Diabetic kidney disease (DKD), the leading cause of kidney failure and end-stage renal disease globally, will affect almost half of all patients with type 2 diabetes. With the prevalence of type 2 diabetes increasing, early identification and management of DKD is critical in clinical practice.
This review provides a comprehensive clinical update on DKD in patients with type 2 diabetes, with an emphasis on novel treatment options.
Traditional DKD preventive and treatment measures, such as glycemic control and blood pressure management, have only minor benefits in reducing the decrease in glomerular filtration rate and progression to end-stage renal disease.
While cardiovascular outcome trials of SGLT-2i show a beneficial effect on various kidney disease-related endpoints, the effect of GLP-1 RA on kidney disease endpoints beyond decreased albuminuria has yet to be proven. Nonsteroidal mineralocorticoid receptor antagonists also have cardiovascular and renal protective properties.
With these novel medications and the prospect of further compounds in clinical development, doctors will be better equipped to customize DKD treatment in type 2 diabetic patients.
The current state of diabetes
According to the International Diabetes Federation, 537 million people (20-79 years old) worldwide were living with diabetes mellitus in 2021, with the figure anticipated to rise to more than 780 million by 2045.1
Approximately 90-95 % of them have type 2 diabetes (T2D).2,3 Nearly half of people with T2D develop diabetic kidney disease (DKD), formerly known as "diabetic nephropathy".4,5
DKD is the most prevalent cause of kidney failure and end-stage kidney disease (ESKD), requiring kidney replacement therapy (dialysis or transplant) worldwide.6,7 Moreover, DKD is a major cause of cardiovascular disease and total mortality among diabetics.8,9
Given the rising prevalence of T2D, early identification and treatment of DKD are critical therapeutic considerations. This review offers an update on DKD pathogenesis, clinical symptoms, and current advances in DKD therapy.
Pathophysiology
Multiple diabetes-related mechanisms, including hyperglycemia and accompanying metabolic abnormalities, glomerular hemodynamic alterations, and proinflammatory and profibrotic factors, all contribute to kidney damage in DKD.10-13
These pathways often result in glomerular hyperfiltration and hypertrophy, and evidence shows that this might progress to sclerosis, especially in the presence of concomitant hypertension.11
Obesity and systemic hypertension, which are frequent among T2D patients, increase glomerular hyperfiltration.14
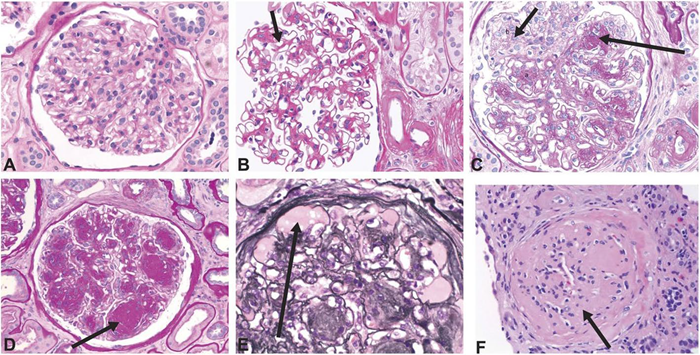
Fig 1. Histology images showing structural changes related to diabetic glomerulopathy. A Normal glomerulus. B Diffuse mesangial expansion with mesangial cell proliferation. C Prominent mesangial expansion with early nodularity and mesangiolysis. D Accumulation of mesangial matrix forming Kimmelstiel-Wilson nodules. E Dilation of capillaries forming microaneurysms, with subintimal hyaline (plasmatic insudation). F Obsolescent glomerulus. A–D and F were stained with period acid-Schiff stain. E was stained with Jones stain. Original magnification ×400. Reprinted with permission from American Society of Nephrology. Image Credit: Alicic, R.Z., et al. (2017)11
