2. What should I know before I use TRELEGY ELLIPTA?
Do not use if you have ever had an allergic reaction to TRELEGY ELLIPTA or any of
the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines,
or are pregnant or plan to become pregnant or are breastfeeding. TRELEGY ELLIPTA should
not be used in children or adolescents under the age of 18 years.
3. What if I am taking other medicines?
4. How do I use TRELEGY ELLIPTA?
The usual dose is one inhalation once a day. Use TRELEGY ELLIPTA at the same time
each day.
5. What should I know while using TRELEGY ELLIPTA?
|
Things you should do
|
Remind any Healthcare Professional (HCP) you visit that you are using TRELEGY ELLIPTA.
Contact your doctor if you are concerned about any side effects.
Tell your doctor immediately if you become or intend to become pregnant while using
TRELEGY ELLIPTA.
Tell your doctor if you feel you are getting breathless or wheezy more often than
normal, or if you are using a quick-acting inhaler (such as VENTOLIN/salbutamol) more
than usual.
|
|
Things you should not do
|
Do not stop using this medicine suddenly or change the dosage without checking with
your doctor.
Do not take any other medicines to help you breathe without checking with your doctor.
|
|
Driving or using machines
|
Be careful driving or operating machinery until you know how TRELEGY ELLIPTA affects
you.
|
|
Looking after your medicine
|
Do not open the foil lid until you are ready to use the inhaler for the first time.
Once TRELEGY ELLIPTA has been removed from the tray, you may store it for a maximum
period of one month or when your counter reads "0", whichever occurs first.
Store TRELEGY ELLIPTA in a cool dry place (below 30°C) away from moisture, heat, or
sunlight.
If you store TRELEGY ELLIPTA in the refrigerator, allow the inhaler to return to room
temperature for at least 1 hour before you use it.
|
6. Are there any side effects?
Less serious side effects include common cold, oral thrush, infection of the nose,
sinuses or throat, infection of the upper airways or viral respiratory tract infection,
headache, changes in taste, tremor, cough, oropharyngeal pain, hoarseness, itchy,
runny or blocked nose, pain in the back of the mouth and throat, inflammation of the
sinuses, bronchitis, influenza, dry mouth, constipation, hyperglycaemia, painful or
discomfort passing urine and frequent urination, joint pain, back pain, weakening
of the bones leading to fractures, muscle spasms or anxiety. Serious side effects
include eye disorders, cardiac disorders, allergic reaction, pneumonia, immediate
breathing difficulties or wheezing. For more information, including what to do if
you have any side effects, see Section
6. Are there any side effects? in the full CMI.
Active ingredient(s):
Fluticasone furoate 100 or 200 micrograms, umeclidinium (as bromide) 62.5 micrograms
and vilanterol (as trifenatate) 25 micrograms per inhalation
Consumer Medicine Information (CMI)
This leaflet provides important information about using TRELEGY ELLIPTA. You should also speak to your Healthcare Professional (HCP) if you would like further
information or if you have any concerns or questions about using TRELEGY ELLIPTA.
Where to find information in this leaflet:
1. Why am I using TRELEGY ELLIPTA?
TRELEGY ELLIPTA contains the active ingredients fluticasone furoate, umeclidinium
(as bromide) and vilanterol (as trifenatate). Fluticasone furoate belongs to a group of medicines called corticosteroids, often
simply called steroids. Umeclidinium and vilanterol belong to a group of medicines
called bronchodilators.
TRELEGY ELLIPTA is used to treat adults with asthma and chronic obstructive pulmonary
disease (COPD).
Asthma is a condition affecting the lungs. Symptoms of asthma include shortness of
breath, wheezing, chest tightness and cough. Two main causes of asthma symptoms are
bronchoconstriction (tightening of the muscle surrounding the airways) and inflammation
(swelling and irritation of the airways).
Chronic obstructive pulmonary disease (COPD) is a long-term condition affecting the
lungs. Symptoms of COPD include shortness of breath, cough, chest discomfort and coughing
up phlegm. The COPD symptoms are mainly due to bronchoconstriction (tightening of
the muscle surrounding the airways) and inflammation (swelling and irritation of the
airways).
