Organoids are three-dimensional tissue cultures able to replicate the structure and function of organs. They are created from stem cells, including adult, embryonic, or induced pluripotent stem cells (iPSCs).
They are grown in a supportive matrix, but it is also possible to develop organoids by coculturing epithelial progenitors with endothelial and mesenchymal cells.
Their 3D microenvironment allows organoids to accommodate both cell-cell and cell-matrix interactions that closely resemble interactions in native tissue. These capabilities have significant potential to impact the fields of cell therapy, personalized diagnostics, and drug discovery, and can aid in the modeling of human-specific aspects of development and disease.1
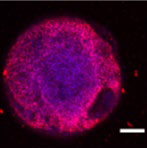
Figure 1. Embryoid Body (EB) Oct4 DAPI. Scale bar 1 mm. Image Credit: Scintica Instrumentation Inc.
Cartilaginous spheroids
Cartilaginous spheroids are multicellular three-dimensional constructs suitable for use in cartilage regeneration, tissue engineering, and as in vitro models for a wide range of studies. Cartilaginous microtissues are typically much larger than other spheroids.
Spheroid production has primarily depended on random self-assembly of spheroids within constrained containers. This approach offers only limited control over size and architectural features, however.
More automated and controlled biofabrication methods are required to increase the precision of cartilaginous spheroid assembly with specific sizes and architectural features. Thankfully, this is now possible via laser-assisted bioprinting.
Benefits of laser-assisted bioprinting
Bioprinting technologies have historically been employed in the production of multicellular spheroids, but these technologies face a number of drawbacks.
For example, extrusion-based bioprinting encapsulates spheroids in hydrogel bioink, which typically produces low spheroid densities and only offers limited control over spheroid localization within the final construct. This technology is also prone to nozzle clogging issues, especially when spheroids fuse into larger agglomerates before bioprinting.
Aspiration-based techniques have been developed to enable the picking up and depositing of single spheroids to address this issue, but this bioprinting method is still limited in throughput.
In contrast, laser-assisted bioprinting (LAB) is a nozzle-free technique able to deliver improved precision when manipulating cell suspensions at a single-cell resolution.
This nozzle-free spheroid bioprinting process helps to avoid the issues commonly associated with extrusion-based methods. LAB works by focusing a pulsed laser on a donor substrate coated with a metallic layer and bioink.
The resulting laser energy causes the metal to ablate, generating a plasma that creates a cavitation bubble. This bubble then collapses, resulting in a liquid jet that deposits bioink droplets containing a defined number of cells onto a receiver.
This approach helps to overcome the limitations of traditional methods by enabling precise spheroid deposition.
Proof of concept
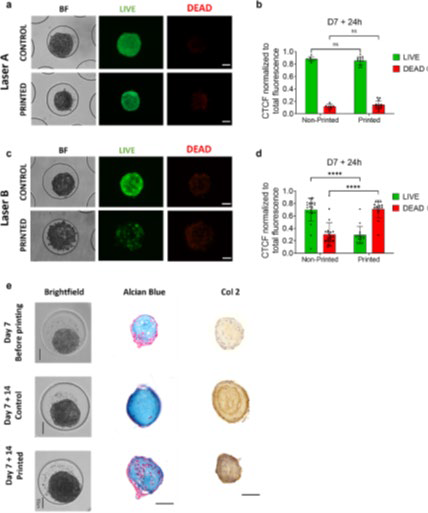
Figure 2. Spheroid laser-assisted bioprinting: Cell spheroids are imaged, targeted, and transferred by the laser to form a predesigned pattern. Image Credit: Hall, et al. (2024)1
Hall et al. were able to successfully bioprint cartilaginous spheroids formed from human periosteum-derived cells (hPDCs).1 These spheroids were shown to maintain high cell viability while also retaining their capacity for chondrogenic differentiation after printing (Figure 3).
It was also possible to bioprint smaller spheroids (100–150 μm in diameter) using the Laser-Induced Forward Transfer (LIFT) method. However, it proved difficult to print larger spheroids using this method.
A novel method, Laser-Induced Propulsion of Mesoscopic Objects (LIPMO), was implemented, enabling the bioprinting of larger spheroids up to 300 μm. High-density, multilayered spheroid populations were successfully fabricated by combining bioprinting with computer-aided image analysis.
Next Generation Bioprinting System (NGB-R) by Poietis™
Poietis’ Next Generation Bioprinting Systems (NGB-R) LAB system was used in this study. NGB-R is a multimodal, 3D bioprinting platform that has been designed and developed to print live organs and tissues.
The system combines extrusion and laser-assisted bioprinting, affording researchers the versatility of bioprinting a range of structures from single cells to 3D spheroids, while also allowing the use of a large number of biomaterials and hydrogels.
Integrated robotics and microscopes allow the entire bioprinting process to be automated, offering a robust combination of resolution and speed.

Figure 3. NGB-RTM Robotic-Assisted LAB System.
References and further reading
- Hall, G. N., et al. (2024). Laser-assisted bioprinting of targeted cartilaginous spheroids for high density bottom-up tissue engineering. Biofabrication, 16(4), 045029–045029. DOI: 10.1088/1758-5090/ad6e1a. https://iopscience.iop.org/article/10.1088/1758-5090/ad6e1a.
About Scintica Instrumentation Inc.
At Scintica, we advance science and medicine by supplying researchers with reliable research instrumentation and equipment. Our carefully selected portfolio of imaging systems, research tools, and supporting technologies is designed to reduce complexity and help scientists focus on what matters most, generating
meaningful results.
We partner closely with the preclinical research community to connect teams with solutions that are scientifically robust and built to support research challenges. From system selection through long-term support, our goal is to make research more productive, efficient, and impactful.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.