Sponsored Content by MaxCyte, Inc.Reviewed by Maria OsipovaAug 24 2023
The first trial to show that vaccination could confer HIV protection was the RV144 HIV vaccine trial. This trial reported an efficacy of 31%.
A key element of the RV144 trial was gp120, generated in Chinese Hamster Ovary (CHO) cells and deficient in N-linked glycosylation sites vital for binding several families of anti-HIV broadly neutralizing antibodies (bN-mAbs).
Researchers consider the absence of N-linked glycosylation to have possibly contributed to the modest efficacy of the vaccine.
Numerous epitopes bound by bN-mAbs have oligomannose terminal glycans, which are early intermediates in the N-linked glycosylation pathway.1
Dr. Berman’s research group at the University of California Santa Cruz showed that recombinant gp120 (rgp120) expressed by MaxCyte® electroporation of a HEK 293 cell line deficient in the N-acetylglucosaminyltransferase I enzyme (GnTI- 293; ATCC CRL-3022) led to epitopes mainly comprised of mannose-5 terminal glycans.2
These glycan moieties enhanced the binding of three major bN-mAb families, indicating that utilization of this rgp120 during vaccination may lead to increased efficacy. However, GnTI- 293 cells are inappropriate for large, clinical-scale bioproduction, hindering progression to in vivo efficacy studies.
CHO cells remain the gold standard for biotherapeutic development. A CHO cell line lacking alpha-1,3-mannosyl-glycoprotein 2-beta-N-acetylglucosaminyltransferase (MGAT1) would create proteins predominantly containing oligomannose glycans and could significantly enhance the production and purification of HIV gp120, possibly also enhancing vaccine efficacy.
Aim
The researchers aimed to produce a CHO MGAT1- cell line via CRISPR gene knockout that preserves growth properties appropriate for use in manufacturing proteins.
An additional aim was to transiently express rgp120 in the engineered CHO MGAT1- cell line and to characterize protein glycosylation patterns and bN-mAb binding.
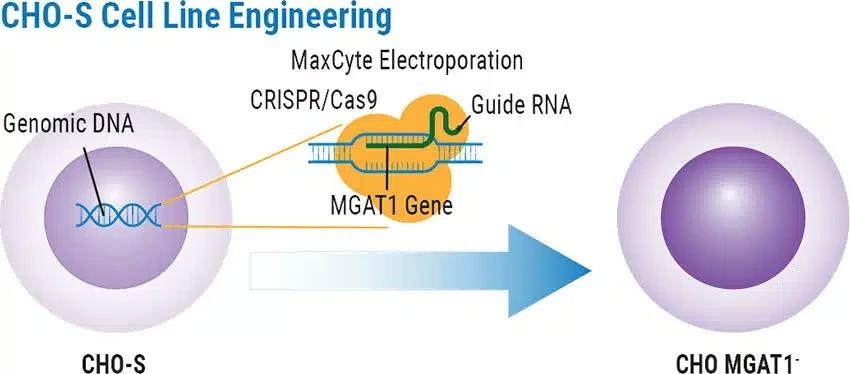
Image Credit: Adapted from Byrne et al (2018) under Creative Commons License Attribution 4.0 International (CC BY 4.0)
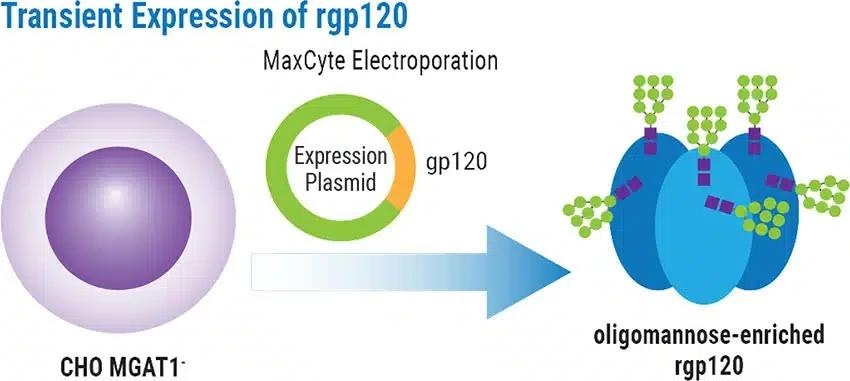
Image Credit: Adapted from Byrne et al (2018) under Creative Commons License Attribution 4.0 International (CC BY 4.0)
CHO electroporation
- For CHO MGAT1- construction, CHO-S cells were resuspended in MaxCyte Electroporation Buffer at 2 x 108 cells/mL
- CRISPR-Cas9, tracrRNA with guide sequence plasmid was added to a final concentration of 300 μg/mL
- For large-scale transient rgp120 production, an expression plasmid encoding the gp120 from the A244 HIV strain was electroporated into CHO MGAT1- cells
- Following electroporation, cells were seeded at 4 x 106 cells/mL in OptiCHO media and subsequently cultured in 125 mL Erlenmeyer shake flasks
- Sodium butyrate (1 mM) was added to the cultures 24 hours after electroporation and the temperature was reduced to 32 °C
- The complete methods for CHO MGAT1- development, screening and selection, in addition to glycosylation analysis and binding assays are presented in PLoS Biol, 16(8): e2005817, 2018.
