2. What should I know before I use ADYNOVATE?
Do not use if you have ever had an allergic reaction to ADYNOVATE, octocog alfa (a
medicine called ADVATE), or you are allergic to mouse or hamster proteins or any of
the ingredients listed at the end of the CMI. Talk to your doctor if you have or have
had any other medical conditions, if you have had or at risk of any heart problems,
if you take any other medicines, or if you are pregnant or plan to become pregnant
or if you are breastfeeding or plan to breastfeed. For more information, see Section
2. What should I know before I use ADYNOVATE? in the full CMI.
3. What if I am taking other medicines?
Tell your doctor or Haemophilia Treatment Centre if you are taking or using any other
medicines including any that you get without a prescription from your pharmacy, supermarket,
or health food shop. For more information, see Section
3. What if I am taking other medicines? in the full CMI.
4. How will I be given ADYNOVATE?
ADYNOVATE injection will be prepared and administered by a qualified healthcare professional
who is experienced in the care of patients with haemophilia. Some individuals may
be trained to use ADYNOVATE at home.
Your doctor will decide on your dose of ADYNOVATE depending on your condition and
body weight.
The frequency of infusions you receive, and how long you will use ADYNOVATE for, will
depend on how well ADYNOVATE is working for you. Your doctor may change the dose you
use during your treatment.
ADYNOVATE is given slowly by injection directly into your veins.
ADYNOVATE comes in a vial of drug powder and a diluent vial is also supplied. These
need to be mixed before use.
5. What should I know while using ADYNOVATE?
|
Things you should do
|
Tell your doctor of Haemophilia Treatment Centre immediately if you notice
- any sudden signs and symptoms of a severe, sudden allergic reaction.
- your bleeding is not controlled or worsens after using ADYNOVATE.
Tell any doctors, dentists, or pharmacists you visit that you are using ADYNOVATE
or if you are about to have any blood tests.
Keep all your appointments with your doctor and any blood tests.
|
|
Things you should not do
|
Do not give your medicine to anyone else, even if they appear to have the same condition
as you.
Do not stop using your medicine or change the dosage without checking with your doctor.
|
|
Looking after your medicine
|
Keep ADYNOVATE in the pack until it is time to use it so that it is protected from
light.
Keep ADYNOVATE in the refrigerator at 2°C to 8°C. Do not freeze.
|
6. Are there any side effects?
A very common side effect is Headache. Common side effects include dizziness, nausea,
diarrhoea, rash, hives. A serious side effect includes severe, sudden allergic reaction
with signs or symptoms of rash or hives, wheals, or generalised itching, swelling
of the face, lips, tongue or other parts of the body, shortness of breath, wheezing,
tightness or discomfort in the chest, and dizziness, which may progress to difficulty
in breathing, chest pain and fainting. For more information, including what to do
if you have any side effects, see Section
6. Are there any side effects? in the full CMI.
Active ingredient(s):
rurioctocog alfa pegol
Full Consumer Medicine Information (CMI)
This leaflet provides important information about using ADYNOVATE. You should also speak to your doctor or pharmacist if you would like further information
or if you have any concerns or questions about using ADYNOVATE.
Where to find information in this leaflet:
1. Why am I using ADYNOVATE?
ADYNOVATE contains the active ingredient rurioctocog alfa pegol (a PEGylated human
recombinant DNA derived blood clotting factor VIII).
ADYNOVATE is used for the management of congenital haemophilia A (an inherited bleeding
disorder caused by a lack of blood clotting factor VIII in the body).
ADYNOVATE is used to:
control and prevent bleeding episodes,
routinely prevent and reduce the frequency of bleeding episodes,
prevent or reduce bleeding before, during and after surgery.
The blood clotting factor VIII is essential for the blood to form clots and stop bleedings.
In patients with inherited haemophilia A, there is a low level of factor VIII in the
blood circulation.
ADYNOVATE is similar to the blood clotting factor VIII in human blood, and ADYNOVATE
works as a replacement therapy so that blood can form clots at the site of bleeding.
ADYNOVATE is produced by recombinant DNA technology and has been modified chemically
to prolong its duration of action.
This medicine helps to control your condition but does not cure it.
Ask your doctor if you have any questions about why this medicine has been prescribed
for you.
Your doctor may have prescribed it for another reason.
