Several methods are used for microbiological sampling in pharmaceutical manufacturing facilities.
- Conducting active aerosol (air) environmental sampling
- Passive aerosol (air) environmental sampling
- Surface sampling
Each of these procedures yields a result based on the formation of microbial colonies on the surface of a collection medium (usually agar), but the specific instruments and techniques differ between them.
Because the results of any individual aspect of sampling do not constitute evidence of asepsis, all elements of sampling should be employed together to support sterility assurance, as specified in the current edition of EU GMP Annex 1: 2022.1
9.1 The site’s environmental and process monitoring program forms part of the overall CCS and is used to monitor the controls designed to minimize the risk of microbial and particle contamination. It should be noted that the reliability of each of the elements of the monitoring system (viable, nonviable, and APS) when taken in isolation is limited and should not be considered individually to be an indicator of asepsis. When considered together, the results help confirm the reliability of the design, validation, and operation of the system that they are monitoring.
EU GMP Annex 1: 2022
More specific rules, as well as surface and passive monitoring equipment (settle plates), are covered in the regulation document.2 For all sampling procedures, it is critical to ensure that the sample appropriately reflects the environmental microbiological conditions.
This is often assessed and evaluated in terms of collection efficiency. The primary focus of this study is active air sampling, but there are sampling features for each method of sampling that assure optimal collection efficiency:
- For surfaces, optimal surface collection efficiency depends on the methodology used (swab or contact plate). If a swab is used, the efficiency is governed by the motion performed on small or large surface regions. When a contact plate is used for surface collection, a combination of pressure and contact/residence time affects collection efficiency. Each of these elements requires site validation because variances in media choice provide varied results, as do local environmental limits such as temperature and humidity.
- For passive monitoring, local environmental factors play a crucial role in collection performance. Exposure to airflows can promote desiccation of media surfaces, diminishing efficiency. Since airflow velocities vary within confined spaces, the plate's position will also have an impact on the results.
Active air sampling
Collection efficiency for active air samplers is divided into two components: physical and biological collection efficiency. This article will evaluate the factors that are likely to influence the collection efficiency of these devices.
The primary technique driving collection efficiency is microbial impaction on the collection media; however, additional functions must also be considered.
- Impaction: The inlet velocity and impaction velocity demonstrate direct correlation, enhancing the device's collecting efficiency. In some cases, an excess of sample air that cannot be evacuated from the impaction plane causes buffeting at the impaction surface, which affects collection dynamics.
- Isokinetic sampling: Isokinetic sampling involves the balancing of the sampled inlet to match the entering airflow velocity, resulting in smooth airflow to the collection device.
- Flow rate: The flow rate via the collection device or instrument can vary from 1–2 LPM to ∼200 LPM, but is typically in the range of 25–100 LPM. Different flow rates provide distinct advantages in specific contexts and influence the pace of a sample duration.
- Desiccation of media: Accelerated desiccation of localized airflow can be achieved by increasing the instrument flow rate, similar to settle plates.
- False positives identification: Identifying false positives is important for determining whether a microbiological contaminant is caused by improper handling or a microorganism from the sampling environment, which impacts efficiency.
Impaction
The BioCapt® Adjustable Height impactor is designed to maximize physical efficiency and can be adjusted to a variety of heights, allowing for the use of diverse media and pour depths without compromising the design's excellent biological efficiency.
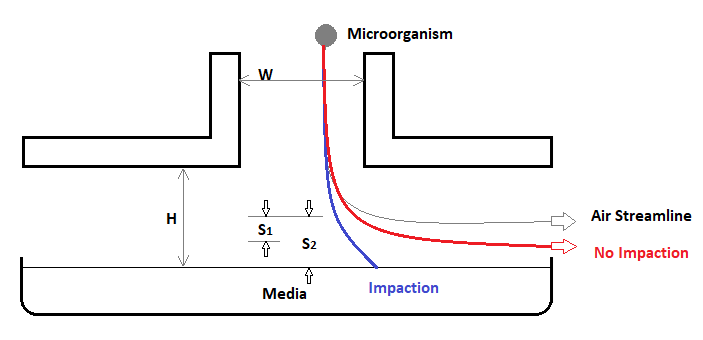
Figure 1. Diagram of Biocapt Adjustable Height Impactor. Image Credit: Particle Measuring Systems
W = width of impactor opening
H = height above media
S1 = Trajectory - No impaction
S2 = Trajectory - Impaction
When the impaction trajectory (S2) is large in comparison to the nozzle width (W), the microorganism will affect the collection media. The Stokes number (Stk) quantifies the relationship between the particle's trajectory and the width of the slit (hole), where the trajectory is determined by the particle's relaxation time and the fluid velocity.