Inhaled corticosteroids are used to reduce inflammation. They reduce the swelling
and irritation in the small air passages in the lungs and so ease breathing problems.
Inhaled corticosteroids also help to prevent attacks of asthma.
Bronchodilators relax the muscles of the small air passages in the lungs. This helps
to open the airways and makes it easier for air to get in and out of the lungs. When
it is used regularly, it helps the small air passages to remain open.
When TRELEGY ELLIPTA is used regularly, it can help to control your breathing difficulties.
TRELEGY ELLIPTA should not be used to relieve a sudden attack of breathlessness or
wheezing. If you get this sort of attack, you must use a quick-acting inhaler (such
as VENTOLIN/salbutamol).
Your doctor may have prescribed TRELEGY ELLIPTA for another reason.
Ask your doctor if you have any questions about why this medicine has been prescribed
for you.
This medicine is not addictive.
This medicine is available only with a prescription.
2. What should I know before I use TRELEGY ELLIPTA?
Warnings
Do not use TRELEGY ELLIPTA if:
you are allergic (hypersensitive) to fluticasone furoate, umeclidinium, vilanterol,
or any of the ingredients listed at the end of this leaflet.
you are allergic (hypersensitive) to lactose or milk protein.
the expiry date printed on the pack has passed or if the packaging is torn or show
signs of tampering.
Always check the ingredients to make sure you can use this medicine.
Check with your doctor if you:
have any other medical conditions.
take any medicines for any other condition.
have been diagnosed with an intolerance to some sugars, or to milk protein.
have liver disease, as you may be more likely to have side effects. If you have moderate
or severe liver disease, your doctor will limit your dose to the lower strength of
TRELEGY ELLIPTA (100/62.5/25 micrograms once daily).
have heart problems or high blood pressure.
if you have ever been told you have diabetes or high blood sugar.
are being treated or have ever been treated for tuberculosis (TB), pneumonia or any
other long-standing or untreated infections.
have eye problems such as glaucoma or cataracts.
have an enlarged prostate, difficulty passing urine or a blockage in your bladder.
have weak bones (osteoporosis).
have any type of viral, bacterial, or fungal infection.
have a problem with your immune system.
Your doctor should give you a personal Action Plan to help manage your asthma or COPD.
This plan will include what medicines to take regularly to control your asthma or
COPD, as well as what "reliever" medicines to use when you have sudden attacks of
breathlessness or wheezing.
Ask your doctor if you have any questions about your Action Plan.
During treatment, you may be at risk of developing certain side effects. It is important
you understand these risks and how to monitor for them. See additional information
under Section
6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
TRELEGY ELLIPTA is not usually recommended for use during pregnancy. Your doctor will
weigh up the benefits to you against the potential risks to your baby when considering
the use of TRELEGY ELLIPTA during your pregnancy.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
It is not known whether the ingredients of TRELEGY ELLIPTA can pass into breast milk.
Children and adolescents
TRELEGY ELLIPTA should not be used in children or adolescents under the age of 18
years.
3. What if I am taking other medicines?
Tell your Healthcare Professional (HCP) if you are taking any other medicines, including
any medicines, vitamins, or supplements that you buy without a prescription from your
pharmacy, supermarket, or health food shop. This should include all of the medicines
that you are using for your asthma or COPD.
Some medicines may interfere with TRELEGY ELLIPTA and affect how it works or make
it more likely that you will have side effects.
These medicines include:
ketoconazole, to treat fungal infections.
ritonavir, to treat viral infections.
other long-acting medicines similar to this medicine that are used to treat breathing
problems (i.e. inhalers used as maintenance or preventer therapy).
medicines to treat depression or mood/mental disorders (such as monoamine oxidase
inhibitors or tricyclic depressants).
Medicines that should be avoided:
A class of medicines known as "beta-blockers" used to treat high blood pressure or
a heart condition.
Medicines which are similar to TRELEGY ELLIPTA should not be used together as an overdose
may result.
Check with your Healthcare Professional (HCP) if you are not sure about what medicines,
vitamins or supplements you are taking and if these affect TRELEGY ELLIPTA.