2. What should I know before I use ADYNOVATE?
Warnings
Do not use ADYNOVATE if:
you have ever had an allergic reaction to ADYNOVATE, or octocog alfa (a medicine called
ADVATE),
you are allergic to mouse or hamster proteins or if you have a known allergy to medicines
of mouse or hamster origin,
you are allergic to any of the ingredients listed at the end of this leaflet.
Always check the ingredients to make sure you can use this medicine.
Check with your doctor if you:
have or have had any other medical conditions.
have had, or at risk of, any heart problems or conditions involving the blood vessels.
are on a controlled sodium diet.
ADYNOVATE may increase the risk of abnormal blood clots forming in your body if you
have risk factors for developing blood clots.
During treatment, you may be at risk of developing certain side effects. It is important
you understand these risks and how to monitor for them. See additional information
under Section
6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant. There is
no information on the use of ADYNOVATE during pregnancy. Your doctor will discuss
the risks and benefits of using ADYNOVATE if you are pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed. It is not known
if ADYNOVATE passes into your milk and if it can harm your baby. Your doctor will
discuss the risks and benefits of using ADYNOVATE if you are breast-feeding.
3. What if I am taking other medicines?
Tell your doctor or Haemophilia Treatment Centre if you are taking any other medicines,
including any medicines, vitamins or supplements that you buy without a prescription
from your pharmacy, supermarket or health food shop.
There are no known interactions of ADYNOVATE with other medicines.
Your doctor or Haemophilia Treatment Centre have more information on medicines to
be careful with or avoid if you have concerns.
4. How do I use ADYNOVATE?
How much to use
Follow all directions given to you by your doctor carefully. They may differ from
the information contained in this leaflet.
Your doctor will decide how much ADYNOVATE you use.
Your dose will depend on:
- your body weight,
- the amount of factor VIII your body is able to make,
- how much, how often and where you are bleeding,
- if your body have built up antibodies to ADYNOVATE.
How to use ADYNOVATE
Treatment with ADYNOVATE will be started in a hospital or Haemophilia Treatment Centre
and supervised by your doctor who is experienced in the care of patients with haemophilia.
After starting ADYNOVATE treatment, some individuals may be trained to use ADYNOVATE
at home.
ADYNOVATE is given by slow injection directly into your vein.
Do not attempt to inject ADYNOVATE by yourself unless you have received proper training
by your doctor or Haemophilia Treatment Centre on how to use the product.
Preparing ADYNOVATE
ADYNOVATE is provided as a powder in a vial, and a diluent vial containing water for
injections is also supplied in the pack. These vials need to be mixed together to
form a clear solution before use.
Follow carefully the step-by-step instructions at the end of this leaflet or in the
pack insert on how to prepare and inject ADYNOVATE.
Do not mix ADYNOVATE with any other medicines or solvent other than the water for
injections diluent supplied with the pack.
Use only the reconstitution device provided with each pack to prepare the solution
for injection.
After mixing the powder and the diluent, use the solution immediately. If the solution
is not used straight way, you can keep the solution for a maximum of 3 hours when
stored at room temperature (below 30°C).
Do not refrigerate the solution after it is prepared.
ADYNOVATE is for single use in one patient only.
Dispose of all unused solution, empty vials, and used needles and syringes into a
sharps bin.
If you are unsure about how to prepare the medicine for use, contact your doctor or
Haemophilia Treatment Centre.
Inspecting ADYNOVATE
Always inspect ADYNOVATE before use and after it has been mixed.
After mixing, the solution should be clear to colourless, and free from foreign particles.
Do not inject the solution if it is discoloured, or cloudy, or contains particles.
How often to use ADYNOVATE
Your doctor will tell you how often or at what intervals you will receive the injection.
The frequency of injections you receive, and how long you will use ADYNOVATE for,
will depend on how well ADYNOVATE is working for you.
Continue using ADYNOVATE for as long as your doctor tells you. Usually, the replacement
therapy with ADYNOVATE is a life-long treatment.
If you forget to use ADYNOVATE
Do not inject a double dose to make up for the forgotten dose.
If you inject a double dose, this may increase the chance of you getting an unwanted
side effect.
If you are not sure what to do, ask your doctor or Haemophilia Treatment Centre.
If you use too much ADYNOVATE
If you think that you have used too much ADYNOVATE, you may need urgent medical attention.
You should immediately:
contact your doctor or Haemophilia Treatment Centre, or
go to the Emergency Department at your nearest hospital, or
phone the Poisons Information Centre by calling 13 11 26 (if you are in Australia), or by calling 0800 764 766 (if you are in New Zealand).