Results
CRISPR delivery and generation of CHO MGAT1- cell line
CHO-S cells were electroporated with a plasmid encoding tracrRNA, CRISPR-Cas9, and a complete guide gRNA to target the MGAT1 gene.
CHO MGAT1- clones were firstly screened for staining utilizing fluorescein-labeled Galanthus nivalis lectin (GNA), which is a lectin that binds glycans with terminal mannose but not complex, sialic acid-containing glycans.
GNA bound to MGAT1- CHO but not the parental CHO-S cell line, as expected (Figure 1).
The four GNA-binding clones that demonstrated optimum growth rates were transiently transfected with a plasmid encoding A244-rgp120.
Secreted rgp120 underwent purification and subsequent analysis for overall titer and binding to the glycan-dependent bN-mAb PG9.
The chosen CHO MGAT1- cell line had rgp120 yields like the parental CHO-S line, in addition to comparable doubling times and the capability of growing at high density.
These characteristics were significantly better than the growth time, yield, and cell density restrictions of the GnTI- HEK 293 cell line employed in earlier studies.3 The chosen clone was sequenced to verify the disruption of the MGAT1 gene.
Identification of MGAT1- CHO cells via GNA binding of oligomannose
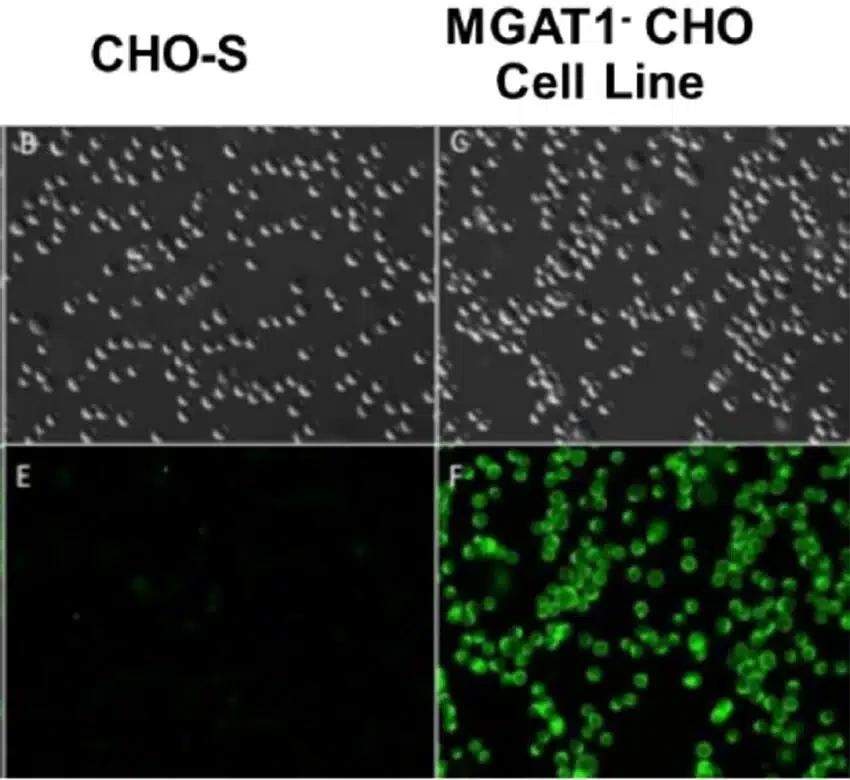
Figure 1. CHO-S parental cells or those electroporated with MGAT-targeting CRISPR machinery were incubated with fluorescein-labeled GNA to identify cells with surface proteins containing terminal mannose residues. Image Credit: Adapted from Byrne et al (2018) under Creative Commons License Attribution 4.0 International (CC BY 4.0)
Transient Expression of rgp120
A224-rgp120 transiently generated by the chosen MGAT1- CHO cell line or parental CHO-S cells was characterized using MALDI-TOF mass spectroscopy to analyze glycan composition. MGAT1-deficiency led to rgp120 containing more than 99% oligomannose glycans, compared to only 25% when generated in CHO-S cells (Figure 2).
