This article is based on a poster originally authored by Justina Then, Isavella Georgiades, Emily Richardson, and Tomasz Kostrzewski.
Pharmaceutical development often fails due to drug-induced liver injury (DILI), with idiosyncratic, immune-mediated forms representing the most significant challenge.1
Immune drug-induced liver injury (iDILI) can occur through reactive metabolites or drug-protein complexes that activate antigen presentation, T-cell activation, and the amplification of inflammatory pathways.
Combined, these triggers can ultimately result in hepatocellular injury.2 Susceptibility is further influenced by genetic factors, especially HLA polymorphisms.3
The complexity of the immune system required to capture these human-specific mechanisms is insufficient in conventional preclinical systems, motivating the use of liver microphysiological systems (MPS). Incorporating PBMCs into these platforms permits mechanistic analysis of immune-driven hepatotoxicity.
In alignment with the FDA’s 2025 roadmap advocating for New Approach Methodologies (NAMs), this article describes the use of the PhysioMimix® DILI assay in conjunction with circulating PBMCs to provide enhanced translational relevance for the assessment of monoclonal antibodies (mAbs) and other modalities.4
Materials and methods
LifeNet Health provided the cryopreserved Primary Human Hepatocytes (PHHs), while PBMCs were obtained from STEMCELL Technologies. Cells were seeded at a 1:10 ratio, with PHHs seeded at 400,000 cells per chip in the PhysioMimix Multi-chip Liver-12 plates and cultured under perfusion for 8 days using the PhysioMimix® Core System (Figure 1).
On the fourth day of culture, PBMCs were co-cultured with liver microtissues and dosed with two concentrations of the mAbs Ipilimumab or Infliximab, both of which have documented DILI risks, with one acting intrinsically and the other idiosyncratically. Daily dosing was conducted, and each condition was evaluated in triplicate.
Albumin production was quantified using ELISA (AssayPro), while LDH release was measured using the Cytotox96 assay (Promega). ALT activity was assessed with the human ALT Assay kit (Abcam), and CRP, IL-6, IL- 10, TNF-α, and IFN-γ were measured via ELISA (R&D Systems).
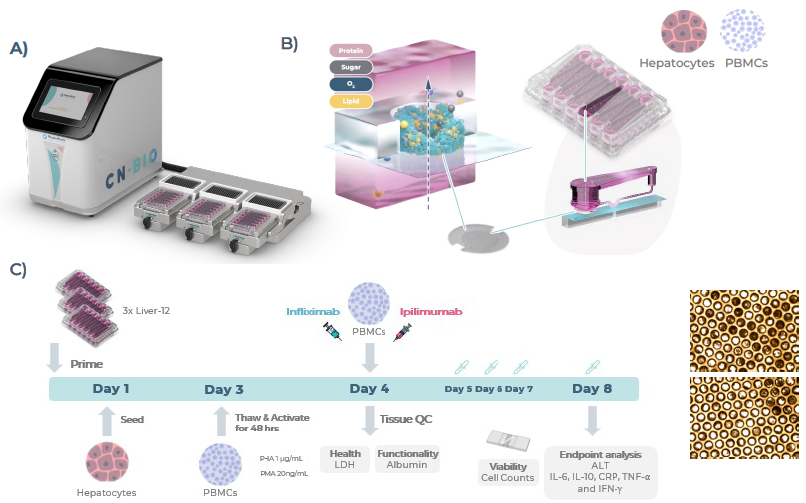
