Plant-derived medicines are common, but today biopharming has changed the nature of the plants themselves, making use of gene therapy to produce transgenic plants which yield non-native proteins targeting deadly viruses like Ebola or HIV, and some cancers.

Tobacco field. Image Credit: Ivana Vrnoga / Shutterstock
This is different from bioengineering in which plant biological drugs are produced in bioreactors that ferment cultures of animal cells: this method is costly, and biopharming hopes to cut down the cost of producing recombinant antibodies, vaccines, enzymes, and other regulatory proteins to a tenth. It will also boost production capacity and drug yield immensely.
Other advantages include:
- High production of biomass
- Faster production time from gene to protein
- Low capital investment
- Low operating costs
- Easily scaled operations
- Presence of post-translational protein modifications specific to eukaryotes, such as folding, glycosylation, or assembly of multimeric proteins
- Low pathogenic and endotoxic contamination
- Easy manipulation of plant cells via transfection with agrobacterium, transformation, and other techniques to introduce the desired genetic material
How to grow an Ebola vaccine with a tobacco plant
Tobacco and Transgenic Drug Production
The first plant to be used to produce a recombinant antibody, back in 1988, was the tobacco plant. Other plants have also been used for pharmaceutical production, such as maize, wheat, tomato, potato, mustard, banana, and soybean. Tobacco is preferred as the host plant because of its rapid growth and high volume of production. It expresses foreign products much more quickly than with bioreactors, and at a significantly reduced cost.
Tobacco is easy to infect with foreign genes coding for the protein as it shows a high tendency to accept foreign proteins. It grows abundantly, is nontoxic, and does not need much maintenance. When grown for this purpose, manure is not used to promote growth which is modified using air pressure, humidity levels, and light.
The new interest in biopharming using tobacco plants lies in the great and increasing demand for biologic drugs. These are protein derivatives which are synthesized from the basic biochemical building blocks, such as antibodies targeting viral or bacterial antigens in order to prime and stimulate the immune system to eliminate them. In 2013, the biologics market around the world was estimated to be $200 billion, because of their high efficacy, accuracy, and targeted approach.
While tobacco yields of recombinant protein are high, the method of protein expression varies. Plastid expression is easier and more rapid but the products do not show proper post-translational modification. On the other hand, viral expression may introduce the possibility of wild virus spread.
Downstream purification is another potential challenge, because of the high cost factor, often equal to that of the biopharming process itself. This cost may be lowered using two main pathways:
- The use of oleosin-fusion techniques to obtain heterologous proteins from oilseeds
- Constant drawing of the drug product from roots or leaf exudates; this also allows nonstop production of protein and utilizes the plant’s ability to synthesize the chemical of interest over its whole lifetime rather than sacrificing it after a single harvest to extract the product.
The Process
The production of biologic drugs from the tobacco plant takes about eight weeks. The plants are allowed to grow in a greenhouse for the initial six weeks, and then the agrobacterium is introduced. This is allowed to proliferate and the plant expresses the recombinant protein, being stimulated to grow rapidly using controlled plant growth stimulators. At the end of the period, the plants are harvested and blended, to produce a slurry from which the drug is then isolated.
New plant breeding technologies have made it possible to further exploit the possibilities of the tobacco plant, using CRISPR gene editing, advanced forms of grafting, and temporary expression of certain genes by infiltration. Vacuum infiltration consists of soaking tobacco plants, which have grown for five weeks, in a liquid containing the bacterium which carries the antigen of interest. Air is pulled out of the leaves and the agrobacteria are sucked in. Once the gene is expressed, the tobacco plant leaves produce plenty of antibody against the antigen introduced via the bacteria.
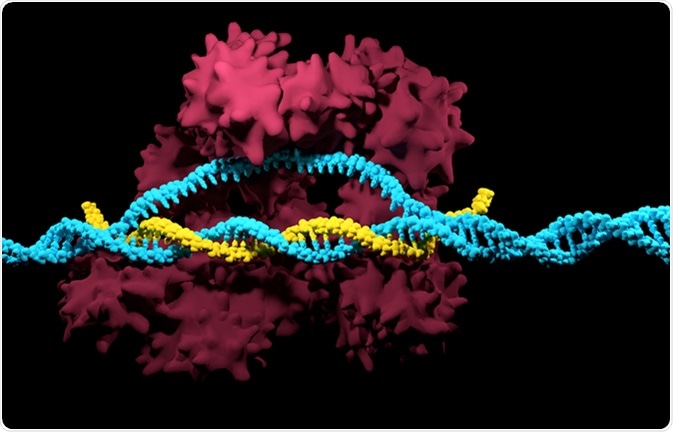
CRISPR-Cas9. Image Credit: Meletios Verras / Shutterstock
This is extracted from the green slurry, purified, and finally blended with other virus components, to yield the initial basis of a vaccine, for instance. The benefit of this type of workflow is that the genetic makeup of the plant is not altered permanently, but rather the bacteria express their own genes inside the tobacco host cells.
This type of process could be effective, but further work is necessary to ensure that clean extraction is possible, and that the product will conform to pharmaceutical and manufacturing norms.
Some of the products on the anvil include antibodies against HIV for topical use as a vaginal gel, and a vaccine against malaria.
Another potential use envisages eating the leaves as such, instead of purifying the extracted drug, provided the drug can be expressed at high enough levels in the plant. This has been tested with several molecules in animal trials and shows promising results both with vaccines and with biologic drugs.
Further Reading
Last Updated: Jan 14, 2019