the K and α constants of lysozyme, the molecular weight of lysozyme can be calculated.
Table 2. Particle sizes of lysozyme at different concentrations at 25℃. Source: Bettersize Instruments
| Concentration |
Z-ave (nm) |
Calculated Molecular
Weight (Da) |
| 10 mg/mL |
3.47±0.31 |
10.3 K |
| 5 mg/mL |
3.79±0.90 |
12.4 K |
| 30 mg/mL |
3.63±0.22 |
12.6 K |
.jpg)
Figure 5. Size distribution of 30 mg/mL lysozyme at 25℃. Image Credit: Bettersize Instruments
.jpg)
Figure 6. Size distribution of 30 mg/mL lysozyme at 45℃. Image Credit: Bettersize Instruments
.jpg)
Figure 7. Size distribution of 30 mg/mL lysozyme at 56℃. Image Credit: Bettersize Instruments
.jpg)
Figure 8. Size distributions of 30 mg/mL lysozyme at different temperatures. Image Credit: Bettersize Instruments
It shows at the concentration of 30 mg/mL that the calculated molecular weight of lysozyme (12.6 KDa) was very near to the theoretical value (14.4 KDa) of lysozyme.
The particle size of lysozyme was small at first and had a narrow distribution at room temperature (25°C), as can be seen in Figures 5-8. Large lysozyme aggregates were formed because of protein denaturation at high temperatures.
Figures 9 and 10 demonstrate that the Z-average mean and scattering intensity of lysozyme were stable at multiple temperatures below 50°C, and they increased dramatically when the temperature went higher than 55°C.
.jpg)
Figure 9. Z-average sizes of 30 mg/mL lysozyme at different temperatures. Image Credit: Bettersize Instruments
.jpg)
Figure 10. Scattering intensities of 30 mg/mL lysozyme at different temperatures. Image Credit: Bettersize Instruments
This is because of a structural alteration caused by the denaturation of lysozyme at high temperatures, resulting in the generation of a large number of aggregates.
Conclusion
In this study using the BeNano 90 Zeta, at the concentration of 30 mg/mLm the particle size of lysozyme was measured to be 3.63 nm ± 0.22 nm and the molecular weight of lysozyme was calculated to be 12.6 KDa through the empirical Mark-Houwink equation.
By using the precise temperature-control system of the BeNano 90 Zeta, the study on lysozyme denaturation at high temperatures was performed successfully.
Acknowledgments
Produced from materials originally authored by Zhibin Guo, Shelly Zhang and Hui Ning from Bettersize Instruments.
About Bettersize Instruments

With over 25 years experience developing and manufacturing particle characterization instruments, Bettersize has introduced breakthrough technology in the field of particle size & shape measurement.
By achieving high quality and superior performance, our instruments provide precise analysis results of particle size, particle shape, and powder characteristics, helping scientists and engineers to understand material properties, facilitate research and improve production efficiency.
Bettersize product line for particle size and shape analysis includes instruments of all needs and budgets, from basic to advanced research models. These instruments are widely applied in Pharmaceuticals, Battery materials, Mining and minerals, Metals, Chemicals and Surface coatings, measuring materials with size ranges from nanometer to millimeter.
Focused on technology innovation, instruments manufacturing, application support and after-sales services, Bettersize provides expertise and professional solutions and assures customers the highest confidence in our products.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.