Isolating nuclei is an essential step in both single nuclear RNA-sequencing and ATAC-sequencing workflows.
It is important to ensure that samples obtained during library preparation are non-clustered and debris-free, but nuclei isolated from tissue samples in areas like the brain are prone to high debris in comparison with samples obtained from cultured cells.
It is also important that nuclei are accurately counted for single cell analysis, even in instances where debris does not impact upon library preparation.
The CellDrop™ FL Automated Cell Counter’s dual fluorescence counting capabilities can clearly distinguish debris, nuclei and unlysed intact cells via acridine orange/propidium iodide (AO/PI) counting.
Eliminate Costs | Automate Cell Counting
Methods
The 10x Genomics® protocol “Nuclei Isolation from Embryonic Mouse Brain for Single Cell Multiome ATAC + Gene Expression Sequencing. CG000366 Rev B” was used to isolate nuclei from mouse embryonic brains.
Minimization of large clusters is critical for the downstream workflow of single-cell sequencing because these have the potential to clog fluidic chips, leading to failed sequencing experiments or a reduction in library quality.
Nuclei suspensions should be filtered to remove large clusters, and it is recommended to refer to the manufacturer’s protocol in cases where large clusters of nuclei are to be observed. It is also important to remove any intact cells that did not lyse during the procedure.
Single cell sequencing procedures like those employed by 10x Genomics® are reliant on properly isolated nuclei if the technology is to appropriately detect expression differences within a cellular population.
A large number of applications (such as ATAC-seq) will only work properly with intact nuclei. The sample volume for these methods is often a limiting factor, so it can be beneficial to use a single analysis volume for multiple quality control purposes.
CellDrop Series Automated Cell Counters’ unique DirectPipette™ Technology facilitates counting without the need for disposable slides, while the variable chamber volume enables counting volumes between 5 and 40 µL of sample.
The CellDrop is also able to accommodate common reusable or disposable plastic slides, meaning that the user can quantify nuclear isolation on the CellDrop before transferring the same slide to a higher magnification microscope for nuclear integrity analysis.
It is possible to measure the success of nuclei isolation via cell viability applications. The AO/PI dye combination stains live cells during traditional cell viability testing, prompting live cells to fluoresce green and dead cells to fluoresce red.
In this specific application, however, the stain will label successfully isolated nuclei red while any remaining intact cells are labeled green.
This process allows the user to calculate the number of residual intact cells that carry over as a percentage of total counted in order to confirm that the experimental workflow can proceed.
As well as cell count, the CellDrop is capable of measuring cell viability using either acridine orange/propidium iodide (AO/PI) or trypan blue.
Isolated nuclei in the examples presented here were measured from mouse brain using both AO/PI and trypan blue, utilizing the settings presented in Tables 1 and 2.
Table 1. Recommended Settings for Counting Isolated Nuclei. Source: DeNovix Inc.
| . |
| Count Application |
AO/PI |
| Chamber Height |
100 µm |
| Dilution Factor |
2 |
| Diameter (min) |
4 µm |
| Diameter (max) |
20 µm |
| Live Roundness |
1 |
| Dead Roundness |
1 |
| Green Fluorescence Threshold |
10 |
| Red Fluorescence Threshold |
1 |
Table 2. Recommended Settings for Counting Isolated Nuclei Using Trypan Blue. Source: DeNovix Inc.
| . |
| Count Application |
Trypan Blue |
| Chamber Height |
100 µm |
| Dilution Factor |
2 |
| Diameter (min) |
6 µm |
| Diameter (max) |
30 µm |
| Live Roundness |
50 |
| Dead Roundness |
15 |
| Stained Threshold |
15 |
Results
Figure 1 displays the results of cell counting and viability assessment. Viability using trypan blue was found to be 54%, while viability in AO/PI was found to be 1.8%, significantly lower than that of trypan blue.
Observing the samples using a microscope revealed that most were stained with trypan blue (Figure 2), indicating the presence of non-viable cells.
Manual inspection of CellDrop trypan blue images showed that where debris resembles live cells, this may be counted erroneously. Some nuclei were not counted (Figure 3).
AO/PI is specific for live/dead cells and intact nuclei, meaning that this does not stain debris. It can be clearly seen in these examples that trypan blue is prone to underestimating total cell count and overestimating live cell number in comparison with AO/PI.
Differentiation of debris and cells can be achieved using the two fluorophores investigated in this study. The nuclear pore complex can facilitate passive diffusion up to 30-60 kDa.