The Stokes number is a dimensionless number that characterizes particles moving in a fluid (such as air): in this application, the Stk number captures the effect of inertial impaction on the media.
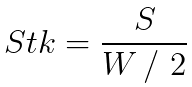
A particle with a low Stokes number follows the air streamline, whereas a particle with a high Stokes number continues on its original course and impacts the media (inertial impaction).
The number Stk50 represents a 50% physical collection efficiency and is given when 50% of particles are collected, while the other 50% remain entrained in the airflow and pass through without being collected.
Furthermore, because the slit is wider than the particle, the particle can enter the aperture from multiple points along the plane of entrance; each entry point affects the particle's trajectory. The height (H) between the slit opening and collecting media influences collection efficiency and is proportional to slit width (W).
The Reynolds number is another dimensionless variable that depicts airflow as a ratio of inertial and viscous forces in the sampler's air and distinguishes between turbulent and unidirectional flow inside the impaction zone.
The Reynolds number is mostly determined by the flow rate employed and the sampler's physical dimensions. Because slit width (W) and airflow are constant, they can be normalized for efficiency calculations.
The height (H) is a variable that depends on the media used, and differences in plate dimensions and pour depths result in variation in this parameter, affecting impaction performance (Stk50).
In the BioCapt® Adjustable Height Microbial Air Sampler Impactor and MiniCapt® Mobile Microbial Sampler, impaction occurs when the agar is 0.5–1.5 mm from the bottom surface of the impactor top plate, with an ideal height of one mm (+/- 20 %).
The bottom plate has a rotary adjustment: one full revolution moves the plate height by 1 mm, and each ‘click’ represents a 0.1 mm change. Setting the plate to the correct height allows for optimal collection efficiency across diverse media types.
Physical d50 efficiency results
The d50 value is the cutoff size at which 50% of particles in the airstream are predicted to impact and 50 % to pass through; this value corresponds to the Stk50 number. There are three techniques for calculating d50 efficiency based on physical efficiency test results.
- Estimate the impingement velocity and compare it to Mays data set
Source: Particle Measuring Systems
| Instrument |
Slit
length
(mm) |
Slit
width
(mm) |
No.
slits |
Total swept
area
(mm2) |
Volume
flow rate
(l/min) |
Impinging
vel. (m/s) |
| MiniCapt (25) |
20 |
0.1 |
20 |
40 |
25 |
10.42 |
| MiniCapt (50) |
20 |
0.1 |
20 |
40 |
50 |
20.83 |
| MiniCapt (100) |
20 |
0.2 |
20 |
80 |
100 |
20.83 |
- Calculate the impingement d50 directly
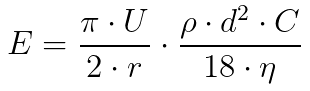
U is the air velocity through the instrument, r is the radius of the streamline’s curvature, p is the density of the sampled air, d is the equivalent particle diameter, C is the Cunningham correction factor (reliant on the particle diameter, mean free path, and several calculated and tabulated coefficients), and n is the viscosity of room air (1.81 x 10-5 Pa * s).3
- The estimation of d50 based on EN-17141, section E.5.2
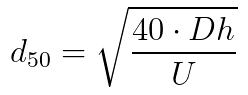
40 is the constant factor for air viscosity (°C), Dh is the equivalent hydraulic diameter of air inlet nozzle(s) in mm, and U is the impact velocity in m/s. For a circular opening, the equivalent hydraulic diameter is the hole diameter, whereas for a rectangular slit, the equivalent diameter is equal to approximately twice the width of the slit.3
The d50 physical collection minimum values applied:
Source: Particle Measuring Systems
| Instrument flowrate |
Average d50 value |
d50 using EN17141 formula |
| 25 LPM |
1 μm |
0.6 μm |
| 50 LPM |
1 μm |
0.4 μm |
| 100 LPM |
1 μm |
0.6 μm |
The MiniCapt® Mobile Microbial Air Sampler, MiniCapt® Remote and Pro Microbial Air Samplers, BioCapt® Microbial Impactor, and Biocapt® Single Use Microbial Impactor all share the same design.