The MGAT1-deficiency led to homogeneity of glycosylation, which positively affected purification. The rgp120 utilized in the RV144 HIV clinical trial exhibited vast net charge heterogeneity that significantly reduced the yield and increased the manufacturing complexity.
The rgp120 generated by MGAT1-deficient cells was secreted at high titers with homogeneous glycosylation, enabling purification with more conventional chromatography.
MGAT1-deficient CHO cells create rgp120 containing more than 99% oligomannose glycans
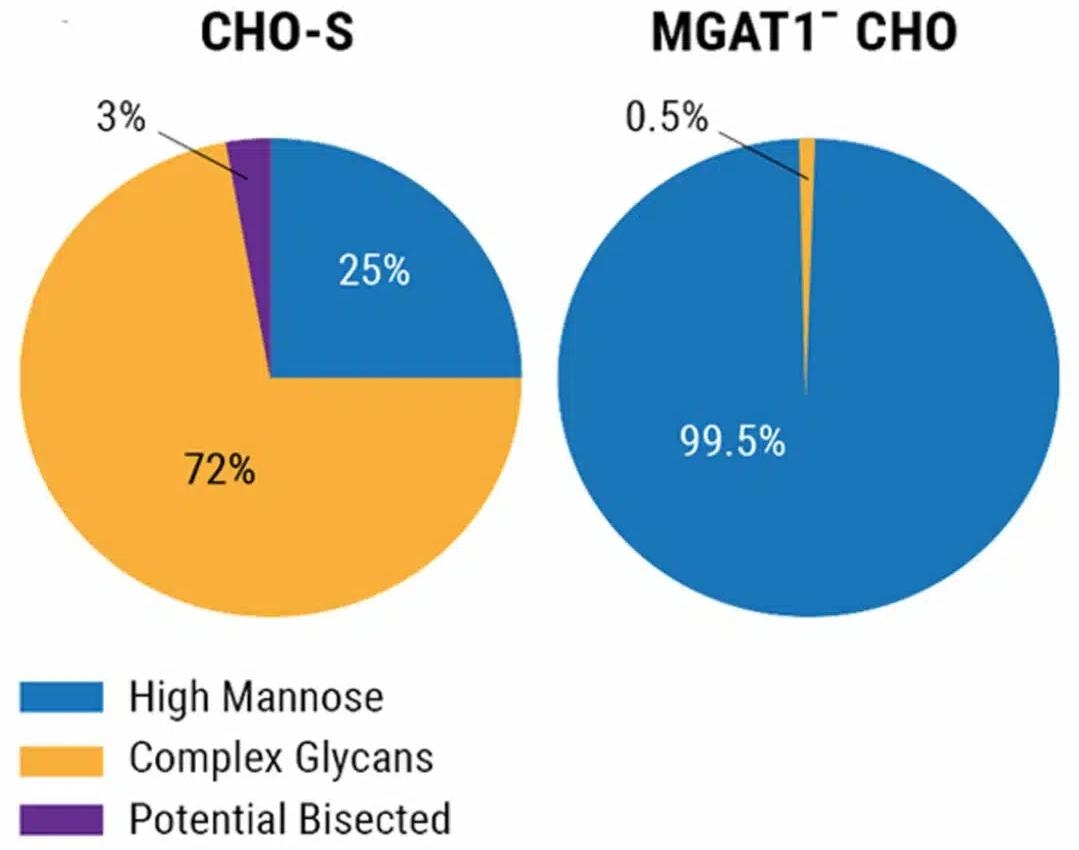
Figure 2. Purified rgp120 from the selected CHO MGAT1- cell line or the parental CHO-S cell line were analyzed via MALDI-TOF mass spectroscopy. Image Credit: Adapted from Byrne et al (2018) under Creative Commons License Attribution 4.0 International (CC BY 4.0)
MGAT1-deficiency generates rgp120 with enhanced binding to multiple bN-mAbs
The binding of rgp120 generated following MaxCyte electroporation of MGAT1- CHO or CHO-S cells to a panel of bN-mAbs was examined using fluorescence immunoassay.3 With the exception of a single antibody, all had modest to significant improvements in binding to rgp120 generated in the MGAT-deficient cells (Figure 3).