Here, both AO and PI (~0.6 kDa) are able to freely pass into the nucleus, displaying a red signal due to a FRET interaction between these two fluorophores.
While both fluorophores are able to pass into the nucleus easily, only AO is able to diffuse across an intact cell membrane. This fluorophore highlights live, intact cells as green while simultaneously labeling successfully isolated nuclei as red.
It is important to minimize the number of intact cells in isolation. Accurately enumerating intact cells with AO/PI can enhance quality control while improving consistency of results in downstream workflows.
Because downstream workflows must be highly precise, the use of well characterized dyes such as AO and PI should be utilized alongside automated counting platforms able to improve the precision of cell counting.
This approach will help ensure accurate and reproducible results within labs, between labs and in future experiments with isolated nuclei.
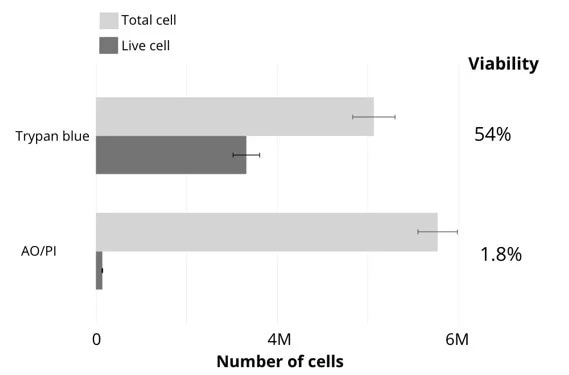
Figure 1. Comparison of viability counts using either brightfield (trypan blue) or fluorescence (acridine orange / propidium iodide) methods. Image Credit: DeNovix Inc.
Utilizing AO/PI fluorophores alongside the CellDrop Automated Cell Counter results in a significant improvement in comparison with automated systems using trypan blue or manual cell counts.
CellDrop offers users the ability to count cells without the need to use disposable plastic slides or to change the chamber height as required to accommodate these.
The instrument is also able to work with a wide dynamic range of cell concentrations, eliminating harsh conditions associated with constantly spinning and concentrating cells.
Overall, CellDrop offers a clear advantage over other automated cell counters when working with nuclei isolation protocols.
Figure 2. Automated image analysis of trypan blue stained cells. Image Credit: DeNovix Inc.
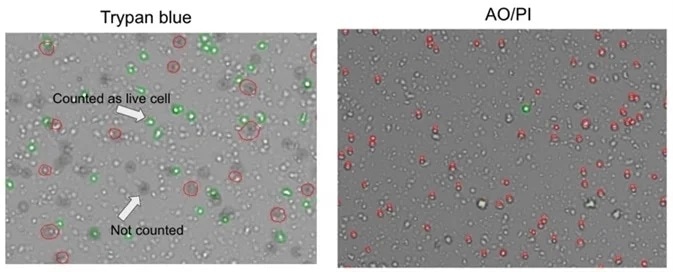
Figure 3. Comparison of trypan blue and acridine orange / propidium iodide methods for assessing cell viability. Image Credit: DeNovix Inc.
Acknowledgments
Produced from materials originally authored by Dr. Masakazu Ishikawa and Dr. Kazuo Yamashita, KOTAI Biotechnologies, Inc.
About DeNovix, Inc.
WELCOME TO DENOVIX
Award-Winning products for Life Science
Our multi-award winning products include the Reviewers’ Choice Life Science Product of the Year and Platinum Seal awarded- DS-11 Series Spectrophotometer / Fluorometer and CellDrop™ Automated Cell Counter. CellDrop is the first instrument of its kind to Count Cells Without Slides. These powerful instruments integrate patented DeNovix technology with easy-to-use software designed by life scientists for life scientists.
Researchers tell us they love the industry leading performance, smart-phone-like operation, and the flexible connectivity of the instruments. When support is needed, the DeNovix team is here to help. DeNovix received the prestigious Life Sciences Customer Service of the Year based on independent reviews posted by scientists worldwide!
CellDrop: Sustainable laboratory product of the year
The CellDrop Automated Cell Counter has been awarded Sustainable Laboratory Product of the Year in the SelectScience® Scientists’ Choice Awards®!
CellDrop’s patented DirectPipette™ technology distinguishes it as the only cell counter to eliminate the need for cell counting slides. This innovation saves millions of single-use plastic slides from use and disposal each year.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.