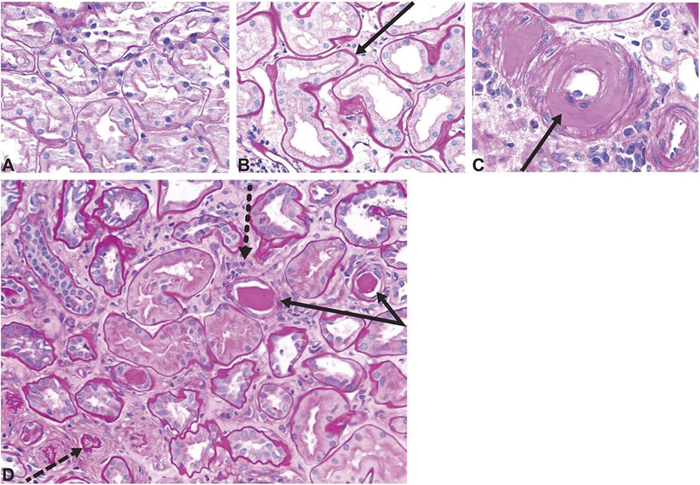
Fig 2. Histology images showing tubulointerstitial changes seen in diabetic kidney disease. A Normal kidney cortex. B Thickened tubular basement membrane and interstitial widening. C Arteriole with an intimal accumulation of hyaline material with significant luminal compromise. D Renal tubules and interstitium in advancing diabetic kidney disease, with thickening and wrinkled tubular basement membranes (solid arrows), atrophic tubules (dashed arrow), some containing casts, and interstitial widening with fibrosis and inflammatory cells (dotted arrow). All sections stained with period acid-Schiff stain, original magnification ×200. Reprinted with permission from American Society of Nephrology. Image Credit: Alicic, R.Z., et al. (2017)11
Arteriolar hyalinosis, tubulointerstitial inflammation, and fibrosis are also prominent hallmarks of DKD (Figures 1 and 2).11
Progressive glomerular damage increases albumin permeability, leading to elevated albuminuria.15 Albuminuria typically precedes filtration loss; in DKD, eGFR reduction can occur without albuminuria.16-18
People with reduced eGFR but no albuminuria often have extensive vascular lesions and interstitial fibrosis in their kidney tissue.18 Table 1 describes the usual results of glomerular lesion biopsies in people with DKD.
Clinical manifestations
DKD frequently escalates to renal failure or cardiovascular events, resulting in mortality in almost half of those affected.11,20 As a result, early recognition, identification, and intervention are critical to improving clinical outcomes.
Diagnostic tools and laboratory practices for DKD
A chronic rise in urine albumin-to-creatinine ratio (UACR, ≥ 30mg/g [≥ 3 mg/mmol]) and/or a persistent decline in eGFR (< 60 mL/min/1.73 m2) in a diabetic patient are key indicators of DKD.21 To qualify as DKD, these lesions must be caused solely by diabetes-related causes.21
The American Diabetes Association (ADA) Standards of Medical Care recommend that persons with T2D be checked for DKD at the time of diagnosis and then once a year after that.21 As shown in Figure 3, there are three types of albuminuria.22
- Stage A1, normal to mildly increased albuminuria: < 30 mg/g (< 3 mg/mmol) UACR in urine sample
- Stage A2, moderately increased albuminuria, microalbuminuria: 30-300 mg/g (3-30 mg/mmol) UACR; occurring ≥ 2 times, 3-6 months apart.21 This low-grade albuminuria is a less effective predictor of disease progression than macroalbuminuria.23
- Stage A3, severely increased albuminuria, macroalbuminuria: > 300 mg/g (> 30 mg/mmol) UACR; occurring ≥ 2 times, 3-6 months apart.21
The Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation is the most widely used formula for estimating GFR from serum creatinine.
Recently, the American Society of Nephrology and the National Kidney Foundation recommended using race-agnostic approaches to diagnose and classify chronic kidney disease as a step toward more equitable healthcare.24,25
A significant development is the updated CKD-EPI 2021 eGFR equation. This new equation does not contain a word for race, with the goal of raising awareness of chronic kidney disease and encouraging more timely identification and treatment interventions for all populations.