These results indicate that modifying glycosylation while preserving the amino acid sequence can positively affect bN-mAb binding. Assessment of the impact of oligomannose engineering on both in vivo immunogenicity and vaccine efficacy are crucial next steps.
Improved binding of V1/V2 and V3 domain bN-mAbs to rgp120 generated in MGAT1-deficient CHO cells
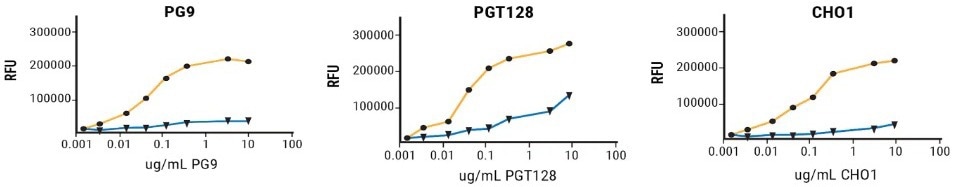
Figure 3. A224-rgp120 purified from CHO-S or MGAT1- CHO cells were coated onto microtiter plates and binding of a panel antibodies with broadly neutralizing capacity examined via FIA. Image Credit: Adapted from Byrne et al (2018) under Creative Commons License Attribution 4.0 International (CC BY 4.0)
Conclusion
The high-performance cell engineering technology of MaxCyte delivered the CRISPR-Cas9 machinery efficiently while maintaining CHO-S cell viability, facilitating the rapid production of an MGAT1-deficient CHO cell line.
The established MGAT1- CHO cell line grew robustly at high density in suspension in serum-free medium. It generated rgp120 at high titers upon transient transfection.
The rgp120 generated with the newly engineered MGAT1- CHO cells contained the desired early oligomannose glycans. These oligomannose glycans improved the binding of key bN-mAbs. This cell line promises to be a vital tool in the clinical translation of more effective HIV vaccines.
References and further reading
- High-mannose Glycan-dependent Epitopes Are Frequently Targeted in Broad Neutralizing Antibody Responses during HIV-1 Infection. (2012) J Virol, 86(4):2153-2164.
- Glycan Modification to the gp120 Immunogens Used in the RV144 Vaccine Trial Improve Binding to Broadly Neutralizing Antibodies. (2018) PLoS ONE, 13(4):e0196370.
- CRISPR/Cas9 Gene Editing for the Creation of an MGAT1-deficient CHO Cell Line to Control HIV-1 Vaccine Glycosylation. (2018) PLoS Biol, 16(8): e2005817.
About MaxCyte, Inc.
MaxCyte is a leading commercial cell-engineering company focused on providing enabling platform technologies to advance innovative cell-based research as well as next-generation cell therapy discovery, development and commercialization. Over the past 20 years, we have developed and commercialized our proprietary Flow Electroporation® platform, which facilitates complex engineering of a wide variety of cells.
Our ExPERT™ platform, which is based on our Flow Electroporation technology, has been designed to support the rapidly expanding cell therapy market and can be utilized across the continuum of the high-growth cell therapy sector, from discovery and development through commercialization of next-generation, cell-based medicines. The ExPERT family of products includes: four instruments, the ATx™, STx™, GTx™, and VLx™; a portfolio of proprietary related processing assemblies or disposables; and software protocols, all supported by a robust worldwide intellectual property portfolio.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.