The design parameters and testing were completed in accordance with ISO 14698-1:2003 and EN 17141:2020 standards. The conformity testing to this standard was carried out by CAMR (PHE Biosafety Group) of Porton Down, UK, and is documented in document n° 670/00.
Isokinetic sampling
In unidirectional situations, there is an optimal airflow rate required to serve as sheathing air. Historically, this flow rate was specified at 90 feet per minute, equivalent to roughly 0.45 meters per second.
Isokinetic Sampling must successfully sample the air without disrupting airflow patterns to obtain the most accurate representation of the particles entrained within that airflow. Isokinetic sampling operates on the following principles:
- The air entering the sampling probe nozzle has the same velocity as the surrounding undisturbed air
- Particles that pass through a specific location and time must be captured without disrupting their journey
- The nozzle is designed to disturb the air as little as possible, allowing only the same particles to enter as would have passed through without it
When sampling with an incorrect velocity into the sampling probe, anisokinetic sample conditions (superisokinetic or sub-isokinetic) influence the data from a total count and particle distribution standpoint, since more or less particles might be collected under these conditions.
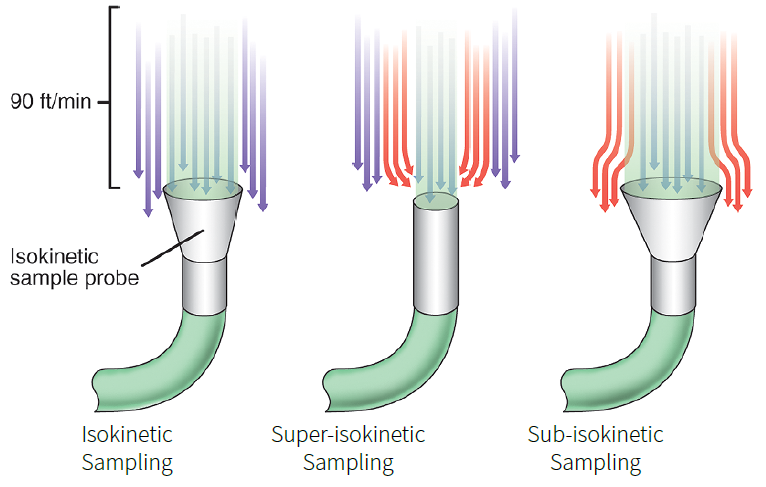
Figure 2. Isokinetic Sampling Diagram. Image Credit: Particle Measuring Systems
Sub-isokinetic sampling: occurs when the velocity of the air surrounding the sample probe exceeds the velocity of the air entering it. Heavy particles will continue to migrate in the straight down unidirectional flow line near the probe’s edges because their momentum carries them in. Lighter particles in those flow channels will form part of the sheath layer and pass through/ around the probe. This leads to oversampling of larger particles.
Super-isokinetic sampling: occurs when the velocity of the air surrounding the sample probe is lower than the velocity of the air entering it. Heavy particles at the probe’s edges will move in a straight, unidirectional flow line past the probe due to momentum. Lighter particles along these flow lines will be drawn into the probe, leading to an undersampling of bigger particles.
As shown in Figure 2, the probe has a single point of entry and is designed to efficiently dissect the air supply for sampling from the overall volume of air used in the flowing air supply. It has a sharp edge (around one to two mm wide and balanced against operator safety).