4. How do I use TRELEGY ELLIPTA?
How much to use
Always use TRELEGY ELLIPTA exactly as your doctor has told you to. Check with your
Healthcare Professional (HCP) if you are not sure.
Asthma:
The dose for asthma is one inhalation of TRELEGY ELLIPTA 100/62.5/25 micrograms once
daily, at the same time each day OR one inhalation of TRELEGY ELLIPTA 200/62.5/25
micrograms once daily, at the same time each day.
Your doctor will decide which strength of TRELEGY ELLIPTA is required to treat your
asthma.
COPD:
The dose for COPD is one inhalation of TRELEGY ELLIPTA 100/62.5/25 micrograms once
daily at the same time each day.
The higher strength of TRELEGY ELLIPTA 200/62.5/25 micrograms is not suitable for
the treatment of COPD.
When to use TRELEGY ELLIPTA
Use TRELEGY ELLIPTA once daily at the same time each day.
It is very important that you use TRELEGY ELLIPTA every day, as instructed by you
Healthcare Professional (HCP). This will help to keep you free of symptoms throughout
the day and night.
Use TRELEGY ELLIPTA for as long as your Healthcare Professional (HCP) recommends.
It will only be effective as long as you are using it. Do not stop unless your Healthcare
Professional (HCP) advises you to, even if you feel better.
How to use TRELEGY ELLIPTA
To use TRELEGY ELLIPTA, you breathe it into your lungs through your mouth using the
Ellipta inhaler.
The full instructions for using TRELEGY ELLIPTA are given on a leaflet inside the
pack. A brief summary of the instructions is provided below.
TRELEGY ELLIPTA is ready to use straight away. No preparation or checks of the inhaler
are required.
Do not open TRELEGY ELLIPTA until you are ready to inhale a dose of medicine.
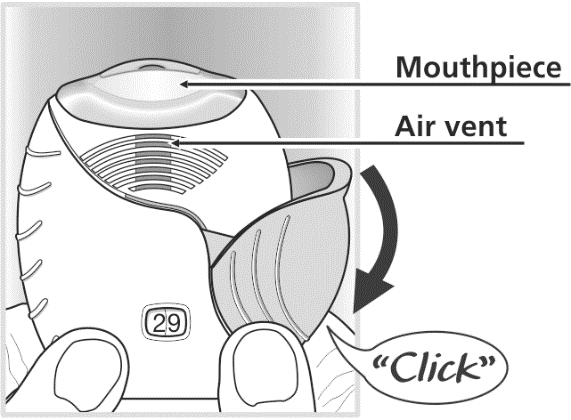
Step 1: Prepare a dose
Wait to open the cover until you are ready to take your dose. Slide the cover fully
down until you hear a "click".
If the dose counter does not count down as you hear the "click", the inhaler will
not deliver medicine. Take it back to your pharmacist for advice.
Do not shake the inhaler at any time.
Step 2: Inhale your medication
While holding the inhaler away from your mouth, breathe out as far as is comfortable.
Do not breathe out into the inhaler.
Put the mouthpiece between your lips and close your lips firmly around it.
Do not block the air vent with your fingers.
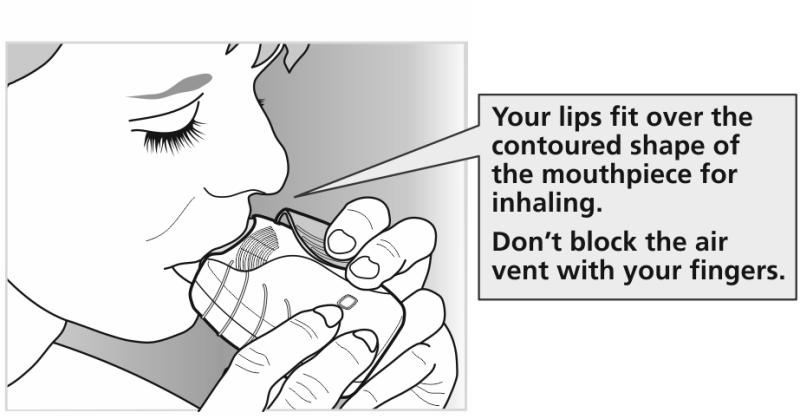
Take one long, steady, deep breath in. Hold this breath for about 3-4 seconds or as
long as is comfortable.