The addition of serum cystatin-C to the CKD-EPI 2021 eGFR equation enhances both accuracy and precision.25 Although the blood cystatin-C test is accessible in some parts of the world, it is not frequently utilized due to high prices and a lack of assay standardization.26-29
In both general and high-risk groups, albuminuria and lower eGFR are related to higher risks of cardiovascular events and death, as well as all-cause mortality.30,31
To assess renal and cardiovascular risks, individuals with diabetes and UACR > 30 mg/g (> 3 mg/mmol) and/or eGFR < 60 mL/min/1.73 m2 should have these tests at least twice a year.21
In addition to monitoring kidney damage and function, people with T2D should have their glycated hemoglobin (HbA1c) checked every 3-6 months to ensure proper blood glucose management.32
Table 1. Overview of classes and biopsy findings seen in glomerular lesions associated with diabetic kidney disease (DKD). Source: Tervaert, T.W.C., et al. (2010)
| Class |
Biopsy findings |
| I |
Thickening of glomerular basement membrane > 430 nm in males ages 9 years and older, > 395 nm in females ages 9 years and older |
| II |
Mild to severe expansion of mesangial extracellular material: width of interspace exceeds two mesangial cell nuclei in two or more glomerular lobules; also known as “diffuse diabetic glomerulosclerosis.” |
| III |
Nodular sclerosis, Kimmelstiel-Wilson lesions: focal, lobular, mesangial lesions with acellular, hyaline/matrix core. Generally, these lesions indicate a transition from early to later stages of diabetic kidney disease |
| IV |
More than 50 % global glomerulosclerosis is attributed to diabetes: fibrotic lesions with a build-up of extracellular matrix proteins in the mesangial space. Presence indicates advanced diabetic kidney disease |
| Other changes, lesions |
Interstitial fibrosis and tubular atrophy; hyalinosis of the efferent, and possibly the afferent, arterioles; insudative lesions known as “capsular drop lesions” when found in Bowman’s capsule, as “hyalinized afferent and efferent arterioles when found in the afferent and efferent arterioles, and as fibrin cap lesions or hyalinosis when found in glomerular capillaries; “tip lesion” refers to abnormality in the tubuloglomerular junction, with atrophic tubules and no visible glomerular opening, and related to advanced DKD and macroalbuminuria |
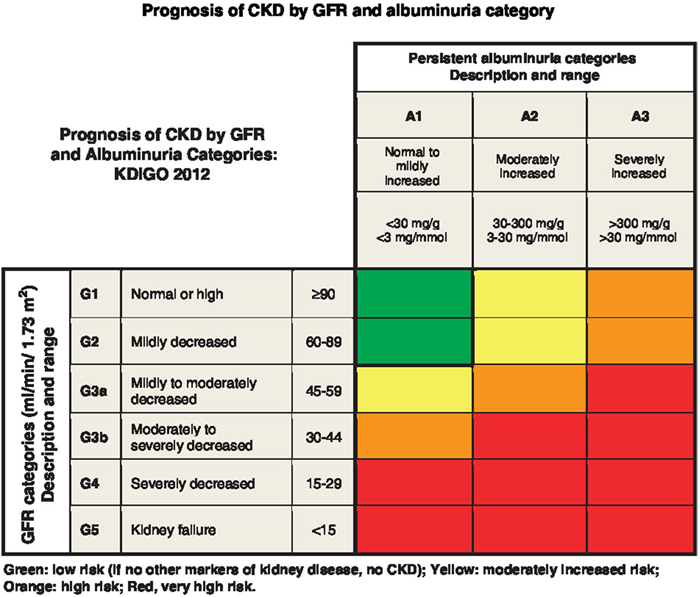
Fig 3. Prognosis of chronic kidney disease by GFR and albuminuria category. This figure was developed by Kidney Disease Improving Global Outcomes (KDIGO) and reproduced with permission from KDIGO. Image Credit: KDIGO (2012)22
The ADA suggests that patients with T2D engage with their physician to develop a personalized objective for glycemic management, avoiding hypoglycemia while maintaining a general aim of HbA1c < 7 % (53 mmol/mol).32
Acknowledgments
Produced from materials originally authored by Thomas Forst, Chantal Mathieu, Francesco Giorgino, David C. Wheeler, Nikolaos Papanas, Roland E. Schmieder, Atef Halabi, Oliver Schnell, Marina Streckbein, and Katherine R. Tuttle from hVIVO.
