Introduction
This guide on troubleshooting will enable users to get reliable results when quantifying nucleic acids through various devices available in the Eppendorf photometer series (Figure 1). Therefore, the important factors which help to attain accurate measurements and problem-solving recommendations are given below. Figure 1 shows a pictorial example of Eppendorf BioPhotometer® D30.
Figure 1. Eppendorf BioPhotometer D30 – one member of the Eppendorf Photometer family
Fundamentals of Measurements on Photometry – Beer-Lambert Law
In order to evaluate the sample liquid concentration in a cuvette, the transmission (T) is first determined with a photometer, while measuring the ratio of ingoing light (I0) and outgoing light (I1) (Figure 2). The absorbance (A) value is the common negative logarithm of transmission. The absorbance that is measured is based on optical path length; specific sample factor; absorbance coefficient; and concentration. This forms the basis of the Beer-Lambert Law.
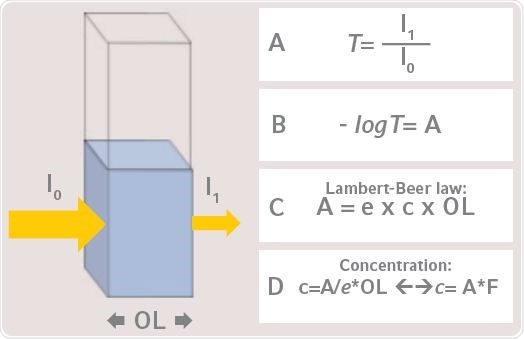
Figure 2. Measuring of light absorbance in a photometer with a liquid sample (Yellow arrow: Light beam of the photometer). A: Transmission, B: Calculation of absorbance, C: Lambert-Beer Law: A=absorbance, e= absorbance coefficient, c= sample concentration, OL=optical path length, D: Calculation of sample concentration via Lambert-Beer Law: F=1/e.
The sample concentration as in Figure 2D can be analyzed using this law. Table 1 enlists various prime parameters with their corresponding SI-units. Correct calculation of the concentration can be done when the SI-units of both absorbance coefficient and concentration coincide with each other.
Table 1. Physical constants/parameters and corresponding SI-units of the Lambert-Beer law
|
Absorbance coefficient
|
(Mol/L)-1* cm-1, (g/L) -1* cm-1, (mg/mL) -1* cm-1, (μg/mL) -1* cm-1
|
|
Concentration
|
mol/L, mg/mL, μg/mL, g/L
|
|
Optical Path Length (OL)
|
mol/L, mg/mL, μg/mL, g/L
|
Calculation Example for dsDNA
Measurements done in a cuvette with 1cm optical path length (OL) can be calculated for concentration values using the formula c=A*F (Figure 2D). The F factor is the reciprocal value of the coefficient of absorbance. The coefficient of absorbance is specific at specific wavelengths for specific samples, e.g. nucleic acid coefficient is valid at 260nm. Table 2 enlists the coefficient of absorbance of certain molecules of nucleic acids at a wavelength of 260nm with their corresponding specific sample factors.
Table 2. Factors and absorbance coefficients for nucleic acids
|
Molecule
|
Absorbance coefficient 260nm
|
Factor (in 1cm cuvette)
|
|
dsDNA
|
0.020 (μg/mL) -1 * cm-1
|
50μg/mL
|
|
RNA
|
0.025 (μg/mL) -1 * cm-1
|
40μg/mL
|
|
ssDNA
|
0.027 (μg/mL) -1 * cm-1
|
37μg/mL
|
A cuvette containing a dsDNA sample with an absorbance of 1 and optical path length of 1cm can be calculated for its concentration of 50µg/mL as follows:
Beer-Lambert:
A=e*c*OL < > c=1/(e*OL)*A < > c=1/e *1/OL*A< > c=F*1/OL*A< > c= 50µg*cm/mL* 1/1cm *1
= 50µg/mL * 1 = 50µg/mL
Beer-Lambert law:
A = e * c * OL
c = 1/(e*OL) * A
c = 1/e * 1/OL * A (1/e = F)
c = F * 1/OL * A (F = 50 (µg*cm)/mL, OL = 1cm A = 1)
c = 50 (µg*cm)/mL * 1/1cm * 1
c = 50µg/mL
The results measured for a sample of nucleic acid are analyzed using the Eppendorf BioSpectrometer® and Eppendorf BioPhotometer®D30. Before measuring, it is mandatory that the right parameters for evaluation are set. Using the Eppendorf BioSpectrometer and the Eppendorf BioPhotometer, these criteria can be directly edited before taking measurements (Figure 3). Figure 4 shows the measuring result of a nucleic acid using the Eppendorf BioSpectrometer or BioPhotometer D30.