Figure 1. Experimental setup of Liver MPS for toxicity DILI testing, including human PHHs and PBMCs. A) The PhysioMimix Core System B) Schematic of the Liver-12 plate, including the perfusion path, scaffold and cross-section of the liver microtissue within the scaffold. C) Timeline and cells used for toxicity experiments: in-house validated PHH and HLA-matched PBMCs (coculture). Endpoint assays for cell health (LDH and ALT) and cytokine expression (IL-6, IL-2, IL-10, TNF-α, IFN-γ, and CRP), cell counts, and viability assessment of PBMCs, and representative light microscopy images of the scaffolds. Image Credit: CN Bio
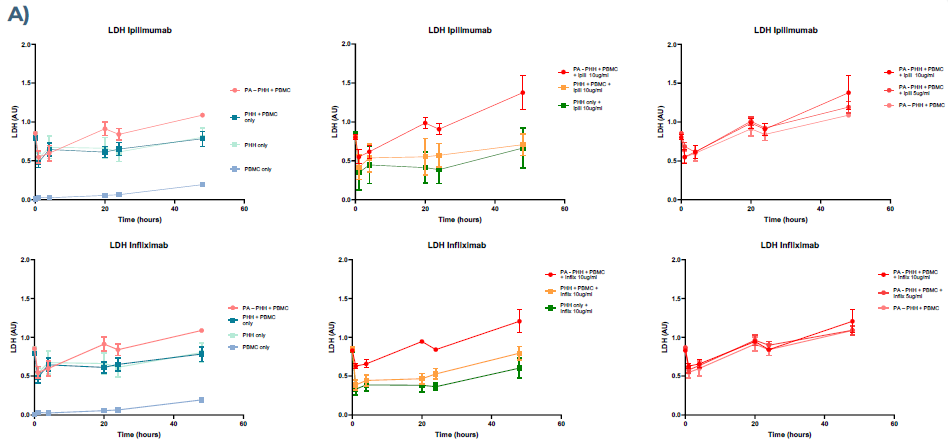
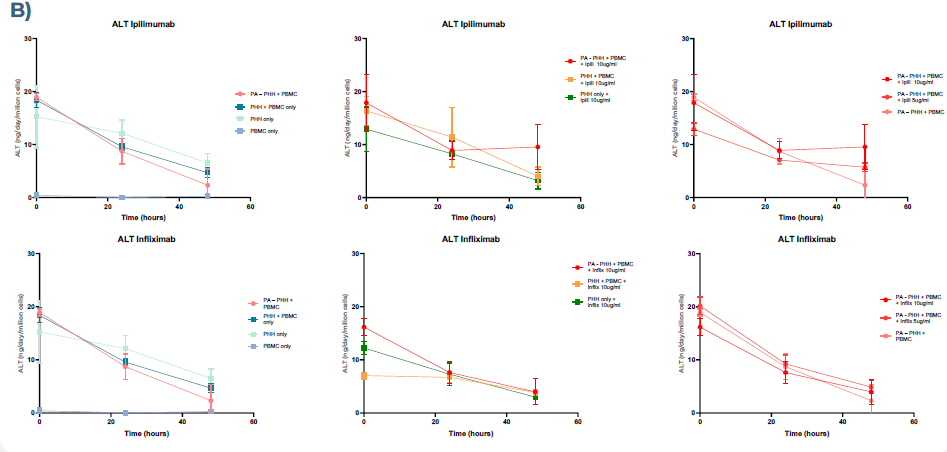
Figure 2. Health and functional biomarker profiles for PHHs co-cultured with PBMCs in MPS. A) PHHs show signs of cell stress by increased production of LDH in the presence of preactivated PBMCs and mAbs (Ipilimumab and Infliximab), compared to co-cultured PHH and PBMC conditions. B) The clinically relevant PHH health biomarker, ALT, indicates cell stress when co-cultured with preactivated PBMCs and mAb. Image Credit: CN Bio
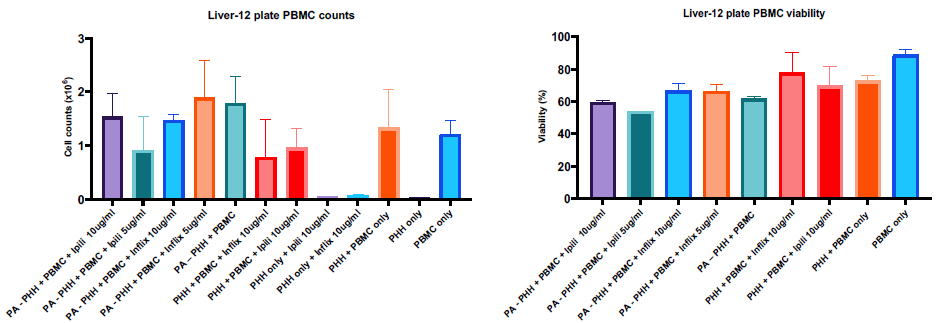
Figure 3. PBMC recovery and viability. From a seeding of 4 x 106, roughly 40 % of the cells were recovered from the Liver-12 plate (left) while viability remained high over the 48h co-culture (right). Image Credit: CN Bio
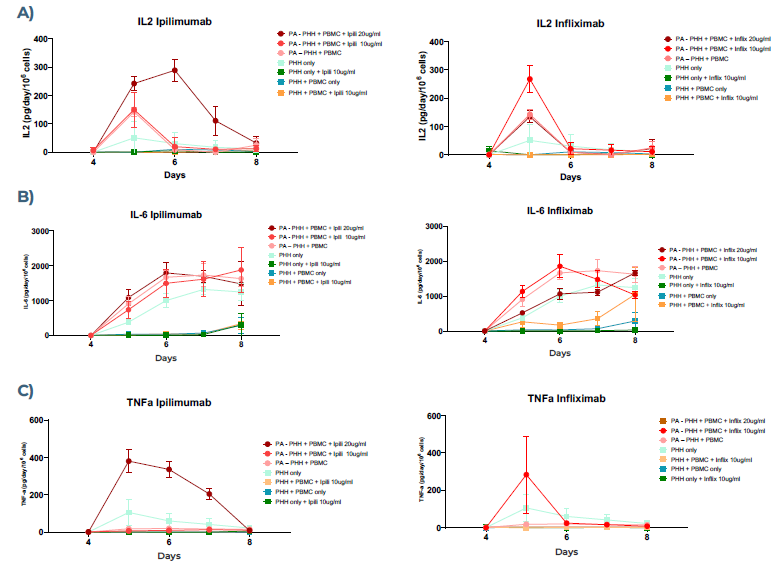
Figure 4. IL-2, IL-6, and TNF- α cytokine expression for PHHs co-cultured with PBMCs in MPS in the presence of Ipilimumab (left) or Infliximab (right). A) IL-2 expression increased and spiked on day 5 with both drugs in the presence of preactivated PBMCs; however, Ipilimumab induced consistent expression over time. B) IL-6 expression increased over time for all conditions with preactivated PBMCs in the presence of mAbs, showing the highest expression for Ipilimumab. C) TNF-α expression showed an increase in Ipilimumab at higher concentrations of mAb and consistent expression over time, whereas Infliximab spiked at day 5 at the lower concentration only. Image Credit: CN Bio
Conclusion
Integrating human HLA-matched PBMCs into the PHH Liver MPS offers a promising, human-relevant methodology for assessing the iDILI potential of mAbs.
The PhysioMimix® Core system facilitates continuous perfusion of nutrients, oxygen, and immune cells through 3D liver microtissues, sustaining physiological flow, fostering long-term hepatocyte function, and enabling dynamic immune-hepatic interactions that more accurately replicate in vivo human liver physiology.
Evidence of mAb-specific iDILI impacts is demonstrated by elevated PHH stress markers (ALT) and heightened cytokine expression only when both preactivated PBMCs and mAbs are present. These levels are greater and more uniform over time in the presence of Ipilimumab.