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using ADYNOVATE?
Things you should do
Tell you other doctors, dentists and pharmacists you are using this medicine.
If you are about to have any blood tests, tell your doctor that you are using ADYNOVATE.
Keep all your doctor's appointments so that your progress can be checked. Your doctor
may do some blood tests before you start your treatment and from time to time during
your treatment to monitor your progress.
Follow all instructions from your doctor. Your doctor may change the dose you use
using your treatment.
Tell your doctor and/or Haemophilia Treatment Centre straight away if you notice:
any sudden signs and symptoms of a severe allergic response.
your bleeding is not controlled or worsens after using ADYNOVATE.
Things you should not do:
Do not give your medicine to anyone else, even if they appear to have the same condition
as you.
Do not use ADYNOVATE to treat any other complaints unless your doctor tells you to.
Do not stop using ADYNOVATE unless advised by your doctor or healthcare professional
or unless you have an allergic reaction.
Do not change the dosage without checking with your doctor.
Do not use ADYNOVATE after the expiry date which is printed on the label after the
word 'EXP'. The expiry date refers to the last day of the month.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how ADYNOVATE
affects you.
ADYNOVATE is not expected to have an influence on your ability to drive and use machines.
Looking after your medicine
Keep ADYNOVATE in the pack until it is time to use it. This will protect the medicine
from light.
Keep ADYNOVATE in the refrigerator at 2°C to 8°C in a refrigerator. Do not freeze.
If necessary, you can keep ADYNOVATE out of the refrigerator for a single 3-month
period when stored in the original packaging in a cool dry place at room temperature
(below 30°C). The date that the product is removed from the refrigerator should be
recorded on the carton.
Do not return the medicine to the refrigerator after it has been stored at room temperature.
Do not use any ADYNOVATE that has been out of the refrigerator for more than 3 months.
Follow the instructions in the carton on how to take care of your medicine properly.
Keep ADYNOVATE out of reach of children.
Getting rid of any unwanted medicine
Medicines should not be disposed of via wastewater or household waste.
If your doctor tells you to stop using this medicine, or if the medicine is out of
date, or if the medicine has not been stored properly, ask your doctor or Haemophilia
Treatment Centre what to do with any unwanted medicine that is left over.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of
them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you
have any further questions about side effects.
Less serious side effects
Serious side effects
Your body can make antibodies (also called "inhibitors") against ADYNOVATE, which
may stop ADYNOVATE from working properly. Signs or symptoms may include easily bruising
or bleeding, swelling and pain or tightness in joints.
Tell your doctor, Haemophilia Treatment Centre, or pharmacist if you notice anything
else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
By reporting side effects, you can help provide more information on the safety of
this medicine.
Always make sure you speak to your doctor, Haemophilia Treatment Centre, or pharmacist
before you decide to stop using any of your medicines.
7. Product details
What ADYNOVATE contains
This medicine is only available with a doctor's prescription.
Powder in a vial
|
Active ingredient
(main ingredient)
|
rurioctocog alfa pegol
|
|
Other ingredients
(inactive ingredients)
|
calcium chloride dihydrate
glutathione
histidine
mannitol
polysorbate 80
sodium chloride
trehalose dihydrate
trometamol
|
Diluent in a vial
|
Other ingredients
(inactive ingredients)
|
water for injections
|
Do not take this medicine if you are allergic to any of these ingredients.
What ADYNOVATE looks like
ADYNOVATE is supplied as a white to off-white powder in a single-dose glass vial.
Each pack of ADYNOVATE contains:
1 drug powder vial of ADYNOVATE
1 diluent vial of water for injections (either a 5 mL diluent or a 2 mL diluent is
provided to dissolve the powder for injection)
1 reconstitution device (either BAXJECT II Hi-Flow or a pre-assembled BAXJECT III
system is provided for mixing the powder and diluent).
ADYNOVATE is available in 7 strengths:
ADYNOVATE 250 IU ( 5 mL or 2 mL) - AUST R 273517
ADYNOVATE 500 IU (5 mL or 2 mL) - AUST R 278727
ADYNOVATE 750 IU (5 mL or 2 mL) - AUST R 300850
ADYNOVATE 1000 IU (5 mL or 2 mL) - AUST R 278728
ADYNOVATE 1500 IU (5 mL or 2 mL) - AUST R 300851
ADYNOVATE 2000 IU (5 mL) - AUST R 278729
ADYNOVATE 3000 IU (5 mL) - AUST R 300852
Not all presentations may be marketed.