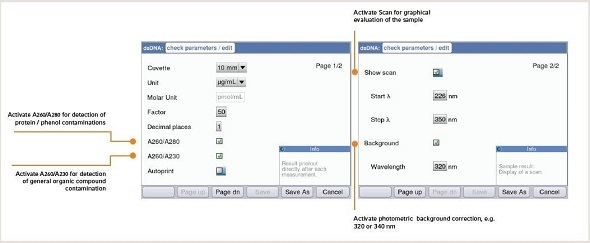
Figure 3. Parameter settings for nucleic acid quantification – Example BioSpectrometer.
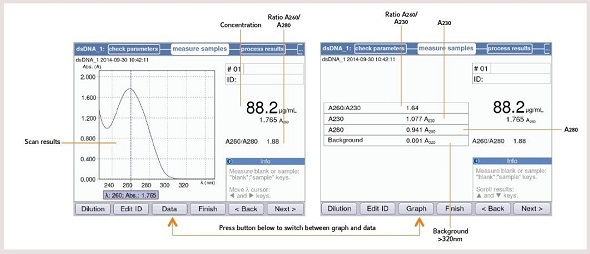
Figure 4. Measuring result of a nucleic acid sample with the BioPhotometer D30 or BioSpectrometer: You can switch between a graphical and table view of the result. In the table-view more details about the sample quality is displayed.
Evaluation of Measuring Results
Reading of absorbance at 260nm (A260) – Concentration of nucleic acid sample.
The following steps should be followed when evaluating the measuring results.
Check
If the measuring range of linear absorption of the sample is 0.05 – 2A, then the corresponding concentrations of nucleic acid should be as follows:
1cm (10mm): UVette® 10mm path length
- dsDNA: 2.5 - 100µg/mL
- RNA: 2 - 80µg/mL
- ssDNA: 1.85 - 74µg/mL
0.2cm (2mm): UVette® 2mm path length
- dsDNA: 12.5 - 500µg/mL
- RNA: 10 - 400µg/mL
- ssDNA: 9.25 - 370µg/mL
0.1cm (1mm): Eppendorf µCuvette® G1.0 with 1mm path length
- dsDNA: 25 - 1000µg/mL
- RNA: 20 - 800µg/mL
- ssDNA: 18.5 - 740µg/mL
Problem
Absorbance >2: Absorbance greater than 2 implies that the displayed result may no longer be in the linear range owing to the effects of stray light on the sample.
Solution
- Sample should be diluted until its absorbance lies in the linear range of 0.05 - 2.
- A cuvette having a shorter optical path length should be used: At high concentrations, the UVette can be turned using a path length of 2mm (Figure 5), or alternatively μCuvette G1.0 with path length of 1mm can be used (Figure 6). The principle to reduce the path length measurement is elaborated in Figure 7.
Problem
Absorbance <0.05: With extinctions lower than 0.05, all error sources become very significant to enable accurate measurements.
Solution
The sample can be concentrated, or if the sample is diluted, sample dilution can be reduced. For samples with very low concentration, the same can be measured by fluorescence, e.g. using the fluorescence Eppendorf BioSpectrometer.
