Jul 13 2016
Bioarray, S.L., IVF SPAIN S.L. and iGLS will present clinical outcomes of IVF treatments with PGS by NGS, after two years providing this genetic test as part of a cooperation supported in a framework agreement. The upcoming Annual Meeting of ESHRE, European Society of Human Reproduction and Embryology (Helsinki, 3 to 6 July) will be the chosen context for this release, being a key event for the fertility community in Europe and worldwide.
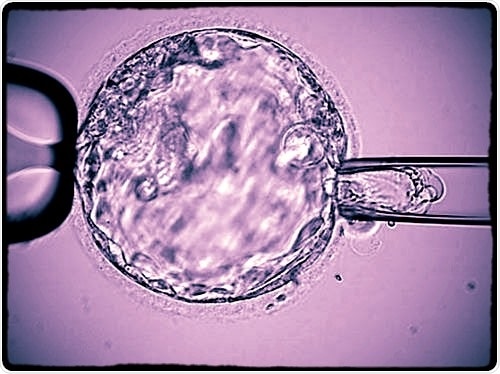
Blastocyst biopsy for PGS
PGS, Preimplantation Genetic Screening, is a genetic test that analyses biopsied cells from embryos produced by in vitro fertilization (IVF) techniques. PGS determines whether the embryos are chromosomally normal (euploid) or not, thus giving the chance to transfer chromosomally normal embryos that are more apt to successfully implant and develop into a pregnancy.
In 2014 Bioarray developed, validated and released PGS using NGS, Next Generation Sequencing, currently the most advanced molecular technology for the study of human genome. Since then, the alliance between Bioarray genetic lab and IVF-Spain fertility clinic has provided PGS to more than hundred European couples, with the first born baby screened by this method in September 2015. To date an overall of 64 born babies and 53 ongoing pregnancies have been achieved.
Moreover, this alliance has also provided PGD, Preimplantation Genetic Diagnosis by NGS for a number of couples carrying a single-gene disorder, who had their embryos checked to prevent the disease being passed to their children. In this regards Bioarray integrated PGD setup + PGD + PGS in a single fast robust NGS workflow, providing unparalleled benefits to patients.