Fentanyl manufactured by illicit means is driving an overdose epidemic, according to data released by the Centers for Disease Control and Prevention (CDC), which shows that more than half of all deaths due to overdose of opioids was caused by fentanyl made in small laboratories. This poses various enforcement problems, as well as for first responders and staff of forensic laboratories. Fentanyl is highly potent, as are its analogs. In fact, just a few milligrams of fentanyl can cause fatality, which makes it 50-100 times as potent as morphine. As for the analog carfentanil, it is 100 times stronger than fentanyl itself. This means that even accidental exposure to fentanyl or its analogs may endanger life whether by inhaling, ingesting or just being in skin contact with the drug.
For such reasons, CDC has put out a health alert dealing with the increase in accidental overdoses of these drugs following illicit production and sale, either as counterfeit pills or after being used to adulterate heroin. New opioid compounds are also being synthesized at a rapid pace by slight changes to the chemical formulae of the older analogs. This means there is great pressure to keep abreast of all the new drugs and to research their pharmacological and toxicological properties.
Multiple techniques are therefore absolutely necessary, both in the field and the laboratory setting to help detect, identify and screen for these synthetic opioids. According to CDC guidelines, a double-tiered testing is required for the identification of fentanyl in blood and urine, using an enzyme-linked immunosorbent assay (ELISA) followed by gas chromatography/mass spectrometry (GC/MS). In many situations, however, the concentration of the drug may be very low, and many other drugs may also be present. This requires the availability of sensitive technology for low limit detection, for which ion mobility spectrometry (IMS), liquid chromatography/mass spectrometry (LC/MS), and thermal desorption direct analysis in real time mass spectrometry (TD-DART-MS) are being studied.
A technique which has proved its worth over decades of illicit drug detection and identification is Raman spectroscopy, and is now being actively explored for use in this area of fentanyl and its analogs. FT-Raman spectroscopy goes a step ahead by using a laser of increased wavelength, 1064 nm, which yields less fluorescence while generating spectra with high signal-to-noise ratios. This is extremely useful in this field as narcotic samples often show fluorescence, and also because it allows sampling of drugs in a range of containers with little or even no sample preparation at all, through blister packs made of polymer, plastic evidence bags or glass vials.
Moreover, it does not destroy the sample, keeping the custody chain intact. This means that the sample can be evaluated qualitatively and quantitatively to make sure it has not been tampered with from the time it was seized until it is put into the evidence file.
In this article, the results of a set of FT-Raman spectroscopy analyses dealing with fentanyl and other illicit drugs is presented, along with a description of the advantages of this method.
Materials and methods
All the samples came through the Albuquerque Police department after preparation. They were therefore handled as per the guidelines of Schedule I controlled substance handling. They were obtained inside double bags, blister packs or glass vials. The analyses in all cases were performed using a Thermo Scientific™ Nicolet™ iS50 FT-IR spectrometer fitted with a calcium fluoride beam splitter as well as an FT-Raman module that had a 1064 nm laser, as shown in Figure 1. For each spectrum, a cycle of acquisition took about 75s, including 64 scans coadded at a resolution of 8 cm-1, the laser power being set at 0.5 mW. The spectra were then compared with data available in the Thermo Scientific Law Enforcement and Security (LEnS) Raman and DEA Raman libraries, with the help of the OMNIC library search and Specta multicomponent search tools.
Results and discussion
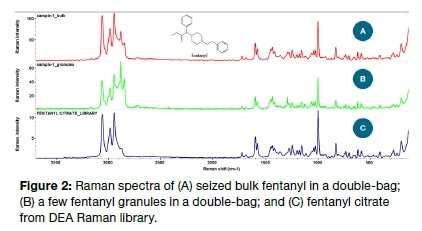
The Raman spectrum obtained from double-bagged bulk fentanyl by direct acquisition, in the absence of any sample manipulation, is shown in Figure 2A. The small spot size of the laser, approximately 60 microns, allows easy analysis of even a few grains of the analyte, as seen in Figure 2B. The only difference of significance is the small increase in noise in Figure 2B, yet this did not pose any difficulty in having both definitely identified as fentanyl citrate following a library search.
The advantages of such an analysis are that it reflects both the chemical and physical properties of the analyte, while it follows unique selection criteria that result in the production of a molecular fingerprint specific enough to distinguish illicit compounds from their analogs.
