Introduction
How does monkeypox present itself in humans?
Therapy and management for monkeypox
References
Monkeypox virus is a zoonotic Orthopoxvirus and a possible biothreat agent that causes human disease with varying morbidity and mortality. Today, monkeypox is considered the most critical orthopoxvirus infection in human beings since the eradication of smallpox. Symptoms of this potentially serious illness include a high fever and the appearance of a characteristic rash.
Image Credit: MIA Studio/Shutterstock.com
How does monkeypox present itself in humans?
After exposure to the virus and infection, there is an incubation period that varies between 6 and 14 days, followed by a prodromal period of about 1-3 days before the appearance of the typical maculopapular rash. During this prodrome phase, the affected individual can experience fever, malaise, and swollen lymph nodes, with the possible occurrence of other symptoms such as sweats, headache, backache, shortness of breath, sore throat, and cough.
Lymph node enlargement can arise in the submandibular, cervical, or inguinal regions, and such lymphadenopathy is essential when distinguishing monkeypox from smallpox, as it is not a common feature in the latter disease. It is observed in approximately 90% of all human monkeypox infections, and the enlarged lymph nodes are tender, firm, and sometimes even painful.
After the prodrome period, a typical maculopapular rash develops with the mean diameter of the skin lesions from 0.5 to 1 centimeter. The number of lesions can range from a few to thousands, and during 2-4 weeks, they progress from macules to papules, vesicles, pustules, and finally, a crusting phase characterized by umbilication, scabbing, and desquamation.
The lesions most often affect oral mucous membranes (in 70% of cases) and genitalia (in 30% of cases) but can also affect eyelids and the cornea. Extracutaneous manifestations, such as secondary skin or soft-tissue infections, have been observed in 19% of unvaccinated monkeypox patients. In less than 1% of cases, encephalitis can also occur.
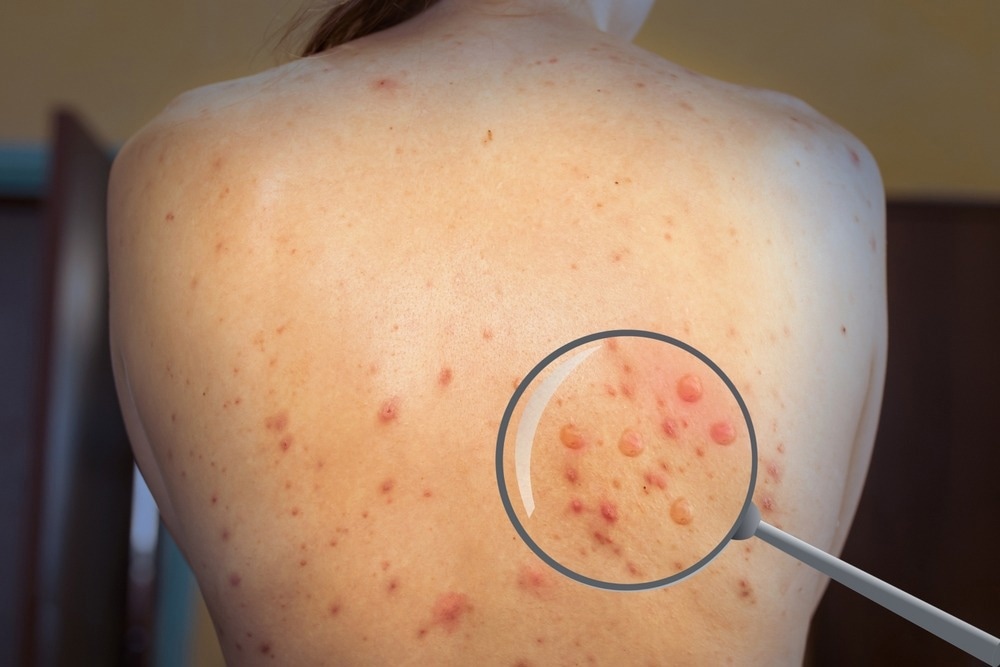
Image Credit: Pavlova Yuliia/Shutterstock.com
Therapy and management for monkeypox
Currently, there are no licensed antiviral drugs for the treatment of monkeypox. However, several agents are promising as antiviral therapeutics against the genus Orthopoxvirus viruses, including monkeypox. By inhibiting viral DNA polymerase, cidofovir shows antiviral activity against a myriad of viruses, although it has not been used to manage orthopoxvirus infection in humans.
There is insufficient data to assess whether severe monkeypox infection could benefit from the treatment with immune globulin. Using that treatment may be considered prophylactic in persons with severe immune deficiency of T-cell function where vaccination against smallpox is contraindicated.
Vaccination with vaccinia virus (i.e., the smallpox vaccine) results in a significant amount of protection against infection with monkeypox. Still, the vaccine is currently not used in monkeypox-endemic areas due to the concerns about severe adverse events in an immunocompromised population. Furthermore, the eradication of monkeypox is not feasible because of the existence of an animal reservoir.
The Centers for Disease Control and Prevention recommends pre-exposure vaccination for individuals investigating animal or human monkeypox cases, people with direct contact with animals that could carry the pathogen, health care workers taking care of patients with monkeypox, and laboratory workers who handle specimens that may contain this virus.
Close contact with patients during the disease outbreak represents the most significant risk factor for monkeypox virus infection. The lack of specific treatments and vaccines means that raising awareness about risk factors and educating the public about prevention measures are the only efficient ways to reduce the burden of this disease.
References:
- www.who.int. (n.d.). Monkeypox. [online] Available at: https://www.who.int/en/news-room/fact-sheets/detail/monkeypox
- Weaver JR, Isaacs SN. Monkeypox virus and insights into its immunomodulatory proteins. Immunol Rev. 2008 Oct;225:96-113. doi: 10.1111/j.1600-065X.2008.00691.x. PMID: 18837778; PMCID: PMC2567051.
- Andrea M. McCollum, Inger K. Damon, Human Monkeypox, Clinical Infectious Diseases, Volume 58, Issue 2, 15 January 2014, Pages 260–267, https://doi.org/10.1093/cid/cit703
- Robert A. Weinstein, Aysegul Nalca, Anne W. Rimoin, Sina Bavari, Chris A. Whitehouse, Reemergence of Monkeypox: Prevalence, Diagnostics, and Countermeasures, Clinical Infectious Diseases, Volume 41, Issue 12, 15 December 2005, Pages 1765–1771, https://doi.org/10.1086/498155
- Fenner F. Human Monkeypox, A Newly Discovered Human Virus Disease. In: Morse S. Emerging Viruses. Oxford University Press, 1993; pp. 176-183.
Last Updated: Aug 10, 2022