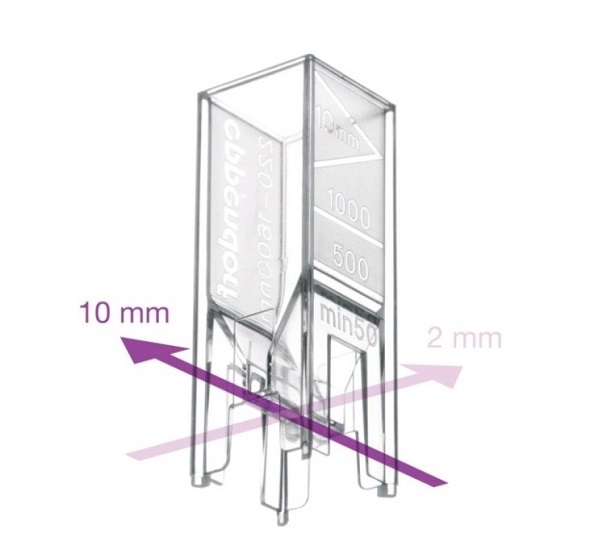
Figure 5. UVette with two optical path lengths: 10mm and 2mm.

Figure 6. Eppendorf µCuvette G1.0 1mm optical path length (orange circle).
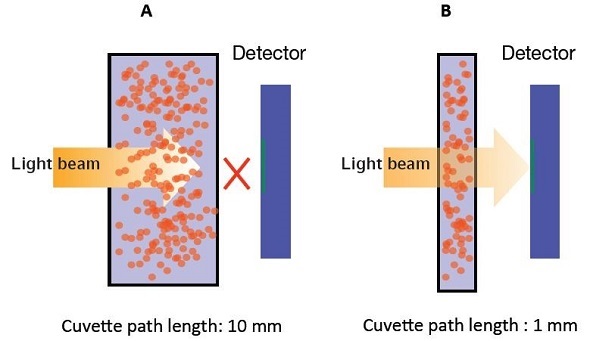
Figure 7. Smaller path lengths for higher concentrations.
The reading of absorbance to be checked at 280nm
The quality of the nucleic acid sample and potential contamination by phenols or proteins / determination of the ratio of purity A260/A280.
Check
The A260/A280 ratio can be analyzed by absorbance measurement at 260 and 280nm. Pure samples of nucleic acid should be in the ratio of 1.8 - 2.0.
Problem
A260/A280 <1.8: Since phenols or proteins show an absorbance which is high at this range, a very low ratio can possibly indicate sample contamination. For instance, a 1.5 ratio corresponds to a protein/DNA ratio of 50%.
Solution
Sample purification can be done, following which quantification using fluorescence can be tried.
Check the reading of absorbance at 230nm
The sample quality of nucleic acid and probable contamination by organic compounds / purity ratio determination A260/A230.
Check
By determining the absorbance at 230nm and 260nm, the A260/A230 ratio can be evaluated. This ratio must be >2.0.
Problem
A260/A230 <2.0: The value of the ratio must be greater than 2.0 for the RNA and DNA samples to be pure. Values which are less than 2.0 show contamination caused by organic solvents, salts, or sugars. A protein contamination will also influence the 260/230 ratio.
Solution
Sample purification can be done, following which quantification using fluorescence can be tried.
Check the reading of absorbance at >320nm
The photometric background and sample quality of nucleic acid.
Check
Above the wavelength 320nm, no nucleic acid absorbance is seen or probable organic compound contamination (e.g. proteins). A320 should be equal to 0.0.
Problem
A320nm >0.0: Pure sample of nucleic acid should not display absorbance within this range. If an absorbance is seen, photometric background is found in the sample.
Following are the reasons for quantified photometric background:
- Air bubbles
- Turbidity: precipitation, magnetic beads, microbial growth
- Insufficient liquid in the cuvette
- Absence of liquid column due to low volume (µCuvette)
- Unclean optical parts of the cuvette
Solution
If <0.03 photometric background is seen, the background correction of the photometer can be activated; all values measured are deducted by the value of absorbance procured at 320nm.
In case of >0.03 photometric background, the below steps should be followed:
- Clean the cuvette’s optical parts
- Purify the sample
- Increase sample volume
- Remove air bubbles
Figure 8 shows different kinds of contamination that could impact the measurement results of a nucleic acid sample at 260nm.