Who distributes ADYNOVATE
ADYNOVATE is supplied in Australia by:
Takeda Pharmaceuticals Australia Pty Ltd
Level 39, 225 George Street
Sydney NSW 2000
Australia
Telephone: 1800 012 612
www.takeda.com/en-au
ADYNOVATE is supplied in New Zealand by:
Takeda New Zealand Pty Limited
Level 10, 21 Queen Street
Auckland 1010
New Zealand
Telephone: 0508 169 077
www.takeda.com/en-au
This leaflet was prepared in April 2023.
ADYNOVATE® is a registered trademark of Baxalta Incorporated.
TAKEDA® and the TAKEDA Logo® are registered trademarks of Takeda Pharmaceutical Company Limited.
8. Instructions for use
IMPORTANT
Contact your doctor or Haemophilia Treatment Centre if you have any questions or if
you experience any problems following this instruction guide.
These instructions are intended only as a visual aid for those patients who have been
trained by their doctor or Haemophilia Treatment Centre on the proper way to self-inject
the medicine.
Do not attempt to inject ADYNOVATE by yourself unless you have received proper training
by your doctor or Haemophilia Treatment Centre on how to use the product.
Use only the water for injections (diluent) and the reconstitution device provided
in the pack to prepare the solution for injection.
If more than one vial of ADYNOVATE is needed for the dose, mix each vial of ADYNOVATE
using a separate BAXJECT II Hi-Flow or a separate preassembled BAXJECT III system
supplied in each pack.
Follow the step-by-step instructions that is specific to the reconstitution device
supplied with your ADYNOVATE.
After preparing ADYNOVATE, use the solution as soon as possible, within 3 hours after
mixing.
Always inspect ADYNOVATE before use and after it has been mixed. After mixing, the
solution should be clear to colourless.
Do not use the solution if it is discoloured, or cloudy, or contains particles.
Preparing ADYNOVATE using aseptic technique
In a quiet place, prepare a clean surface and gather all the materials you will need
for the injection.
Remove ADYNOVATE from the refrigerator and check the expiry date on the package.
Wash your hands and put on clean exam gloves.
If you are self-injecting at home the use of gloves is optional.
Using the BAXJECT II Hi-Flow device
1. Use aseptic technique (clean and germ free) and a flat work surface during the reconstitution
procedure.
2. Allow the ADYNOVATE powder and diluent vials to reach room temperature before use.
3. Remove plastic caps from the ADYNOVATE powder and diluent vials.
4. Cleanse rubber stoppers with an alcohol wipe and allow to dry before use.
5. Open the BAXJECT II Hi-Flow device package by peeling away the lid, without touching
the inside (Figure A). Do not remove the device from the package.
Figure A
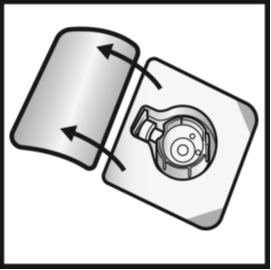
6. Turn the package over. Press straight down to fully insert the clear plastic spike
through the diluent vial stopper (Figure B).
Figure B
7. Grip the BAXJECT II Hi-Flow package at its edge and pull the package off the device
(Figure C). Do not remove the blue cap from the BAXJECT II Hi-Flow device. Do not
touch the exposed purple plastic spike.
Figure C
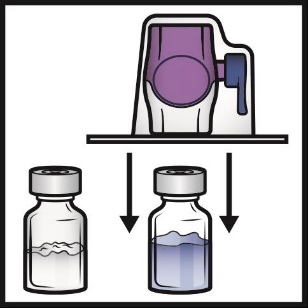
8. Turn the system over so that the diluent vial is on top. Quickly insert the purple
plastic spike fully into the ADYNOVATE powder vial stopper by pushing straight down
(Figure D). The vacuum will draw the diluent into the ADYNOVATE powder vial.
Figure D
9. Swirl gently until the ADYNOVATE powder is completely dissolved. The powder should
dissolve rapidly (usually in less than 1 minute).
Do not refrigerate the solution after reconstitution.
Administration
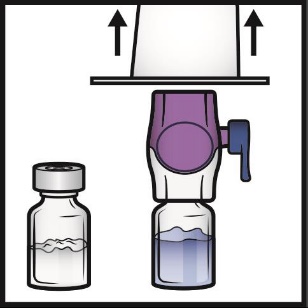
1. Remove the blue cap from the BAXJECT II Hi-Flow device (Figure E). Connect the syringe
to the BAXJECT II Hi-Flow. Use of a Luer-lock syringe is recommended.