Notably, the ability to recover immune cells for downstream phenotyping will enable additional detailed evaluations. Overall, the data underscores the value of Liver MPS as a platform for modeling immune-related DILI to support risk evaluation and accelerate both the development and regulatory acceptance of cutting-edge antibody drugs.
Acknowledgments
Produced from materials originally authored by Justina Then, Isavella Georgiades, Emily Richardson, and Tomasz Kostrzewski.
References and further reading:
- Gerussi, A., et al. (2021). Immune-Mediated Drug-Induced Liver Injury: Immunogenetics and Experimental Models. International Journal of Molecular Sciences, [online] 22(9), p.4557. DOI: 10.3390/ijms22094557. https://www.mdpi.com/1422-0067/22/9/4557.
- Tasnim, F., et al. (2021). Recent Advances in Models of Immune-Mediated Drug-Induced Liver Injury. Frontiers in Toxicology, 3. DOI: 10.3389/ftox.2021.605392. https://www.frontiersin.org/journals/toxicology/articles/10.3389/ftox.2021.605392/full.
- Onaciu, A., et al. (2026). Human Leukocyte Antigen (HLA) Signatures and Idiosyncratic Drug-Induced Liver Injury. International Journal of Molecular Sciences, 27(1), 482. DOI: 10.3390/ijms27010482. https://pmc.ncbi.nlm.nih.gov/articles/PMC12787173/
About CN Bio
CN Bio is a leading organ-on-a-chip (OOC) company that offers a portfolio of products and contract research services to optimize the accuracy and efficiency of bringing new medicines to market. With more than a decade of research and development experience, they aim to transform the way human-relevant pre-clinical data is generated through the development of advanced in vitro human organ models.
CN-Bio's PhysioMimix® Core microphysiological system (MPS) enables researchers to recreate human biology in the lab and is the only microphysiological system with validated performance across single-, multi-organ, and higher throughput configurations. This easy-to-adapt, scalable technology bridges the gap between traditional cell culture and human studies to support the development of safer and more efficacious therapeutics while reducing the dependence on animal model usage.
CN Bio’s portfolio of products (MPS, 3D-validated cells, consumable plates) and services supports researchers who require reliable, data-rich in vitro studies to uncover novel mechanistic insights into the mechanisms of action of drugs or diseases.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.
Last Updated: Apr 13, 2026