Biotech and pharmaceutical products must be manufactured in compliance with the current Good Manufacturing Practice (cGMP) regulations. Monitoring these areas meets the requirement for environmental contamination control - crucial for defining clean manufacturing and demonstrating that the necessary controls are working.
Pharmaceutical companies must have quality departments, which are responsible for routine quality assurance that establishes documented evidence to provide a high degree of assurance that a specific process will consistently produce a product meeting its predetermined specifications and quality attributes.1
To satisfy these requirements, products are manufactured in controlled environments/ cleanrooms. Cleanrooms are used to reduce variability in a production environment: as controlled environments, they can be tested to demonstrate compliance with specific standards.
GMP regulations require these environments to be meticulously tested and monitored to guarantee that there is a full and consistent awareness of current environmental conditions for both viable and non-viable contamination.
Cleanrooms are the fundamental starting points for contamination control.
Cleanroom: A room in which air filtration, air distribution, utilities, materials of construction, and equipment are maintained in a controlled manner.2
Operational procedures are defined and managed so that airborne particle concentrations meet the classifications for particulate cleanliness. The International Standard ISO 14644-1 defines cleanroom contamination levels.3
Pharmaceutical cleanrooms are categorized according to the required air particle concentrations that meet the cleanliness criteria for the manufacturing process.
Determining cleanroom class is based on statistically valid measurements and is a function of the room’s filtration and operational status. Essentially, the room is calibrated to ensure it meets its intended classification. It is not primarily a function of the risk of application.
There are three phases for measurement that involve particle counting in cleanrooms:
- Operational: all equipment is installed and functions according to a specified format, and a specified, agreed number of personnel are present, all working to an accepted procedure.
- As Built: a completed, functional room with all services connected but with no personnel or production equipment within the facility.
- At Rest: all services are connected, all equipment has been installed and operates as agreed, but no personnel are present.
Particles are counted at defined grid locations within the cleanroom to perform the airborne particle count test. The test points should be at work height and evenly spaced throughout the work area to evaluate air cleanliness.
Pharmaceutical cleanrooms generally operate at Class 5 (most aseptic areas), Class 7 (surrounding areas), or Class 8 (support areas).
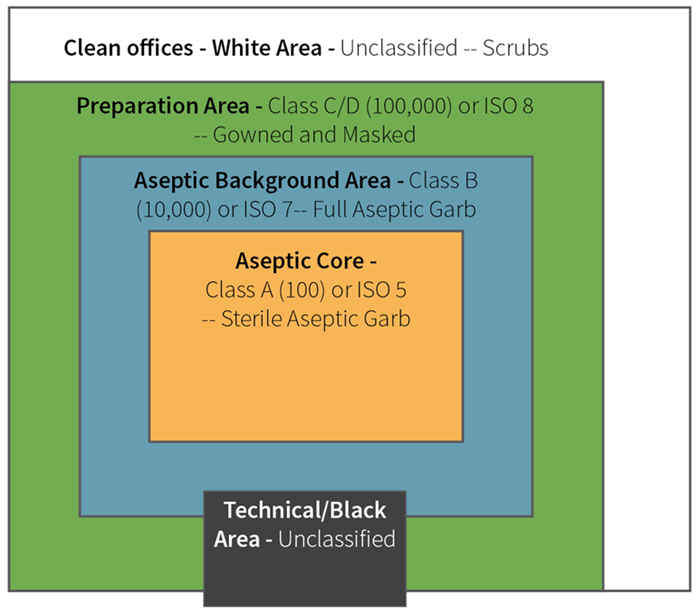
Figure 1. Diagram of Typical ISO Rating in Areas of Cleanroom. Image Credit: Particle Measuring Systems
Pharmaceutical cleanroom utilization
Once a cleanroom has been tested and deemed compliant with cleanroom classification (usually by using a Light Scattering Aerosol Particle Counter (LSAPC)), the achieved classification governs the production activities that can take place in that cleanroom or clean air device. The FDA defines two areas.
- Critical areas
- Supporting clean areas
Critical area: An area containing products that are vulnerable to contamination if exposed is considered critical. It is crucial that the environment in which aseptic operations are performed is controlled and maintained at an appropriate level of quality to maintain product assurance.
The air particle content is one parameter of environmental quality. Particles are important because they can enter a product as extraneous contaminants and biologically contaminate it by serving as vehicles for microorganisms.