References
- International Diabetes Federation. IDF Diabetes Atlas. https://diabetesat las.org/2021. Accessed 30 Aug 2022.
- Xu, G., et al. (2018). Prevalence of diagnosed type 1 and type 2 diabetes among US adults in 2016 and 2017: population based study. BMJ, 362(1), p.k1497. DOI: 10.1136/bmj.k1497. https://www.bmj.com/content/362/bmj.k1497.abstract.
- World Health Organization (2024). Diabetes. (online) World Health Organization. Available at: https://www.who.int/news-room/fact-sheets/detail/diabetes.
- Thomas, M.C., Cooper, M.E. and Zimmet, P. (2015). Changing epidemiology of type 2 diabetes mellitus and associated chronic kidney disease. Nature Reviews Nephrology, 12(2), pp.73–81. DOI: 10.1038/nrneph.2015.173. https://www.nature.com/articles/nrneph.2015.173.
- Gheith, O., et al. (2015). Diabetic kidney disease: world wide difference of prevalence and risk factors. Journal of Nephropharmacology, (online) 5(1), p.49. Available at: https://pmc.ncbi.nlm.nih.gov/articles/PMC5297507/.
- Fu, H., et al. (2019). Diabetic kidney diseases revisited: A new perspective for a new era. Molecular Metabolism, (online) 30, pp.250–263. DOI: 10.1016/j.molmet.2019.10.005. https://www.sciencedirect.com/science/article/pii/S2212877819309263.
- Li, H., et al. (2020). Changing epidemiology of chronic kidney disease as a result of type 2 diabetes mellitus from 1990 to 2017: Estimates from Global Burden of Disease 2017. Journal of Diabetes Investigation, 12(3). doi: 10.1111/jdi.13355. https://onlinelibrary.wiley.com/doi/full/10.1111/jdi.13355.
- Górriz, J.L., et al. (2020). GLP-1 Receptor Agonists and Diabetic Kidney Disease: A Call of Attention to Nephrologists. Journal of Clinical Medicine, 9(4), p.947. DOI: 10.3390/jcm9040947. https://www.mdpi.com/2077-0383/9/4/947.
- Rawshani, A., et al. (2018). Risk Factors, Mortality, and Cardiovascular Outcomes in Patients with Type 2 Diabetes. New England Journal of Medicine, 379(7), pp.633–644. DOI: 10.1056/nejmoa1800256. https://www.nejm.org/doi/full/10.1056/NEJMoa1800256
- Alicic, R.Z., Johnson, E.J. and Tuttle, K.R. (2018). Inflammatory Mechanisms as New Biomarkers and Therapeutic Targets for Diabetic Kidney Disease. Advances in Chronic Kidney Disease, 25(2), pp.181–191. DOI: 10.1053/j.ackd.2017.12.002. https://www.akdh.org/article/S1548-5595(17)30215-X/abstract.
- Alicic, R.Z., Rooney, M.T. and Tuttle, K.R. (2017). Diabetic Kidney Disease. Clinical Journal of the American Society of Nephrology, (online) 12(12), pp.2032–2045. DOI: 10.2215/cjn.11491116. https://journals.lww.com/cjasn/fulltext/2017/12000/diabetic_kidney_disease__challenges,_progress,_and.17.aspx.
- Pichler, R., et al. (2017). Immunity and inflammation in diabetic kidney disease: translating mechanisms to biomarkers and treatment targets. American Journal of Physiology-Renal Physiology, 312(4), pp.F716–F731. DOI: 10.1152/ajprenal.00314.2016. https://journals.physiology.org/doi/full/10.1152/ajprenal.00314.2016.
