In common with SARS-CoV, the current virus, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), also appears to undergo indoor superspreading by aerosols. This is supported by the observation that disease spread is 20 times higher indoors relative to outdoors, and by the fact that many earlier viruses, including respiratory syncytial virus (RSV), Middle East Respiratory Syndrome coronavirus (MERS-CoV), and influenza have been shown to spread this way. However, this is a topic of considerable controversy.
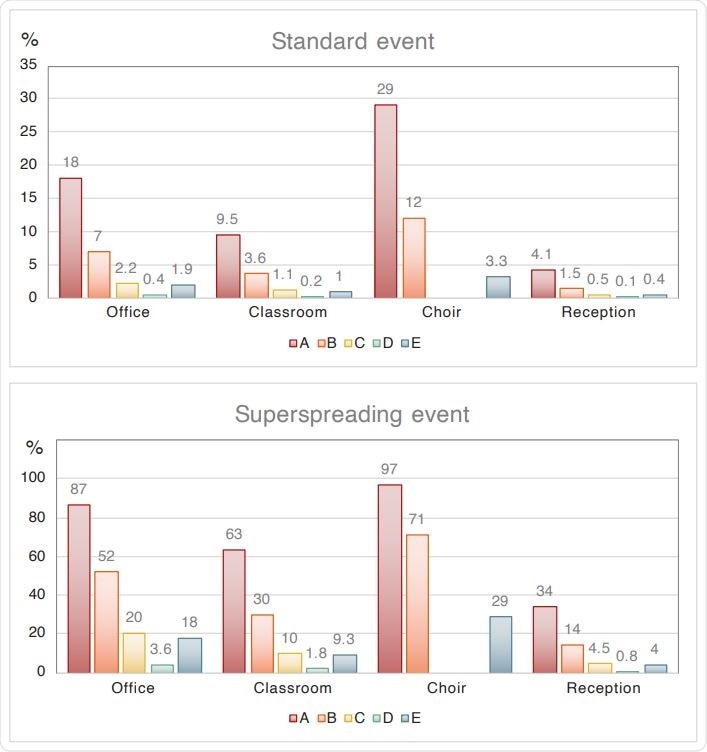
Individual risk of a particular person being infected (equivalent to the fraction of the group being infected) in four indoor environments and five scenarios, for standard and superspreading conditions. Scenario A: passive ventilation, no masks. Scenario B: active ventilation with outside air, no masks. Scenario C: active ventilation, facial masks (not for choir). Scenario D: active ventilation, high-quality masks (not for choir). Scenario E: High-volume filtration with HEPA.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Estimation of Indoor Risk due to Aerosolized Virus
The current study presents a versatile and straightforward but easily understandable spreadsheet algorithm that is used to estimate the chances of indoor viral spread through aerosols. The parameters incorporated into the algorithm include the size of the room, the number of people exposed to the virus, the volume of inhaled air, and the characteristics of aerosol generation via breathing, speaking, singing, and the like.
The viral load and infection dose parameters are based on assumptions made from earlier studies. The study focuses on aerosols, leaving droplets spread out altogether. Each case scenario envisages a single infected person in either the presymptomatic phase, which is thought to be highly infectious or with asymptomatic infection, where infectivity is not clearcut.
The researchers define highly infectious to mean 5×108 viral (RNA) copies/mL, as observed in about one in five positive cases.
Two alternative scenarios are considered: in one, the index patient is very infectious, while in the other, a superspreading event occurs either due to a tenfold higher viral load relative to the former index patient or because of extremely high aerosol production.
Office Spread
The researchers assume that with four individuals being present, one the index case to which the others are exposed, the period of high infectivity is two days at 8 hours each. If only passive ventilation is present, there is a 45% and 18% risk of one other being infected, and of individual infection.
The authors clarify, “The individual infection risk equals the fraction of the group that is at risk, thus being increasingly significant with the number of subjects present.”
Under conditions of active ventilation from outside air, both these risks are 2-2.5 times lower, while with the use of face masks, they decline by 7-8-fold. With high-volume HEPA filtering (HEPA), the risk is 8-9-fold lower while it is 40 times less if high-quality masks are worn.
If a superspreading event occurs, 2-3 individuals would be infected, but with active ventilation only 1-2. Individual risk is 4-5 times less with either masks or HEPA ventilation, while high- quality masks bring down the risk 25 times.
These conditions can be replicated in other surroundings of similar size with the same number of people, such as a hospital ward or nursing home room.
