Caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), coronavirus disease 2019 (COVID-19) is primarily a pneumonic illness – and in a large proportion of patients with severe cases, secondary bacterial or fungal infection is also present. In fact, about 25% of patients with severe acute respiratory distress syndrome (ARDS), and around 40% of patients with severe COVID-19 pneumonia, also show signs of infection with the fungus Aspergillus fumigatus in the lungs.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This fungus causes invasive lung infection, following the inhalation of asexual spores. There are over 250,000 aspergillosis cases every year, and these are associated with many deaths. This fungus is able to grow at the temperature of the human body, is resistant to oxidation, and rapidly develops resistance to antifungals.
The current study is aimed at understanding whether CAPA isolates are different from other A. fumigatus strains regarding their genetic make-up or phenotypic expression. The researchers examined four such isolates from four patients admitted to an intensive care unit (ICU) with ARDS due to critical COVID-19.
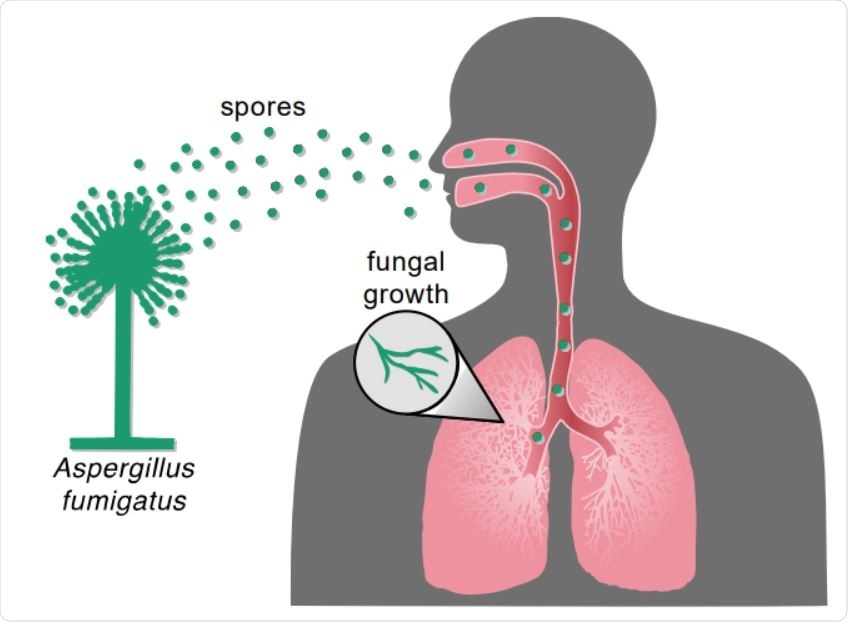
Inhalation of Aspergillus spores can result in fungal infection. Inhalation of Aspergillus spores from the environment can travel to the lung and then grow vegetatively and spread to other parts of the body.
CAPA isolates belong to A. fumigatus
The study found that all patients had infection with A. fumigatus strains. The CAPA isolates were also closely related, and appear to share a common ancestor. The CAPA isolates are also similar to the reference strains Af293 and CEA17.
All the 206 genes concerned with virulence in A. fumigatus were present in the CAPA isolates without copy number variation, but showed single nucleotide polymorphisms (SNPs) as well as indel mutations, which were correlated with variation in the predicted effect.
LOF mutations in virulence genes
The many polymorphisms seen in the CAPA isolates caused early termination or frameshift mutations, both of which may indicate loss of function (LOF) in NRPS8, which is a peptide synthase gene that is responsible for the production of an unknown secondary metabolite. Such mutations in these genes have been found to increase virulence in a mouse experiment.
Many of the (possible) LOF mutations also occurred among genes that had null mutants linked to decreased virulence, such as premature termination of the PPTA gene. Deletion mutants of the latter have been found to result in lower virulence in mice. The researchers pointed out that the monophyletic relationship among the four isolates means that the observed shared SNPs in PPTA in all CAPA isolates can be traced back to a mutational event in the genome of the latest common ancestral strain.
Isolate-specific polymorphisms
However, different isolates also showed specific polymorphisms affecting virulence. One such mutation affects CYP5081A1, a cytochrome enzyme in CAPA isolates B and C, and one in the FLEA gene in the D isolate. The FLEA null mutant is associated with more severe pneumonia and with a higher chance of invasive aspergillosis in mouse studies. This is because this gene binds to macrophages and its presence is required for host recognition by the virus, as well as for macrophage killing.
Biosynthetic gene clusters
Again, all the CAPA isolates had biosynthetic gene clusters (BGCs) that synthesize toxic or modulatory metabolites, such as gliotoxin, which was produced by all the isolates, or fumagillin, which is known to inhibit neutrophil function. All the CAPA isolates showed BGCs of equivalent class and in similar numbers.
The greatest number of LOF mutations among those genes associated with greater virulence were found among CAPA isolate D. This conclusion is supported by pair-wise comparison of pathogenicity among the isolates, which showed that this is the most virulent, with isolate C also showing markedly greater virulence than the reference strains.
The pathogenicity profiling of the CAPA isolates closely resembled that of the reference strains. They were more sensitive to the chemical calcofluor white compared to reference strains, but showed no significant difference in susceptibility to antifungals.
Conclusions
The researchers concluded, therefore, that the CAPA isolates are closely related, and contain genes for virulence with almost no attenuation. Moreover, these genes are expressed as would be expected of this fungus. However, there were genetic and phenotypic differences between virulence determinants of the different isolates.
The D isolate seems to be more virulent than other strains )both CAPA isolates and the reference strains) as assessed in an invertebrate infection model.
“We hypothesize that this difference is because the D isolate contains more putative LOF mutations in genetic determinants of virulence whose null mutants are known to increase virulence than other strains,” said the researchers.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Journal references:
- Preliminary scientific report.
Steenwyk, J. L. et al. (2020). Genomic and phenotypic analysis of COVID-19-associated pulmonary aspergillosis isolates of Aspergillus fumigatus. bioRxiv preprint. doi: https://doi.org/10.1101/2020.11.06.371971, https://www.biorxiv.org/content/10.1101/2020.11.06.371971v1
- Peer reviewed and published scientific report.
Steenwyk, Jacob L., Matthew E. Mead, Patrícia Alves de Castro, Clara Valero, André Damasio, Renato A. C. dos Santos, Abigail L. Labella, et al. 2021. “Genomic and Phenotypic Analysis of COVID-19-Associated Pulmonary Aspergillosis Isolates of Aspergillus Fumigatus.” Edited by Christina A. Cuomo. Microbiology Spectrum, June. https://doi.org/10.1128/spectrum.00010-21. https://journals.asm.org/doi/10.1128/Spectrum.00010-21.