Sponsored Content by HORIBAReviewed by Olivia FrostApr 7 2026
Pellets in enteric capsules typically consist of multi-layered structures with controlled interlayer composition and thickness to achieve delayed release. This study employed Micro-Raman spectroscopy, a reliable instrument for identifying components and analyzing layer thickness.
The coating differences between a reference-listed drug (RLD) and its generic counterpart were compared using Micro-Raman spectroscopy. This approach facilitated a preliminary assessment of the generic pellets and provided solutions for identifying variations in component and layer thickness during coating consistency analysis.

Image Credit: RABEYA AKTAR12/Shutterstock.com
Altering pellet composition
Pellets are a multi-unit dosage form, appearing as smooth-surfaced spherical or semispherical particles with typical dimensions of 500-1500 μm. They can be filled into capsules, compressed into tablets, or used directly as sustained-release, controlled-release, or delayed-release granules, providing substantial pharmacological benefits. Pellets can be designed with a multi-layered structure, as seen in Figure 1.
By altering the interlayer composition, pellets can be customized according to the specific microenvironments of different segments of the gastrointestinal (GI) tract, thereby delivering the therapeutic to a target organ for action.
For instance, enteric-coated capsules exploit the physiological pH variations of the GI tract. They are engineered to remain intact in the stomach and initiate drug release upon reaching the small intestine, with the release process concluding in the colon.
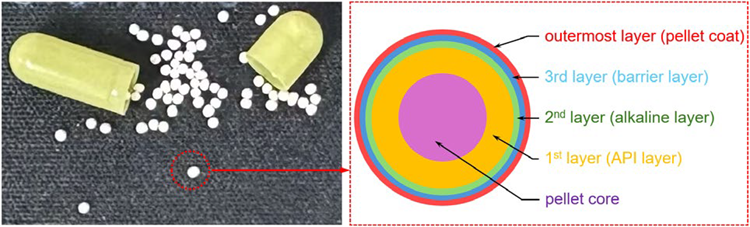
Figure 1. Cross-sectional schematic diagram of the multi-layer structure of the pellets. Image Credit: HORIBA
The overall performance of enteric pellet-based delayed-release formulations generally depends on several factors, including the physicochemical characteristics of the different coating layers, coating thickness, and production technology.1
When coating composition and process parameters remain consistent, coating layer thickness has a significant impact on drug release kinetics.1,3
Traditional methods, such as Scanning Electron Microscopy (SEM), are limited to thickness characterization, whereas Micro-Raman imaging can determine interlayer thickness while also identifying the compositional makeup of each individual layer.
This study used Raman imaging to examine coating differences between pellets from the RLD and its generic formulation, providing a reference for evaluating coating uniformity.
Materials and methods
Samples: The reference and generic capsules containing enteric-coated pellets were sourced from a popular domestic pharmaceutical company.
Methodology: Raman imaging was employed to map the component distribution across the full cross-section of the pellet. Layer thickness was then measured according to the obtained compositional maps.
Instrumentation: HORIBA XploRA™ PLUS Raman system provides high sensitivity for fast imaging and features a confocal pinhole design that guarantees high spatial resolution.
Data Analysis: Multivariate analysis (CLS fitting) in LabSpec 6 software enabled rapid component imaging, while the integrated Layers app enabled quick quantification of coating layer thickness.