Figure E
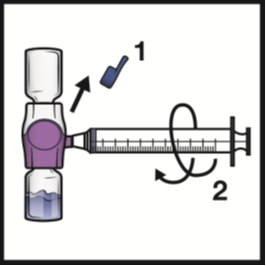
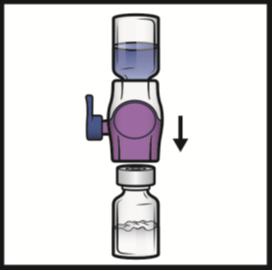
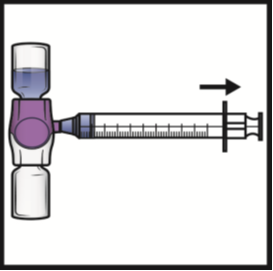
2. Turn the system upside down (ADYNOVATE powder vial now on top). Draw the reconstituted
solution into the syringe by pulling the plunger back slowly (Figure F). Do not draw
air into the syringe.
Figure F
3. Disconnect the syringe; attached a needle suitable for intravenous injection. If more
than one vial of ADYNOVATE is to be used, the contents of multiple vials may be drawn
into the same syringe.
4. Administer the dose by injecting the solution directly into the vein over a period
of up to 5 minutes (maximum infusion rate of 10 mL per min).
Using the BAXJECT III system
The ADYNOVATE powder vial and the diluent vial are supplied preassembled with the
BAXJECT III reconstitution device within a sealed blister package.
Do not use if the lid is not completely sealed on the blister packaging.
1. Take the sealed blister out from the pack and allow the ADYNOVATE powder and diluent
vials to reach room temperature before use.
2. Open the blister package by peeling away the lid. Remove the preassembled BAXJECT
III system from the blister.
3. Place the pre-assembled BAXJECT III system on a flat surface with the diluent vial
on top (Figure 1). The diluent vial has a blue stripe. Do not remove the blue cap
until instructed in a later step.
Figure 1
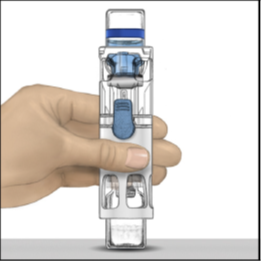
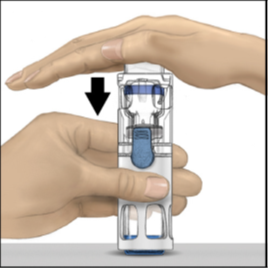
4. Hold the lower end of the pre-assembled BAXJECT III system with one hand, and use
the other hand to press down firmly on the diluent vial until the system is fully
collapsed and the diluent flows down into the ADYNOVATE powder vial (Figure 2). Do
not tilt the system until all the diluent is completely transferred to the ADYNOVATE
powder vial.
Figure 2
5. Check to make sure the diluent transfer is complete. Swirl gently until all the ADYNOVATE
powder is dissolved (Figure 3). The powder should dissolve rapidly (usually in less
than 1 minute).
Figure 3
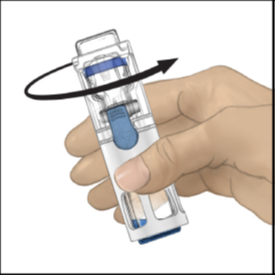
Make sure all the ADYNOVATE powder is completely dissolved, otherwise the undissolved
powder will not pass through the device filter.
Do not refrigerate the solution after reconstitution.
Administration
1. Remove the blue cap from the BAXJECT III device. Connect the syringe to the BAXJECT
III device. Use of a Luer-lock syringe is recommended.
2. Turn the system upside down (ADYNOVATE powder vial now on top). Draw the reconstituted
solution into the syringe by pulling the plunger back slowly. Do not draw air into
the syringe.
3. Disconnect the syringe; attach a needle suitable for intravenous injection. If more
than one vial of ADYNOVATE is to be used, the contents of multiple vials may be drawn
into the same syringe.
4. Administer the dose by injecting the solution directly into the vein over a period
of up to 5 minutes (maximum infusion rate 10 mL per min).
ADYNOVATE® and BAXJECT® are registered trademarks of Baxalta Incorporated.
TAKEDA® and the TAKEDA Logo® are registered trademarks of Takeda Pharmaceutical Company Limited.