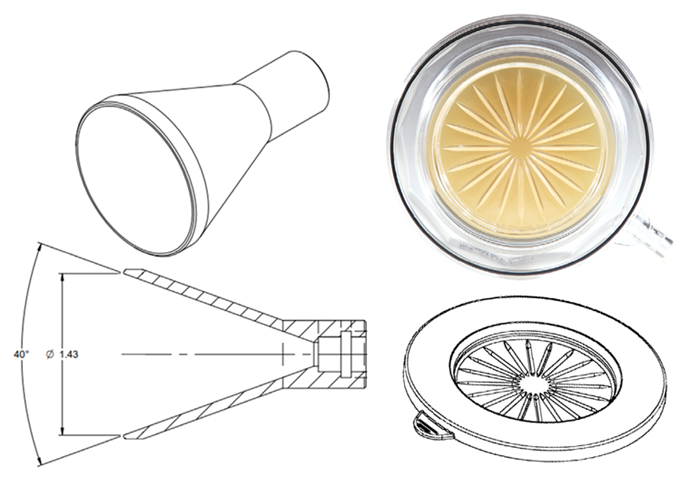
Figure 3. Typical Isokinetic Sample Probe Design (Left) and Biocapt/Minicapt Sample Head Design (Right). Image Credit: Particle Measuring Systems
There are some significant distinctions between the inlet design of an isokinetic probe and that of a microbial impactor.
Source: Particle Measuring Systems
| Parameter |
Isokinetic Probe |
BioCapt inlet |
Alternate Microbial Sampler inlet |
| Profile |
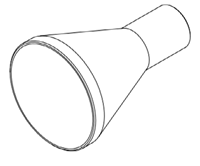
Streamlined designed narrow angle inlet, sharp differentiation to general airflow, no obstruction to sample flow
|
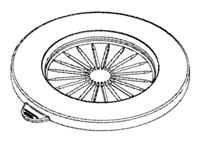
Flat top design to accommodate 90 mm agar plate media and to ensure impaction collection efficiency
|

Flat top design to accommodate 90 mm agar plate media and to ensure impaction collection efficiency
|
| Volume flow rate of the instrument |
28.3 LPM (1 CFM) 50 & 100 LPM available Probe inlet diameter a function of instrument flowrate. |
25 / 50 / 100 LPM Design of probe diameter fixed and based on the accommodation of 90 mm media plates. |
100 LPM Design of probe diameter fixed and based on the accommodation of 90 mm media plates. |
Approximate
inlet opening |
28.3 LPM – 36 mm
50 LPM – 48 mm
100 LPM – 68 mm |
Fixed opening – 73 mm |
Fixed opening – 70 mm |
| Total Sample area |
Volumetric as all flowing air passes through the whole area of the probe, 1 CFM ≈ 1000 mm2 |
The BioCapt uses 20 slits, each 20 mm long and 100 μm wide.
Total swept area = 35 mm2 |
The sampler uses 300 openings, each 6 mm in diameter.
Total swept area = 85 mm2 |
The comparison shows that microbial samplers are intended to optimize impaction and accommodate compatible media plates. The vast flat surfaces created by the housing, as well as the spaces between the impactor inlets, will serve as impaction planes for particles that are not pulled through the impactor and onto the media surface.
The wider collection plane relative to the flow rate promotes the collection of bigger particles, which are commonly associated with free-floating microorganisms in the air. The collection plane is located within the sampler's lip, preventing extraneous sample exhaust.
The only way to perform isokinetic sampling with a standard microbiological sampler is to use an external isokinetic sample probe designed for the instrument’s flow rate and accompanying air velocity; however, this increases losses due to the movement of large particles through the transit tubes. The direction of EU Annex 1 is:
"9.22 Where aseptic operations are performed, microbial monitoring should be frequent using a combination of methods such as settle plates, volumetric air sampling, glove, gown, and surface sampling (e.g., swabs and contact plates). The method of sampling used should be justified within the CCS and should be demonstrated not to have a detrimental impact on grade A and B airflow patterns."
