The COVID-19 epidemic that began at the end of 2019 in China’s Wuhan city has now spread to over 185 countries and territories, taking over 177,000 lives out of approximately 2.5 million reported cases. Without either a vaccine or a reliable cure at hand, public health authorities are stretched to contain the virus spread.
Currently, many countries are enforcing social distancing and quarantine measures to prevent the pandemic from spreading too fast, overwhelming the healthcare system. Some have gone into lockdown mode to try and prevent the community spread of the virus.
However, such measures depend heavily on accurate knowledge of the incubation time of the virus and of the duration in which an individual remains infectious.
-1.jpg)
Novel Coronavirus SARS-CoV-2 Colorized scanning electron micrograph of an apoptotic cell (blue) infected with SARS-COV-2 virus particles (red), isolated from a patient sample. Image captured at the NIAID Integrated Research Facility (IRF) in Fort Detrick, Maryland. Credit: NIAID
What is the incubation period?
The incubation period is conventionally described as the period after an individual becomes infected in which no symptoms are developed. Incubation periods for many acute respiratory infections range from 2 days to 2 weeks.
Previous estimates arrived at a median incubation period of 5.1 to 5.8 days, which could range from 2-24 days, depending on the study. The incubation period can only be appropriately calculated if contact tracing is carried out efficiently. Contacts are asymptomatic or symptomatic individuals who have been exposed to the infection.
The issues with contact tracing-based incubation
All symptomatic individuals are taken back over their history to find the day when they were possibly exposed to an infected individual or case. This is a cumbersome and exacting procedure, and when the clinical burden of the symptomatic illness is great, it is difficult to scale it up. The scaled-up version of this program would involve collecting history of exposure from hundreds of thousands of individuals.
A more significant issue with the current pandemic is the very high proportion of asymptomatic cases, up to 80%, according to the state of current knowledge. Adding to all this is the limited number of patients traced in this way, making the possibility of error due to the large ‘tail’ ends of the distribution curve much higher.
Lockdown has been observed in almost all countries affected. Nonetheless, new cases keep cropping up, in a manner that is difficult to explain unless the estimate of the incubation period is wrong. This could be due to the low number of cases studied, leading to unreliability.
Why was this study done?
This new study, published on the preprint server medRxiv, attempts to answer the questions:
“Is there an alternative way to estimate this incubation period even if one did not have detailed exposure histories? Or is there an alternative estimation one needs to do at the level of the community with the best quarantine practices possible? Are the new cases in a lockdown scenario arising from incubation or because of other factors?”
How is the incubation period estimated?
The answer involves a new method of estimating the incubation period using only epidemiological data available to the public. This is based on the reported number of infections, which allows a double advantage: first, exposure history is not required; and tens of thousands of reported cases comprise a much more extensive database, enhancing the reliability immensely.
The trend seen with the use of this database is an increase in the number of new cases each day for a brief period following the lockdown, followed by a fall. However, the ideal scenario calls for people infected in the days before the lockdown to become symptomatic certain days afterward, once their incubation period is over.
The researchers consider that the number of new infections each day after the lockdown, say, on day 1, will be derived from the number of asymptomatic infections on day 0 who had an incubation period of 1 day, plus the number of infections the day before (-1) with a 2-day incubation, etc.
They derive the following equation to predict the number of infections I*(t) on any day (t) after the lockdown starts, namely,
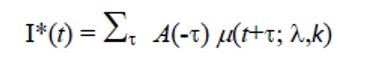
Here μ(t+t; l,k) is the distribution of the incubation period in a Weibull distribution, with the parameters l and k.
Again, the researchers made the assumption that on any day before the lockdown, the number of asymptomatic infections would be in proportion to the reported infections. They analyzed the observed and predicted infections after lockdown for various values of λ and k. They took the values of these parameters, which had the highest regression coefficient as the most likely estimate of the incubation period.
The researchers estimate that the values λ =16, k=2 for the parameters is most predictive for the infections occurring after the lockdown based on those which occurred earlier, in Austria, Germany, and Spain. Based on the epidemiological data, they estimated the incubation period in the community, which is at least theoretically the same as that of the individuals under an ideal quarantine situation.
How do new cases arise beyond the incubation period?
The incubation period now accepted is not enough to explain all the new cases cropping up in different places. For instance, the new infections that have occurred after the announcement of a lockdown in Italy are in excess of what should have been observed from the incubation period of 13.3 days. The same has not occurred in Switzerland, which borders Italy, though the infections in both countries ought to be similar.
The researchers say, therefore, that it is necessary to find reasons for the excessive number of new post-lockdown cases.
This distribution could “be considered as a surrogate of the incubation time in a perfect lockdown”, in which case, even a perfect quarantine for 30 days would keep only 90% of cases separate from healthy people.
The reasons for the longer incubation time may be either a truly longer incubation period that was not picked up by the earlier contact tracing of only a few individuals. Or the failure to maintain an ideal lockdown could lead to a falsely long incubation period because healthy individuals keep being exposed to infected asymptomatic individuals beyond the expected date of exposure.
The researchers favor the former reason, but they point out that regardless of the true reason, their estimation method can yield the same kind of conclusion.
In other words, they say, “Although physiologically it may slightly differ from the incubation of the individual, this incubation at the community level with the lowest possible societal interactions has its own meaning.” This means that the incubation period at the community level is more important in deciding the term of lockdown and quarantine measures.
What does the study show?
The researchers say, “We introduced a new way of estimating the incubation time by studying the new infections that arise after measures that restrict the interactions between the individuals in the community to a minimal or non-existent level.” This is an easy method, and when conducted for three countries in Europe, the median incubation period was 13.3 days.
A more extended incubation period is probably present, according to the current study, between 2 and 16 days. This will mean that to ensure the complete containment of viral spread from asymptomatic cases, the period of lockdown will need to be extended beyond 30 days.
This should serve as a means to estimate community-level incubation over larger areas, to decide on public health measures. This could help distinguish how new cases arising after the incubation period from those which are truly newer.
Important Notice
medRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.