A kidney transplant is a lifesaving but complicated procedure. There are many different points at which it can fail. Of these, the most dreaded complication following a successful surgery has to be organ rejection – when the patient’s body rejects the new kidney as a foreign object. The immune system starts producing antibodies to try and kill the new kidney. This not only wastes a precious donor kidney but can lead to the patient’s death.
To prevent this, doctors usually prescribe medications that reduce the strength of the immune response, called immunosuppressives. This decreases the probability of rejection. There’s a downside, though.
The problem with immunosuppressive drugs
The disadvantage of such drugs is that it disarms the very system that enables us to function normally even while exposed to millions of bacteria, viruses, and other germs that, if allowed, would enter the body and harm or even kill the organism. What keeps them at bay is the immune system – the finely tuned natural defenses of the body. Sometimes it fails, as when the body is exposed to a new and unknown threat (like COVID-19), or it’s taxed beyond its strength. With the immune system no longer operating at full strength, the drawbridge is down – we are much more susceptible to external threats. This is why transplant doctors have to toe a fine line between too little dampening (resulting in organ rejection), and too much (which allows the infection to gain a hold in the body). Moreover, the infections which occur in immunosuppressed patients are just ordinary ones for the most part, which in a healthy person would have been dispelled with ease.
Doctors have to monitor their patients exceptionally closely to ensure that the drug is at the right concentration to prevent either of these things from happening. Such monitoring is usually done via kidney biopsies and blood tests – both expensive, invasive, and time-consuming.
A new way
Urine testing is much simpler and less expensive than blood and kidney tests. And now, scientists have combined this modality with the powerful technology called CRISPR to sense the presence of molecules that signal infection. CRISPR is more usually associated with gene editing. Still, the current use could result in a highly sensitive diagnostic tool to detect early signs of rejection, just like the more common urine tests that screen for urine sugar, pregnancy hormones, and the like.
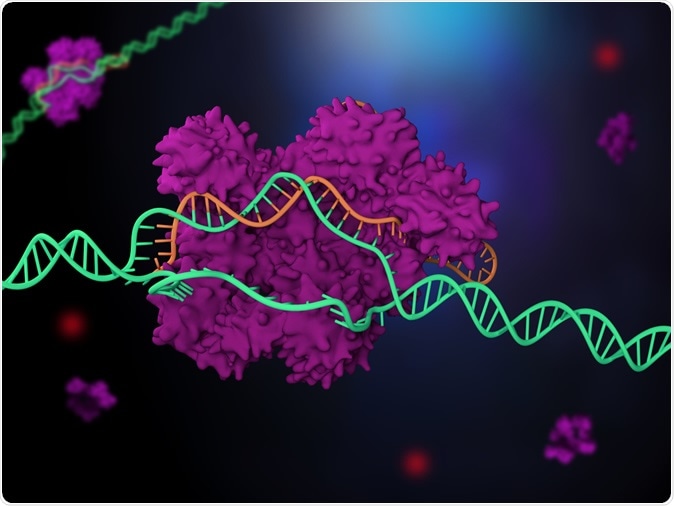
3d illustration of CRISPR-Cas9 genome editing system. Illustration Credit: Meletios Verras / Shutterstock
To adapt a urine test for this purpose, it has to be able to detect nucleic acids – DNA or RNA. And this is why CRISPR becomes so useful because it is able to find tiny segments of nucleic acid with a particular sequence if there is a complementary piece of guide RNA to help it out. It is coupled with an enzyme called Cas, which occurs in several forms, and makes cuts to isolate the sequence being sought. It is also bound to a fluorescent molecule called a reporter, because it lights up when the target sequence is cleaved.

The lateral flow strips show three patient samples that are negative for BK virus (13,14,15) and three patient samples that are positive (16,17,18). Presence of the upper band indicates a positive test result. Image Credit: Michael Kaminski, MDC
Many researchers have proved that CRISPR can pick up diagnostically useful information on man-made samples, but real-world clinical testing is rare. This involves achieving sufficient sensitivity to detect extremely low concentrations, which are typically what is found in biological samples.
Researcher Michael Kaminski comments: “The challenge is getting down to concentrations that are clinically meaningful. It really makes a huge difference if you are aiming for a ton of synthetic target in your test tube, versus if you want to get to the single-molecule level in a patient fluid.”
How is CRISPR-aided urine testing done?
The urine test kit or assay is performed in two stages. The first involves the amplification of viral DNA of the two most common opportunistic pathogens, namely, cytomegalovirus (CMV) and the BK polyomavirus (BKV). This process simply means making multiple copies of a small amount of target DNA to get a high enough concentration so it can be detected by the CRISPR. The amplification proceeds to the extent required to allow the detection of even one target molecule. This is achieved by a CRISPR-Cas13 program called SHERLOCK, which adapts the process for use with viral DNA.
The assay kit is a paper strip that acts somewhat like a home pregnancy strip. Two lines indicate the presence of a virus when the strip is dipped in the sample that has been prepared for testing. If the target sequence is present but at very minute levels, a confusing pale second line may appear. To avoid this, they also designed a smartphone app to ensure a thoroughly impartial analysis of the strip with the result reflecting the intensity of the line.
The steps were repeated this time using the biomarker CXCL9, which signals organ rejection. The mRNA from the sample is isolated and then amplified before detecting it by the use of CRISPR-Cas13.
The researchers refined the technique, testing out the analysis on over 100 real-life kidney transplant samples. They found that it responded to even very low concentrations of BKV or CMV, both of which are linked to acute rejection response mediated by cellular infiltration.
Ex-vivo embryonic kidney culture. Image Credit: Michael Kaminski, MDC
What’s up next for CRISPR?
The researchers are applying for a patent. Kaminski is also planning to perform a clinical study of sufficient size to make a meaningful contribution to the field. This will compare the current test with conventional methods of monitoring these patients. The scientists are working on a smoother, less complicated testing protocol, without having to heat the samples before analysis.
Such dreams could take the test to home-testing level, where a single strip just needs to be tested to obtain quantitative results of various biomarkers. This will allow changes in each area to be compared against the baseline for each marker.
The researchers say, “The assay enables—via simple visualization—the post-transplantation monitoring of common opportunistic viral infections and graft rejection, and should facilitate point-of-care post-transplantation monitoring. Not only will this test be a boon for other people with weakened immune systems, but its CRISPR-mediated diagnostic capability could also lead to the development of new tests for other types of organ transplants as well."
Journal reference:
Kaminski, M.M., Alcantar, M.A., Lape, I.T. et al. A CRISPR-based assay for the detection of opportunistic infections post-transplantation and for the monitoring of transplant rejection. Nat Biomed Eng (2020). https://doi.org/10.1038/s41551-020-0546-5