- Cappelli C, et al. (2020). The TGF-β profibrotic cascade targets ecto-5’-nucleotidase gene in proximal tubule epithelial cells and is a traceable marker of progressive diabetic kidney disease. Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease. DOI: 10.1016/j.bbadis.2020.165796. https://www.sciencedirect.com/science/article/pii/S0925443920301411.
- Chagnac, A., et al. (2008). Obesity-induced glomerular hyperfiltration: its involvement in the pathogenesis of tubular sodium reabsorption. Nephrology Dialysis Transplantation, 23(12), pp.3946–3952. DOI: 10.1093/ndt/gfn379. https://academic.oup.com/ndt/article-abstract/23/12/3946/1885676.
- Benzing, T. and Salant, D. (2021). Insights into Glomerular Filtration and Albuminuria. New England Journal of Medicine, (online) 384(15), pp.1437–1446. DOI: 10.1056/nejmra1808786. https://www.nejm.org/doi/abs/10.1056/NEJMra1808786.
- Penno, G., et al. (2011). Clinical significance of nonalbuminuric renal impairment in type 2 diabetes. Journal of Hypertension, 29(9), pp.1802–1809. DOI: 10.1097/hjh.0b013e3283495cd6. https://journals.lww.com/jhypertension/abstract/2011/09000/clinical_significance_of_nonalbuminuric_renal.19.aspx.
- Dwyer, J.P., et al. (2012). Renal Dysfunction in the Presence of Normoalbuminuria in Type 2 Diabetes: Results from the DEMAND Study. Cardiorenal Medicine, (online) 2(1), pp.1–10. DOI: 10.1159/000333249. https://karger.com/crm/article-abstract/2/1/1/97278/Renal-Dysfunction-in-the-Presence-of.
- Deng, L., Li, W. and Xu, G. (2021). Update on pathogenesis and diagnosis flow of normoalbuminuric diabetes with renal insufficiency. European Journal of Medical Research, 26(1). DOI: 10.1186/s40001-021-00612-9. https://link.springer.com/article/10.1186/s40001-021-00612-9.
- Tervaert, T.W.C., et al. (2010). Pathologic Classification of Diabetic Nephropathy. Journal of the American Society of Nephrology, 21(4), pp.556–563. DOI: 10.1681/asn.2010010010. https://journals.lww.com/jasn/abstract/2010/04000/pathologic_classification_of_diabetic_nephropathy.7.aspx.
- Ballew, S.H. and Matsushita, K. (2018). Cardiovascular Risk Prediction in CKD. Seminars in Nephrology, 38(3), pp.208–216. DOI: 10.1016/j.semnephrol.2018.02.002. https://www.seminarsinnephrology.org/article/S0270-9295(18)30023-8/abstract.
- American Diabetes Association (2019). 11. Microvascular Complications and Foot Care: Standards of Medical Care in Diabetes−2020. Diabetes Care, 43(Supplement 1), pp.S135–S151. DOI: 10.2337/dc20-s011. https://diabetesjournals.org/care/article/43/Supplement_1/S135/30443/11-Microvascular-Complications-and-Foot-Care.
- Kidney Disease: Improving Global Outcomes Diabetes Work Group. KDIGO 2012 clinical practice guideline for the evaluation and management of chronic kidney disease. Kidney Int Suppl. 2013;3:1–150.
- Perkins, B.A., et al. (2007). Microalbuminuria and the risk for early progressive renal function decline in type 1 diabetes. Journal of the American Society of Nephrology: JASN, (online) 18(4), pp.1353–1361. DOI: 10.1681/ASN.2006080872. https://journals.lww.com/jasn/abstract/2007/04000/microalbuminuria_and_the_risk_for_early.39.aspx.
- Delgado, C., et al. (2021). A Unifying Approach for GFR Estimation: Recommendations of the NKF-ASN Task Force on Reassessing the Inclusion of Race in Diagnosing Kidney Disease. Journal of the American Society of Nephrology, 32(12), pp.2994–3015. DOI: 10.1681/asn.2021070988. https://journals.lww.com/jasn/fulltext/2021/12000/a_unifying_approach_for_gfr_estimation_.9.aspx.