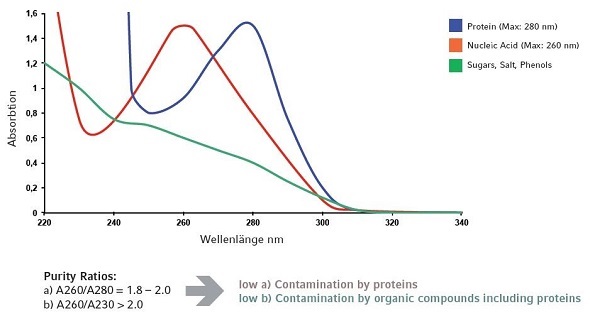
Figure 8. Influence on the measuring result at 260nm by protein or other organic compounds.
Frequent Error Sources
The Dilution
- A dilution factor of 1:10 to 1:50 is recommended, as higher pipetting error increases dilution ratios.
- Check if dilution factor has been rightly set
- Check for correctness of the pipettes through calibration
Sample Mixing
- The sample nucleic acid must be mixed prior to measuring and diluting in order to prevent potential sample concentration gradients.
- Before filling the cuvette with the sample, the same must be briefly vortexed before the measurement. This avoids fluctuation of concentration due to prolonged sample storage
The Measuring Medium
- The behavior of absorption of nucleic acids is governed by the pH value and buffer’s ionic strength
- Certain buffers display self-absorption in the UV range. This can be prevented by using the same buffer
Other Causes
- The device must lie flat to ensure that it is functioning properly
- The shaft of the cuvette must be kept clean
- The pipettes must be calibrated and cleaned properly
- The pipette piston must be cleaned to prevent sample contamination
Conclusion
When in doubt, users can perform a reference measurement instead of using a defined concentration of nucleic acid sample, or alternatively they can utilize the Eppendorf UV-VIS filter set along with different devices.
Acknowledgements
Produced from materials originally authored by Katrin Kaeppler-Hanno, Martin Armbrecht-Ihle and Ronja Kubasch from Eppendorf AG, Hamburg, Germany.
About Eppendorf NA
Eppendorf NA, with headquarters in New York, are a subsidiary of Eppendorf AG (Hamburg, Germany) – manufacturer of laboratory instruments and consumables for the life sciences.
To make it easy for labs in the United States to benefit from Eppendorf quality, they provide total customer support and service: taking/tracking orders; product installation and training; application support; calibration services; general product maintenance and repair, and more!
Eppendorf's products
Eppendorf products are used in all types of life science research and testing settings – from basic laboratory applications to highly specialized cell and molecular biology applications. They are highly regarded for their quality design and performance – beginning with extensive research and development, adding state-of-the-art technology and ending with strict quality-controlled manufacturing are what make their products stand out from the rest. It is what has made them a brand you have been able to rely on for over 70 years.
Over the years Eppendorf has improved upon and added to their flagship products – pipettes and pipette tips, centrifuges and microcentrifuge tubes – to now include the most ergonomic liquid handling devices and automated pipetting stations, quiet centrifuges, fast and fully flexible thermal cyclers, sample-protecting deepwell plates, cell manipulation systems and microcapillaries, ultra-low temperature freezers, shakers, incubators and bioprocessing equipment.
Eppendorf's services
Customer Support Representatives are available M–F, 8:30 AM – 8:00 PM (EST).
Local Sales Representatives cover the entire US to give you the fastest best possible attention. They know their products – and better yet, they know your applications. Feel confident in their abilities to assess your needs and determine the best products and systems to give you the best results.
Field Specialists also cover the entire US to provide post-sales installation and training on their specialty systems and automation.
The Applications Hotline is manned by degreed scientists working in their own fully functional life science laboratory. They are on call and email-accessible to help you troubleshoot your application or answer any technical question you may have. The Eppendorf Services group provides a wide variety of support services such as pipette calibration, instrument maintenance programs, and expedited repair.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.