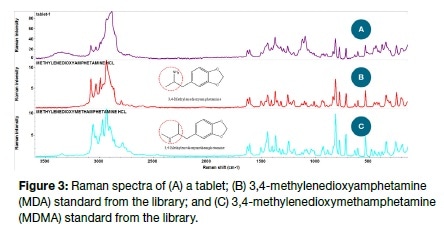
This may be seen in Figure 3 which displays the Raman spectra of a tablet, 3,4-methylenedioxyamphetamine (MDA) and also 3,4-methylenedioxymethamphetamine (MDMA) reference standards drawn from the library. As the inset shows, there is only a slight structural difference between MDA and MDMA but the two are clearly distinguished by their Raman spectra. The tablet of unknown substance was, in this case, clearly seen to be MDA with its score matching MDA at 83 compared to 66 with MDMA.
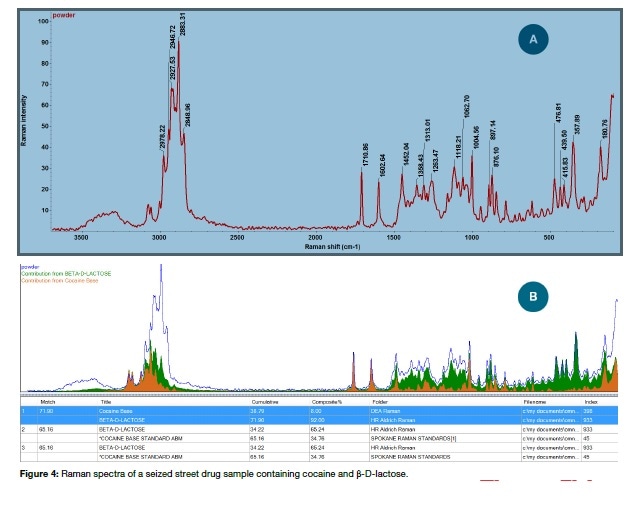
It is well known that street drugs are often mixtures and not pure substances. Figure 4 shows the results of analysis of a sample of some off-white powder in an evidence bag. A preliminary library search matched both lactose and cocaine, thus revealing that the sample was mixed. The next step was the OMNIC Specta multi-component search which identified the sample as a mixture of cocaine and β-D-lactose, the latter often being used as a cutting agent.
Conclusions
The recent upsurge in the use of illicit opioids and their analogs has created great demand for forensic analysis, and the Nicolet iS50 FT-IR spectrometer with its inbuilt iS50 Raman module meets this need. This instrumentation needs little or no sample preparation as well as allowing direct measurements of samples inside glass or plastic or polymer containers. This caters to the safety of laboratory personnel by almost eliminating the possibility of accidental exposure to these very potent drugs. It allows rapid and streamlined analysis of bulk narcotic samples, as well as of a few granules, because of the small laser spot size of 60 microns, allowing high quality Raman spectra to be acquired in both cases. The use of the DEA and LEnS Raman libraries with the OMNIC Spectra allows mixtures of illicit drugs to be confidently identified as well. Thus the use of the iS50 Raman module to carry out Raman spectroscopy offers a rapid, safe and reliable addition to the mode of illicit drug screening, detection and identification.
References and Further Reading
- https://www.cdc.gov
- Edward Sisco, Jennifer Verkouteren, Jessica Staymates, Jeffrey Lawrence, Forensic Chemistry,
- Volume 4, June 2017, Pages 108-115.
- Ciro Augusto Fernandes de Oliveira Penido, Marcos Tadeu Tavares Pacheco, Igor K. Lednev, Landulfo
- Silveira Jr., Journal of Raman Spectoscopy, 47 (1), January 2016, Pages 28–38.
About Thermo Fisher Scientific – Materials & Structural Analysis
 Thermo Fisher Materials and Structural Analysis products give you outstanding capabilities in materials science research and development. Driving innovation and productivity, their portfolio of scientific instruments enable the design, characterization and lab-to-production scale of materials used throughout industry.
Thermo Fisher Materials and Structural Analysis products give you outstanding capabilities in materials science research and development. Driving innovation and productivity, their portfolio of scientific instruments enable the design, characterization and lab-to-production scale of materials used throughout industry.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.