- Williams, W.W., Hogan, J.W. and Ingelfinger, J.R. (2021). Time to Eliminate Health Care Disparities in the Estimation of Kidney Function. New England Journal of Medicine, 385(19), pp.1804–1806. DOI: 10.1056/nejme2114918. https://www.nejm.org/doi/full/10.1056/NEJMe2114918.
- Inker, L.A., et al. (2021). New Creatinine- and Cystatin C-Based Equations to Estimate GFR without Race. The New England Journal of Medicine, (online) 385(19), pp.1737–1749. DOI: 10.1056/NEJMoa2102953. https://www.nejm.org/doi/full/10.1056/NEJMoa2102953.
- KDIGO (2020). KDIGO 2020 clinical practice guideline for diabetes management in chronic kidney disease. Kidney International, 98(4), pp.S1–S115. DOI: 10.1016/j.kint.2020.06.019. https://www.kidney-international.org/article/S0085-2538(20)30718-3/fulltext.
- Chen, D.C., et al. (2022). Association of Intraindividual Difference in Estimated Glomerular Filtration Rate by Creatinine vs Cystatin C and End-stage Kidney Disease and Mortality. JAMA Network Open, (online) 5(2), p.e2148940. DOI: 10.1001/jamanetworkopen.2021.48940. https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2789130.
- Bargnoux, A.-S., et al. (2017). Multicenter Evaluation of Cystatin C Measurement after Assay Standardization. Clinical Chemistry, 63(4), pp.833–841. DOI: 10.1373/clinchem.2016.264325. https://academic.oup.com/clinchem/article-abstract/63/4/833/5613009.
- Hemmelgarn, B.R. (2010). Relation Between Kidney Function, Proteinuria, and Adverse Outcomes. JAMA, 303(5), p.423. DOI: 10.1001/jama.2010.39. https://jamanetwork.com/journals/jama/fullarticle/185313.
- Matsushita, K., et al. (2010). Association of estimated glomerular filtration rate and albuminuria with all-cause and cardiovascular mortality in general population cohorts: a collaborative meta-analysis. Lancet (London, England), (online) 375(9731), pp.2073–81. DOI: 10.1016/S0140-6736(10)60674-5. https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(10)60674-5/abstract.
- American Diabetes Association (2019). Glycemic targets: Standards of medical care in diabetes - 2020. Diabetes Care, 43(Supplement 1), pp.S66–S76. DOI: 10.2337/dc20-s006. https://diabetesjournals.org/care/article/43/Supplement_1/S66/30598/6-Glycemic-Targets-Standards-of-Medical-Care-in.
About hVIVO
hVIVO plc is a science‑led early‑phase drug development company purpose‑built to meet the growing complexity of modern clinical research. The Company operates an integrated early‑phase ecosystem that combines specialist clinical sites, advanced virology and immunology laboratories, human challenge expertise, and early drug development consulting. This unified model enables sponsors to generate rigorous, decision‑ready human data earlier in development, reducing uncertainty and accelerating progression through Phase I and II trials.
With industry‑leading capabilities in respiratory and infectious disease, alongside expanding expertise in cardiometabolic and other high‑growth therapeutic areas, hVIVO supports a diverse global client base that includes seven of the world’s ten largest biopharmaceutical companies. Its London quarantine facilities are the largest purpose‑built human challenge units in the world, complemented by additional early‑phase clinical capacity in Germany and a specialist consulting team providing strategic, regulatory, and biometry expertise.
The Company’s integrated approach delivers a seamless pathway from preclinical planning through early proof‑of‑concept, supported by continuous patient recruitment through FluCamp and a network of outpatient clinical sites for Phase II and III studies. By unifying scientific insight, operational control, and advanced laboratory capabilities, hVIVO provides sponsors with the clarity, speed, and reliability required to advance new medicines with confidence.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.