Remove the inhaler from your mouth.
Breathe out slowly and gently away from the mouthpiece.
You may not be able to taste or feel the medicine, even when you are using the inhaler
correctly.
After using TRELEGY ELLIPTA, you may clean the mouthpiece, using a dry tissue, before
you close the cover.
Step 3: Close the inhaler and rinse your mouth
Slide the cover upwards as far as it will go, to cover the mouthpiece.
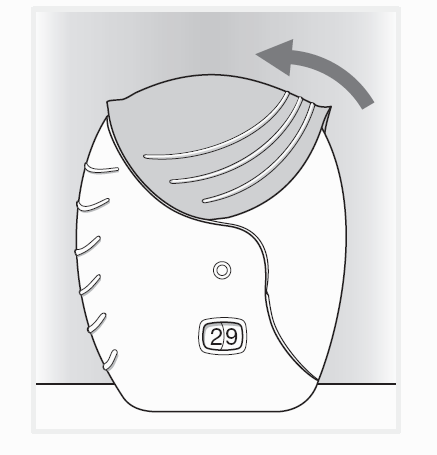
Rinse your mouth out with water without swallowing after you have used the inhaler.
This will make it less likely that you will develop a sore mouth or throat as side
effects.
If you forget to use TRELEGY ELLIPTA
TRELEGY ELLIPTA should be used regularly at the same time each day. If you miss your
dose at the usual time, skip the dose you missed and use your next dose when you are
meant to. Otherwise, use it as soon as you remember, then go back to using it as you
would normally.
Do not take a double dose to make up for the dose you missed.
If you are not sure what to do, ask your Healthcare Professional (HCP).
If you have trouble remembering to take your medicine, ask your pharmacist for some
hints.
If you become wheezy, or breathless, or develop any other symptoms of an asthma attack,
use your quick-acting inhaler (e.g. VENTOLIN/salbutamol), then seek medical advice.
If you use too much TRELEGY ELLIPTA (overdose)
If you think that you have used too much TRELEGY ELLIPTA, you may need urgent medical
attention. You may notice that your heart is beating faster than usual, you feel shaky
or have a headache.
You should immediately:
phone the Poisons Information Centre
(by calling
13 11 26 in Australia), or
contact your doctor, or
go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
If you have used larger doses than instructed for a long period of time, it is particularly
important that you ask your Healthcare Professional (HCP) for advice. This is because
larger doses of TRELEGY ELLIPTA may reduce the amount of steroid hormones produced
naturally by your body.
5. What should I know while using TRELEGY ELLIPTA?
Things you should do
Remind any Healthcare Professional (HCP) you visit that you are using TRELEGY ELLIPTA,
especially if you are about to be started on any new medicine.
Use TRELEGY ELLIPTA for as long as your doctor recommends. It will only be effective
as long as you are using it. Do not stop unless your doctor advises you to, even if
you feel better.
Check with your doctor if you are unsure about which medicines for asthma or COPD
you should continue taking while being treated with TRELEGY ELLIPTA.
Continue to use a quick-acting inhaler (such as VENTOLIN/salbutamol) during an acute
episode.
If you are about to have surgery, tell the surgeon or anaesthetist that you are taking
this medicine.
Keep all of your doctor's appointments so that your progress can be checked.
Call your doctor straight away if you:
experience blurred vision or other visual disturbances.
experience increased thirst, frequent urination, or unexplained tiredness (signs of
high blood sugar).
become pregnant while taking this medicine.
feel that your breathing or wheezing gets worse straight after using TRELEGY ELLIPTA.
Stop using it immediately.
Things you should not do
Do not stop using this medicine suddenly or change the dosage without medical advice.
Do not use TRELEGY ELLIPTA to treat any other complains unless your doctor tells you
to.