Installing a MiniCapt® Mobile instrument in unidirectional airflow has no effect on the airflow pattern within the chamber; as the exhaust is external to the process, there is no additional concern about particle loading of the expelled air post-sample.

Figure 4. Visualization of Airflow With Minicapt Mobile Instrument. Image Credit: Particle Measuring Systems
Desiccation of media
Desiccation of localized airflow can be accelerated with increasing instrument flow rate, as with settle plates, when the media is exposed to active sampling: the higher the flow rate, the faster the desiccation rate.
The BioCapt Single Use impactors have been shown to efficiently collect data from an environment for up to 4 hours (the maximum exposure time is controlled by site-specific environmental factors, such as temperature and humidity at the sampling point).
Traditional, high-flow-rate instruments require only a 10-minute sample to collect a cubic meter (1 m3); any longer will result in increased desiccation and reduced growth promotion of test isolates.
Using the BioCapt Single Use units for continuous sampling follows EU GMP Annex 1 requirements. The Growth Promotion Test, carried out (based on European Pharmacopeia recommendations) after incubation, shows a good recovery rate (≥ 70 %) and a good condition of the media’s appearance.
False positive identification
The BioCapt impactor's unique radial pattern enables the identification of microbes that reside within the impaction zone's geometry and are thought to have entered via the intake.
The overall swept area of the impactor is only 35 mm2, compared to a total area of media on the plate of 6300 mm2, equivalent to 0.5 %. The use of Biocapt® Single Use devices reduces the risk of secondary contamination from plate manipulation, as the agar is well protected by the device.
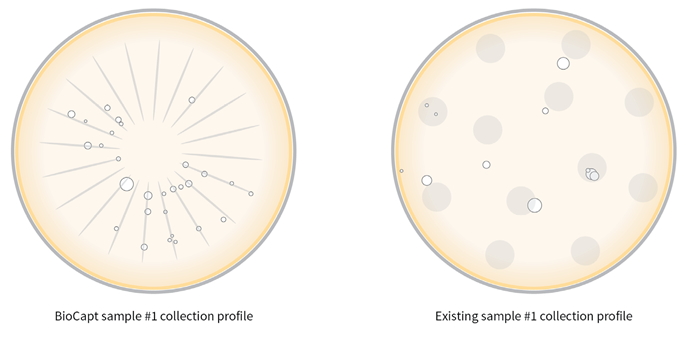
Figure 5. Collection Profiles of Biocapt Plate and Generic Plate. Image Credit: Particle Measuring Systems
Summary
Environmental monitoring to assess sterility assurance is a multistep process, and relying on a single factor is considered inconclusive.
Microbial sampling is a function of environmental monitoring, and in critical, high-risk, Grade A environments, being able to accurately differentiate a zero count (0) from a single count (1) requires empirical quantitative data due to the fact that sterility cannot be determined because zero is a virtual concept; all of the air in all areas cannot be tested.
No single data point can reliably determine pollution levels, leading EU Annex 1 to require continuous microbiological sampling in Grade A regions.
To extend the sample durations, settle plates can be used, although they are qualitative (or, at best, semi-quantitative) and not the best technique for separating zeros and ones.
The BioCapt Single Use product has been proven and validated for extended periods of up to 4 hours (the maximum exposure time is dictated by site-specific environmental factors, such as temperature and humidity at the point of sampling).
To guarantee collection efficiency, the BioCapt and MiniCapt products were validated in accordance with ISO 14698:1, ISO 14698:2, and ISO/EN 17141.
Collection and impaction efficiency are the most important criteria influencing sample accuracy, and the BioCapt has the highest level of collection efficiency, both physical and biological, even over long sample durations, without imposing undue stress on the media or isolates.
The sample is collected within the process zone of a filling line, close to the important process functions identified in site-based Environmental Monitoring Risk Assessments, without disrupting airflow patterns, and exhausted externally in accordance with Annex 1 and ISO 14698.
