Experiments conducted at the University of California, Berkeley, suggest that the odor of food has a direct link with how the body processes the calories.
The new study conducted on mice published in the first week of July in the journal Cell Metabolism implicates that the ability to detect the smell of food is related to whether the calories are burnt rather than stored.
In the experiment, obese mice that had lost their sense of smell also lost weight, whereas mice that retained their sense of smell ballooned to twice their normal weight. Also, super-smeller mice (mice with boosted sense of smell) got fatter than mice with a normal sense of smell when provided with a high-fat diet. Both the smell-deficient mice as well as mice with a sense of odor were fed with the same amount of fatty food.
The findings of the experiment point to a key link between the olfactory system and the regions of the brain that regulate metabolism, especially the hypothalamus, but the neural circuits related to this mechanism remain unknown.
Human become anorexic when they lose their sense of smell caused by aging, injury, or disease like Parkinson’s. Yet, the causes are unclear as loss of pleasure in eating can lead to depression, which in turn results in loss of appetite.
This study implies the role of loss of smell and suggests possible interventions for people who have lost their sense of smell as well as those who would like to lose weight.
"This paper is one of the first studies that really show if we manipulate olfactory inputs we can actually alter how the brain perceives energy balance, and how the brain regulates energy balance," said Céline Riera, a former UC Berkeley postdoctoral fellow now at Cedars-Sinai Medical Center in Los Angeles.
She also points out that sensitivity to smell is more when hungry than after consumption of food in both mice and human; possibly, the lack of smell tricks the body into thinking that it has already eaten. The body stores calories when it is unsuccessful in searching for food, and feels free to burn it when food is secured.
"Weight gain isn't purely a measure of the calories taken in; it's also related to how those calories are perceived," said senior author Andrew Dillin, the Thomas and Stacey Siebel Distinguished Chair in Stem Cell Research, professor of molecular and cell biology and Howard Hughes Medical Institute Investigator. "If we can validate this in humans, perhaps we can actually make a drug that doesn't interfere with smell but still blocks that metabolic circuitry."
Researchers worked on the destruction of olfactory neurons in the nose of adult mice using gene therapy, but the stem cells were left alone so that the animals would only temporarily lose their sense of smell for about three weeks after which the olfactory neurons would regrow.
Upregulation of the sympathetic nervous system increases fat burning, and this was exactly mimicked in the smell-depleted mice. The beige fat cells (subcutaneous fat storing cells accumulate on the circumference of our thighs and midriffs) are turned into brown fat cells in mice, which is capable of generating heat through burning of fatty acids. A few mice became lean, mean, burning machines after they turned all their beige cells into brown cells.
These mice were reported to have shrunk their white fat cells (a storage cell) that clump around the internal organs which are connected with poor health.
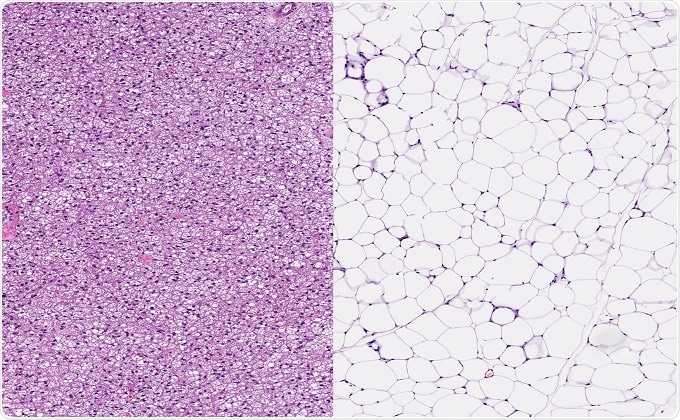
Image: (Left): Normal healthy brown fat (left image) and normal healthy white fat. Brown adipose tissue or hibernating gland with small fat vacuoles. (Right): White adipose tissue with large fat vacuoles ©vetpathologist
The obese mice with glucose bigotry (a status that brings on diabetes) not only lost weight in spite of a high fat diet but also achieved normal glucose tolerance.
The smell loss, in contrast, leads to increased noradrenaline levels resulting in a stress response that is linked with the sympathetic nervous system. Such a continuous raised level of noradrenaline could result in a heart attack in humans.
This might be a drastic step when smell loss is implemented in humans trying to lose weight. Dillin noted that for the morbidly obese contemplating stomach stapling or bariatric surgery, this might be an alternative method, even with increase in noradrenaline. He said: "For that small group of people, you could wipe out their smell for maybe six months and then let the olfactory neurons grow back, after they've got their metabolic program rewired."
Two different techniques to block the sense of smell in adult mice temporarily were developed by Dillin and Riera. In the first technique, they engineered mice genetically in order to express a diphtheria receptor in their olfactory neurons that reaches the olfactory center in the brain from the nose's odor receptors. The neurons died when diphtheria toxin was sprayed into the nose of the mice, making it smell-deficient until the regeneration of the stem cells.
In the second technique, a benign virus was engineered to carry the receptor into olfactory cells only via inhalation. Again, the toxin prevents their smell for nearly three weeks.
Both the smell-deficient mice and the mice that could still smell ate equal amounts of high-fat food in the two techniques. However, the smell-deficient mice gained only a maximum of 10% extra weight (25–30 g to 33 g), while the other type ballooned with a gain of 100% of their normal weight (60 g). Insulin sensitivity as well as response to glucose, which are usually disrupted in metabolic disorders like obesity, remained normal in the former.
For obese mice, after their smell was knocked out, they lost fat weight, slimming down to the size of normal mice even if they were provided with fatty diet; but had no effect on muscle, organ, or bone mass.
The researchers at UC Berkeley by teaming up with colleagues in Germany, who have a strain of mice that are super-smellers with more acute olfactory nerves, discovered that the mice gained more weight on a standard diet than the normal mice.
"People with eating disorders sometimes have a hard time controlling how much food they are eating and they have a lot of cravings," Riera said. "We think olfactory neurons are very important for controlling pleasure of food and if we have a way to modulate this pathway, we might be able to block cravings in these people and help them with managing their food intake."
Source:
https://www.eurekalert.org/pub_releases/2017-07/uoc--syf070317.php