Do not give your medicine to anyone else, even if they have the same condition as
you.
If you are currently on oral or injectable corticosteroids, do not stop taking these
medicines suddenly without checking with your doctor.
Do not use any other long-acting inhalers which contain inhaled corticosteroids or
bronchodilators.
Do not take any other medicines for your breathing problems without checking with
your doctor.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how TRELEGY
ELLIPTA affects you.
This medicine is not expected to affect your ability to drive a car or operate machinery.
Action Plan
If you have an Action Plan for your asthma or COPD that you have agreed with your
doctor, follow it closely at all times.
Looking after your medicine
Store in the original package container in order to protect from moisture and do not
open the foil lid until you are ready to inhale for the first time.
If you store in a refrigerator, allow the inhaler to return to room temperature for
at least an hour before use.
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight where the temperature
stays below 30°C.
Do not store it:
in the bathroom or near a sink, or
in the car or on window sills.
Keep it where young children cannot reach it.
A locked cupboard at least one-and-a-half metres above the ground is a good place
to store medicines.
When to discard your medicine
Ask your pharmacist to safely throw away TRELEGY ELLIPTA one month after you open
the foil tray or when the counter reads "0", whichever comes first. Write the date
the inhaler should be discarded on the label in the space provided. The date should
be added as soon as the inhaler has been removed from the tray.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy
for safe disposal.
Do not use this medicine after the expiry date or if the packaging is torn or shows
signs of tampering.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of
them are minor and temporary. However, some side effects may need medical attention.
Your doctor will consider the risk of side effects when deciding which strength of
TRELEGY ELLIPTA you should use.
Do not be alarmed by the following list of side effects. You may not experience any
of them.
See the information below and, if you need to, ask your doctor or pharmacist if you
have any further questions about side effects.
Less serious side effects
Serious side effects
Tell your doctor if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can
report side effects to the Therapeutic Goods Administration online at
www.tga.gov.au/reporting-problems . By reporting side effects, you can help provide more information on the safety of
this medicine.
Always make sure you speak to your doctor before you decide to stop taking any of
your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What TRELEGY ELLIPTA contains
|
Active ingredient
|
Fluticasone furoate
Umeclidinium (as bromide)
Vilanterol (as trifenatate)
|
|
Other ingredients
|
Lactose monohydrate (contains milk protein)
Magnesium stearate
|
|
Potential allergens
|
Lactose monohydrate (contains milk protein)
Fluticasone furoate
Umeclidinium (as bromide)
Vilanterol (as trifenatate)
|
Each dose contains 100 or 200 micrograms of the active ingredient fluticasone furoate.
The amount depends on which strength of TRELEGY ELLIPTA you have been given. Each
dose also contains the active ingredients 62.5 micrograms of umeclidinium (as bromide)
and 25 micrograms of vilanterol (as trifenatate).
Do not take this medicine if you are allergic to any of these ingredients.
What TRELEGY ELLIPTA looks like
The Ellipta device itself is a plastic inhaler with a light grey body, a beige mouthpiece
cover and a dose counter. It is packaged in a foil laminate tray with a peelable foil
lid.
The active substances are present in powder form in separate blister strips inside
the inhaler. TRELEGY ELLIPTA contains either 14 or 30 doses on each blister strip.
The 14 dose inhaler has 14 blisters on each strip, and the 30 dose inhaler has 30
blisters on each strip, depending on which pack size you have been given.
The tray contains a desiccant sachet, to reduce moisture in the packaging. Once you
have opened the lid of the tray, throw the desiccant away - do not open, eat or inhale
it.
TRELEGY ELLIPTA 100/62.5/25 micrograms is AUST R 284636.
TRELEGY ELLIPTA 200/62.5/25 micrograms is AUST R 335858.
Who distributes TRELEGY ELLIPTA
GlaxoSmithKline Australia Pty Ltd
Level 4, 436 Johnston Street,
Abbotsford, Victoria, 3067
Phone: 1800 033 109
Trade marks are owned by or licensed to the GSK group of companies.
© 2024 GSK group of companies or licensor.
This leaflet was prepared in September 2024.
Version 8.0