Sponsored Content by HORIBAReviewed by Olivia FrostMar 26 2026
Ensuring pharmaceutical product quality, safety, and efficacy is critical to therapeutic development and production.
Regulatory standards impose strict characterization of both active pharmaceutical ingredients (APIs) and excipients, from raw materials to final dosage forms.
Raman microscopy has answered these demands, becoming a mainstay as a reliable, non-destructive analytical method that offers high spatial and spectral resolution for in-depth pharmaceutical analysis.
Application
Conducted in partnership with Zentiva, this collaborative study demonstrates Raman spectroscopy capabilities for pharmaceutical analysis, with a focus on a high-throughput macro-approach for analyzing powder blend mixtures to quantify sugar-based excipients, specifically starch and lactose.
Powder blends analysis
The study was performed using the HORIBA LabRAM Soleil™ Raman microscope, which delivers high throughput without compromising resolution.
This performance is achieved thanks to its unique optical design, which uses dielectric mirrors with minimal signal loss and high-quality gratings – a core area of expertise for HORIBA.
The analysis was conducted by measuring multiple macro-approach spectra of pharmaceutical powder blends at 785 nm excitation and grating of 600 grooves/mm, covering a spectral range of 150-1800 cm-1.

Figure 1. LabRAM Soleil™ Raman Microscope. Image Credit: HORIBA
Results
Sample measurements were carried out using the patented QScan™ module, which is engineered for high-throughput sampling and provides an advanced strategy for rapid, dependable analysis.
Raman measurements were collected over a large surface area (∼3 mm2) of each sample to ensure representative data acquisition. QScan™’s macro-scale scanning method enabled analysis of the entire area in just one minute, producing a single spectrum per powder blend.

Figure 2. QScan dynamic laser scanning of the sample. Image Credit: HORIBA
A Partial Least Squares (PLS) regression model was subsequently developed to accurately predict lactose and starch concentrations, showcasing the QScan™ module’s ability to quantify powder mixtures both efficiently and precisely.
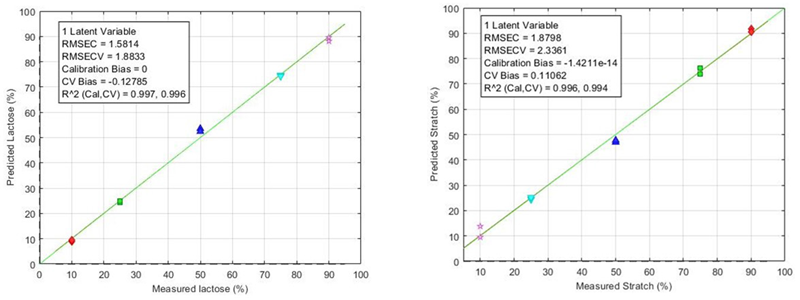
Figure 3. PLS models for Lactose and Starch. Image Credit: HORIBA
Summary
The QScan™ module successfully produced quantitative analysis of the powder blends, enabling representative sampling through a macro-spot for accurate data collection. A PLS model was developed to quantify excipients’ concentrations with an accuracy of 2 %.
The integration of cutting-edge Raman methods, efficient sampling techniques, and chemometric tools provided an effective strategy for the quantitative analysis of pharmaceutical formulations.
This approach offers valuable insights for process control and quality assurance in pharmaceutical production.
About HORIBA

Founded in 1953, HORIBA has explored a wide range of unique measurement and analysis technologies to meet global customer needs from 47 group companies and local sites spread across 29 countries and regions. Under the corporate motto Joy and Fun, the company has expanded and refined its core technologies to solve society's energy issues of today and tomorrow. Our unique measurement and analysis technologies are valued in various fields of society, including the three megatrend business fields of Energy & Environment, Biology & Healthcare, and Materials & Semiconductors. For more Information on HORIBA, visit https://www.horiba.com/int/company/about-horiba/home/
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.