Figure 2. HORIBA XploRA™ PLUS Raman system with LabSpec 6 software and Layers app. Image Credit: HORIBA
Results and discussion
Compositional analysis
The pellets from the reference and generic capsule exhibit similar coating compositions in this study. Figure 3 presents each layer’s spectra, following the name defined in Figure 1.
Two types of spectra were observed in the pellet core layer, identified as sucrose and starch, respectively.
The spectrum of the first layer consists of API and excipients and is designated as the API layer. The second layer primarily contains talc along with a small amount of API and excipients, and is designated as the alkaline layer. The third layer comprises a mixture of polymers and talc and is designated as the barrier layer. The outermost layer is the TiO2 composite layer.
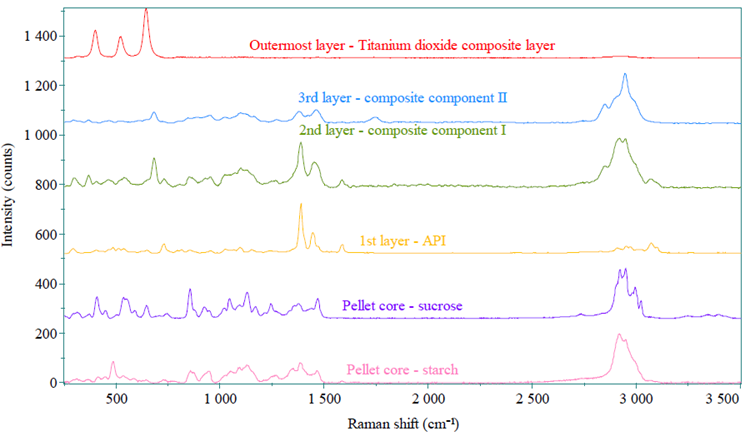
Figure 3. Raman spectra of each different pellet layer. Image Credit: HORIBA
Raman imaging analysis
The Raman imaging results for both the reference and generic pellets are shown in Figure 4. The distinct colors of each layer correspond to those of the Raman spectra in Figure 3, clearly illustrating the stratified structure of the pellets.
In line with the diagram in Figure 1, the Raman images reveal five concentric layers from the inside out: the pellet core (sucrose, starch), the first layer (API layer), the second layer (alkaline layer), the third layer (barrier layer), and the outermost coating (TiO2 composite layer), respectively.
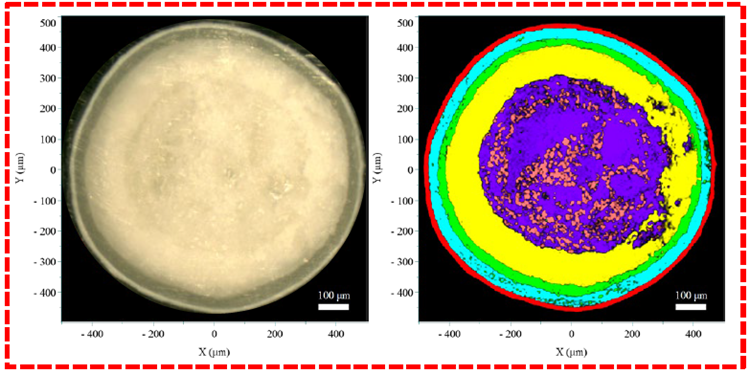
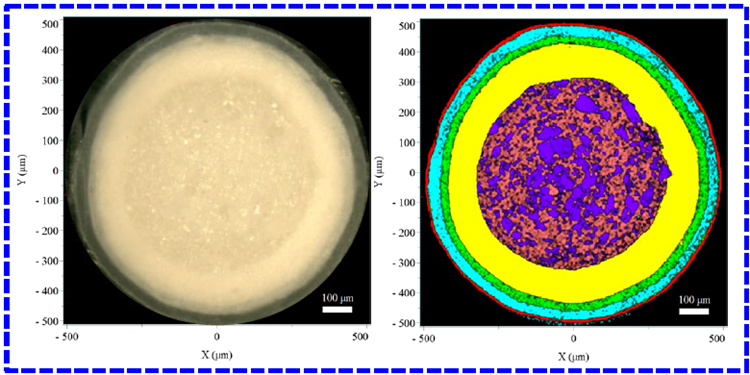
Figure 4. Results of the reference (outlined in red) and generic (outlined in blue) pellets. The left panel shows the video image, and the right panel shows the Raman image. Image Credit: HORIBA
Layer thickness analysis
The Layers app in LabSpec 6 was used to evaluate each layer’s thickness along the transverse, longitudinal, and oblique orientations. The average thickness of each layer in the three directions is summarized in Table 1.
Comparison of the layer thickness between the generic and reference pellet indicates that the most substantial variation occurs in the outermost layer, where the reference pellet is thicker than the generic one. Minor variations were detected in the thicknesses of other internal layers.
Table 1. Results of the layer thickness. Source: HORIBA
| Layer |
Main constituents |
Average (μm) |
| Reference |
Generic |
| 1st layer |
API |
~105.2 |
~109.4 |
| 2nd layer |
Alkaline |
~25.9 |
~26.7 |
| 3rd layer |
barrier |
~36.7 |
~32.6 |
| Outermost layer |
TiO2 composite |
~17.5 |
~9.2 |
Conclusion
Micro-Raman imaging technology can effectively evaluate discrepancies in coating composition and thickness between reference and generic enteric-coated capsule pellets. This technique offers important analytical insight and serves as a useful reference for assessing coating uniformity in such pharmaceutical formulations.
Acknowledgments
Produced from materials originally authored by Zhi-Wei LI, En-Ping HU, Jing SHEN
Analytical Solution Plaza, HORIBA (China) Trading Co., Ltd.
References and further reading:
- Palugan L., et al. (2015). Coated pellets for oral colon delivery. Journal of Drug Delivery Science and Technology, 25, pp.1–15. DOI: 10.1016/j.jddst.2014.12.003. https://www.sciencedirect.com/science/article/abs/pii/S1773224714000069.
- Wise, D.L. (2000). Handbook of Pharmaceutical Controlled Release Technology. CRC Press. DOI: 10.1201/9781482289985. https://www.taylorfrancis.com/books/mono/10.1201/9781482289985/handbook-pharmaceutical-controlled-release-technology-donald-wise.
- Grassi, M., et al. (2004). Modeling of drug release from partially coated matrices made of a high viscosity HPMC. International journal of pharmaceutics, 276(1-2), pp.107–114. DOI: 10.1016/j.ijpharm.2004.02.016. https://www.sciencedirect.com/science/article/abs/pii/S0378517304001280.
About HORIBA

Founded in 1953, HORIBA has explored a wide range of unique measurement and analysis technologies to meet global customer needs from 47 group companies and local sites spread across 29 countries and regions. Under the corporate motto Joy and Fun, the company has expanded and refined its core technologies to solve society's energy issues of today and tomorrow. Our unique measurement and analysis technologies are valued in various fields of society, including the three megatrend business fields of Energy & Environment, Biology & Healthcare, and Materials & Semiconductors. For more Information on HORIBA, visit https://www.horiba.com/int/company/about-horiba/home/
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.