Classroom environment
Young children below ten years of age may be less infective, so the researchers decreased the viral load by 10 for this case. The risk of infection is 21%, while for each individual, it is about 1%. For older students, the overall and individual risk is 91% and 10%, respectively.
The risk is reduced with ventilation from outside for a classroom day of six hours, and for older children, it becomes 58%. In this group, the individual risk drops to 1% if face masks are used.
For high-risk individuals in these surroundings, HEPA may drop the individual risk by 9-10 times, and still, more if other measures are used. This is expensive, of course, and may be used only for this group. Superspreaders among either students or teachers may cause a 63% risk of infection, but this is reduced 6-7-fold by active ventilation and mask-wearing, even more, if high-quality masks are used, though this is unrealistic.
Choir practice
Due to the high aerosol emission during singing compared to breathing, a single highly infectious index case can infect 29% of the others, half this number if active ventilation is present, and 9-fold with HEPA. Face masks are not used in this setting.
Superspreaders can result in infection rates of 71% with active ventilation and 29% with HEPA.
Reception
Guests at a reception desk speak loudly compared to people in an office or classroom, but the space is usually bigger, and the exposure time is shorter. Without other measures, about 4 infections may be expected from the index case but two in the classroom. Active ventilation and HEPA will progressively reduce this risk by 2.5 to 3 times and 10 times, respectively. Face masks and good-quality masks also reduce the risk.
If a superspreading event occurs, about 34% may be infected, but with active ventilation, 14%, and 9% with HEPA. Unless high-quality masks are used, four or more guests are likely to be infected in all these conditions.
If more ordinary conditions are envisaged, as when alcohol is served, a band is present, and dancing is held, the reception is more of a party, and the risk is correspondingly higher.
Cluster Infections
If only one index case affects many individuals, a cluster of over 10 COVID-19 cases can break out, which usually occurs with superspreading. This must be rapidly prevented from spreading further. A party at a reception may cause cluster infections even without a superspreader. Since face masks are not usually worn at such parties, HEPA may be advisable to contain the spread by 9-fold, to about 3 cases instead of 34, and thus prevent cluster infection.
Though schoolchildren may be less infective, they also spend more time together and tend to speak loudly or shout, which may push up the overall cluster infection risk significantly.
Implications
Of course, these parameters need to be adjusted individually, since the aerosol production rate can vary significantly with the viral load, the type of breathing or vocalization, and the duration of exposure. Indoors, asymptomatic, presymptomatic, and mild COVID-19 cases are the primary sources of secondary transmission.
They comment, “Additional scientific research will need time to quantify infection risks and pathways in greater detail. However, by waiting for additional scientific results, valuable time will be lost that could be used to control the pandemic.”
The researchers also glance at the benefits of using masks, especially if they are multi-layered and contain fluffy material, and more during forceful or loud speaking compared to simple breathing.
Concerning prevention of infection, the most effective measures are high-quality masks and HEPA, but in terms of real-life protection, active ventilation and the wearing of face masks are preferable in terms of the cost and the results. High-quality masks are about 5 times more costly than others but are quite unlikely to be used either by choir singers or by party-reception guests.
Still, properly fitted and worn masks, with hygiene measures, represent the best hope, especially if active ventilation mixes room air with outside air, and if HEPA is used. This algorithm can be used to estimate risks. The study ends with an apt quote from De Kai, who said a “mouth-and-nose lockdown is far more sustainable than a full lockdown, from economic, social, and mental health standpoints.”

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Journal references:
- Preliminary scientific report.
Lelieveld, J. et al. (2020). Aerosol Transmission Of COVID-19 And Infection Risk in Indoor Environments. medRxiv preprint. doi: https://doi.org/10.1101/2020.09.22.20199489. https://www.medrxiv.org/content/10.1101/2020.09.22.20199489v1
- Peer reviewed and published scientific report.
Lelieveld, Jos, Frank Helleis, Stephan Borrmann, Yafang Cheng, Frank Drewnick, Gerald Haug, Thomas Klimach, Jean Sciare, Hang Su, and Ulrich Pöschl. 2020. “Model Calculations of Aerosol Transmission and Infection Risk of COVID-19 in Indoor Environments.” International Journal of Environmental Research and Public Health 17 (21): 8114. https://doi.org/10.3390/ijerph17218114. https://www.mdpi.com/1660-4601/17/21/8114.