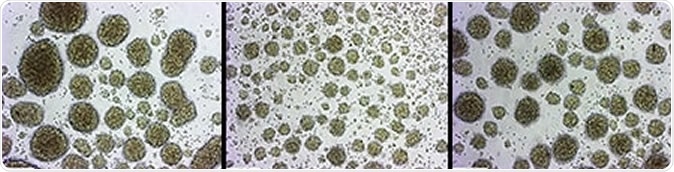
3D human brain organoids. Left: normal, uninfected. Center: infected with Zika virus. Right: infected with Zika virus and treated with cilengitide, which protects the cells from destruction by the virus. Image Credit: UC San Diego Health Sciences
The Zika virus causes an infection that can seriously impair the development of the fetal brain. An epidemic affected several areas of the world in 2014-15, especially in South America. In 2019, Zika infection hit Europe for the first time. Transmitted through mosquito bites, it also spreads from pregnant mothers to the babies in the womb, and through sexual intercourse, putting half the world’s people at risk as a result. There is no cure for this infection, and scientists have been studying how the virus makes its entry into brain cells in order to develop a preventive and therapeutic strategy.
As part of such research, two different teams at the University of California San Diego School of Medicine approached the question in two different ways but came up with the same answer in a surprising meeting. What they found was that the Zika virus uses a molecule called αvβ5 integrin to enter the brain stem cells.
Zika virus in blood with red blood cells, a virus which causes Zika fever found in Brazil and other tropical countries. Image Credit: Kateryna Kon / Shutterstock
Integrins – what are they?
Integrins are molecules that are found within the cell membranes bounding the different cells of the body. They are essential for helping cells adhere to each other and to underlying fascia or tissue, and in facilitating cell-cell communication. These molecules are therefore also involved in the loss of cell adherence and abnormal cell signalling that occurs in spreading cancer, where cells lose their mooring and travel from their host organ to a distant location to proliferate in an uncontrolled fashion and produce a metastatic growth.
Many other integrins have already been found to offer weak points through which adenoviruses, foot-and-mouth disease viruses and rotaviruses sneak into the cells to cause infections. This pair of studies offer the first proof that the αvβ5 integrin is involved in enabling infection by the Zika virus.
Study One
The first approach, published in the journal Cell Reports, was by systematically deleting genes one by one from cultured human glioblastoma cells in a 3D culture. Glioblastomas are particularly nasty brain cancers. Each of the mutant cell cultures was exposed to the Zika virus to see how it responded. This enabled them to detect the key genes and encoded proteins for viral entry into the cell. The virus was labelled with green fluorescent protein (GFP) to make its detection easier following infection.
The experiment showed that the Zika virus requires not less than 92 separate human genes found in brain cancer stem cells to enter and replicate within brain cells. However, among the crowd, the αvβ5 integrin-encoding gene attracted a lot of attention. This is because it is the only integrin that occurs at high levels in brain stem cells - the very cells that are most readily infected by the virus.
Researcher Tariq Rana explains, “I was expecting to find Zika using multiple integrins, or other cell surface molecules also used by other viruses. But instead we found Zika uses αvβ5, which is unique. When we further examined αvβ5 expression in brain, it made perfect sense because αvβ5 is the only integrin member enriched in neural stem cells, which Zika preferentially infects. Therefore, we believe that αvβ5 is the key contributor to Zika's ability to infect brain cells.”
Study Two
In the second experiment, which was published in the journal Cell Stem Cell, the scientists used one specific antibody against each of the many integrins to find out which inhibitor would act most powerfully. Researcher Jeremy Rich describes the results: “When we blocked other integrins, there was no difference. You might as well be putting water on a cell. But with αvβ5, blocking it with an antibody almost completely blocked the ability of the virus to infect brain cancer stem cells and normal brain stem cells.”
They then used two methods to inhibit the integrin - using a blocking antibody and disabling its encoding gene.
They observed that both methods prevented Zika infection, and prolonged survival of treated mice compared to exposed untreated mice. Using glioblastoma cells from surgical patients, they also found that inhibition of this integrin blocked Zika virus infection of the cell.
At this point the stories converge, because the first team also used two experimental cancer drugs called cilengitide and SB273005 in mice to block the αvβ5 integrin. The mice thus treated were then exposed to Zika virus. After 6 days of daily treatment, the researchers found that there was only half as much of the virus in the brains of the treated mice as in the brains of controls.
The findings not only add to the knowledge about the virus but also open up a new field of possibilities in antiviral strategies – blocking the entry of other similar viruses into specific tissues by inhibiting integrins, according to Rana, whose team is now working on developing a unique mouse model. This animal will not have αvβ5 integrin in the cells of the brain. This will prove once for all that this molecule is essential for the entry and replication of the virus in brain cells.
αvβ5 integrin and brain cancer
Talk of serendipity! When Rich first saw the effect of Zika virus on the developing brain, shrinking it drastically, he saw something different – a possible way to kill glioblastoma cells, a goal he has been pursuing for years. This led to a 2017 study which showed that the Zika virus prefers to infect and kill glioblastoma cells. This observation was exciting because glioblastomas are not only very aggressive tumors that have a high mortality rate but are also poorly responsive to standard therapies. This is the major reason for their poor outcomes, with most glioblastomas, if not all, being found to recur after treatment.
The current study by Rich explains why glioblastoma stem cells are preferred by the Zika virus to other brain cells. The scientists found that there are two parts to the αvβ5 integrin, namely, -- αv and β5. Both are expressed at high levels by glioblastomas. However, the first subunit is typically expressed by stem cells, while the second is linked to cancer cells. The complete molecule is essential for glioblastoma cells to survive.
For this reason, the Zika virus preferentially infects and kills glioblastoma stem cells compared to either normal brain stem cells or other types of brain cell. In other words, says Rich, “It turns out that the very thing that helps cancer cells become aggressive cancer stem cells is the same thing Zika virus uses to infect our cells.”
By transplanting human glioblastomas into 3D brain cell cultures or brain organoids, they tested their theory in a more realistic way. They found the Zika virus selectively removed glioblastoma stem cells from the organoids, but this was reversed simply by blocking αvβ5 integrin. Since this inhibition blocked the removal of the cancer cells, it prevented anticancer effects.
The researchers have thus leveraged their findings by exploiting it to block virus entry into brain cells and also how to use this integrin to shrink cancer cells in the brain. They would like to increase the specificity of brain cell destruction by engineering viral genes.
Future research will be twofold: engineering the Zika virus to improve the safety profile in its use in brain tumors and look for drugs that block the integrin to prevent Zika infection.