References
- European Commission (2022). The Rules Governing Medicinal Products in the European Union. Vol. 4 Good Manufacturing Practice Medicinal Products for Human and Veterinary Use – Annex 1 Manufacture of Sterile Medicinal Products. (online) European Commission. Available at: https://health.ec.europa.eu/medicinal-products/eudralex/eudralex-volume-4_en.
- Particle Measuring Systems. Environmental Monitoring Handbook for Pharmaceutical Manufacturer's, Rev A (2023). [Online]. Available: https://www.pmeasuring.com/application_note/environmental-monitoring-book-forpharmaceutical-manufacturers/
- EC, "EN 17141: Cleanrooms and associated controlled environments," Brussels, 2020.
Other references used
- European Medicines Agency.(2023). ICH guideline Q9 on Quality Risk Management (ICH Q9). Available at: https://www.ema.europa.eu/en/documents/scientific-guideline/international-conference-harmonisation-technical-requirements-registration-pharmaceuticals-human-use-ich-guideline-q9-r1-quality-risk-management-step-5-revision-1_en.pdf.
- FDA.Guidance for Industry Sterile Drug Products Produced by Aseptic Processing - Current Good Manufacturing Practice Pharmaceutical CGMPs. (2004). Available at: https://www.gmp-compliance.org/files/guidemgr/Asept_Process_5882fnl.pdf.
- ISO, "ISO 14698-1:2003 - Cleanrooms and associated controlled environments - Biocontamination control," 2003.
- PDA. PDA Technical Report No. 13 Revised 2022 (TR 13) Fundamentals of an Environmental Monitoring Program (single user digital version). (online) Available at: https://www.pda.org/bookstore/product-detail/6644-tr-13-revised-2022-fund-environmental-monitoring.
- Hallworth, Mark, "An Analysis of Acceptable Particle Losses in Tubing," AN 81, Boulder CO, USA, 2012.
- Sandle, Tim, Environmental Monitoring: A Comprehensive Handbook, Volume 8, Bethesda MD, USA: PDA/DHI, 2017
- PDA. (2024). Points to Consider for the Aseptic Processing of Sterile Pharmaceutical Products in Isolators. (online) Available at: https://www.pda.org/bookstore/product-detail/5699-points-to-consider-isolators.
Acknowledgments
Produced using materials originally authored by Giulia Artalli and Mark Hallworth, Product Line Manager Microbiology and Sterility and Senior GMP Scientist, Life Sciences at Particle Measuring Systems.
About Particle Measuring Systems
Particle Measuring Systems has 35 years of experience designing, manufacturing, and servicing microcontamination monitoring instrumentation and software used for detecting particles in air, liquid, and gas streams, as well as molecular contamination monitoring.
Specific applications include cleanroom monitoring, parenteral sampling, filter and in-line testing in deionized water and process chemicals, and point-of-use monitoring of inert gases and in-situ particle monitoring. Specialty monitoring includes parts cleanliness testing with a highly automated solution.
Particle measuring systems
Whether you want to protect a product or meet industry requirements, such as ISO 14644, USP 797, or GMP, Particle Measuring Systems offers a wide range of particle counters and molecular monitors to meet your needs. With 35 years of experience, it has proven reliability to support your application.
Particle counters
Protect your product with PMS' reliable particle counters. PMS has airborne, portable, and liquid particle counters for a wide variety of applications, including DI water, chemicals, and cleanroom monitoring. Compare particle counters or learn how to monitor your cleanroom or product by reading our papers.
Molecular contamination monitors
Molecular contamination causes costly problems for high-value products, production processes, and equipment surfaces. PMS has solutions for both Airborne Molecular contamination (AMC) and Surface Molecular contamination (SMC). With parts-per-trillion limits of detection, real-time sampling, NIST traceable calibrations, and various data analysis packages, you can monitor in confidence.
Gas detectors
If you need gas detectors for process control or continuous emissions monitoring, Particle Monitoring Systems can help. Get real-time, reliable results with its ammonia, hydrogen fluoride, and chlorine detectors for worker protection, CEMs, and pollution monitoring.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.