Air in the immediate proximity of exposed sterilized containers/closures and filling/closing operations would be of appropriate particle quality when it has a per-cubic-meter particle count of no more than 3,520 in a size range of 0.5 µm and larger when counted at representative locations normally not more than 1 foot away from the work site, within the airflow, and during filling/closing operations. This level of air cleanliness is also known as Class 100 (ISO Class 5).
We recommend that measurements to confirm air cleanliness in critical areas be taken at sites where there is the most potential risk to the exposed sterilized product, containers, and closures.4
Supporting clean areas: Supporting clean areas are classified by the FDA as follows:
The nature of the activities conducted in a supporting clean area determines its classification. It is recommended that the area immediately adjacent to the aseptic processing line meet, at a minimum, Class ISO 7 standards under dynamic (operational) conditions. Manufacturers can also classify this area as Class ISO 6 or maintain the entire aseptic filling room at Class ISO 5. An area classified at a Class ISO 8 air cleanliness level is appropriate for less critical activities (e.g., equipment cleaning).4
Environmental monitoring
After a cleanroom or clean-air device has been certified to meet cleanliness requirements, it must also demonstrate that this control can be sustained throughout production. The environment must be thoroughly monitored to ensure a well-developed, sustained awareness of current conditions, including the identification of periodic events that could be devastating if left unobserved.
Constant monitoring means continuous analysis and large volumes of data that can be used to detect trends.
Manufacturing facilities should therefore have thorough environmental monitoring programs that monitor non-viable and viable airborne particulates, surface viable contamination, and, in aseptic areas, personnel.
These procedures should address the locations and frequencies of the monitoring sample points, warning and alarm limits for each area, and any corrective actions to be taken if any areas deviate from expected results.
When limits are exceeded, part of the action taken should include an investigation into the problem’s source, its potential impact on the product, and any necessary measures to prevent recurrence.
Generally, areas of lower classification (ISO 7, ISO 8, or unclassified rooms) require less frequent monitoring. Though this reduced monitoring is performed at a lesser frequency in "controlled" environments (ones with some level of particulate controls), it should be of the same integrity as that sampled in higher classifications.
A portable particle counter can be employed in both critical areas and supporting clean areas. Selecting portable particle counters involves several factors, including ergonomics and suitability for the specific areas to be tested.
- Size channels required: There is a conventional requirement for two primary monitoring channels: 0.5 µm and 5.0 µm. However, these sizes can be complemented with more sizes, both smaller and larger than the given range. Expanding the sizes enables further investigation of out-of-tolerance trends, as events may generate particles within specific bands, indicative of a particular failure mode.
- A sample flow rate of 1 CFM (cubic feet per minute = 28.3 L/minute) is suitable for most cleanroom activities, including certification. This flow rate has suitable particle transport properties, allowing up to two meters of tubing to be used without a significant loss of larger particle counts. Increased flow rates are available (up to 100 LPM). Such flow rates facilitate rapid qualification testing in clean environments with low populations of 0.5 µm particles. The increased flow rate delivers a statistically significant sample volume more quickly.
- Mains- or battery-operated functionality increases the ease of portability and ergonomics of the chosen instrument. When batteries are used, they should last the duration of the required testing or support live exchanges without compromising data quality.
- Mobility, either by hand or cart: Technicians may need multiple instruments to perform environmental monitoring. If small spaces are to be tested, the instrument can be placed in the clean-air device, or the sample can be transported from within an environment to the particle counter using tubing.
- Other accessories, such as a local display for controls and alarming, or a built-in printer for sample point-generated reports, are also sometimes necessary. Electronically transferring data to an LIMS or central software may be necessary, as thermal paper has durability limitations and transposition requirements for reporting.
Portable instruments are often used throughout facilities by moving them from location to location, following each measurement. Each sample point’s data record should be made up of the following details:
Primary data
- Status of instruments (hardware alarms)
- Channel data, results, and units
- Sample date and time
- Sample location
Additional information to support the primary data includes:
- Instrument identification (serial number)
- The operator taking the sample
- Alarm threshold excursions
- Sampling parameters
- Occupancy state
This data can then be locally printed or exported to a software management application for further review.
Data review should be performed in three phases: short-term, medium-term, and long-term trending.
- Short-term trends look at sample-to-sample relationships and process stability. It also enables ‘n’ out of ‘m’ nuisance controls, in which samples might reflect the process and not first air.
- Medium-term trends facilitate the review of day-to-day or batch-to-batch performance. At this scale, trend data reviews show whether a particular occupancy or process impacts particle burden during production.
- Long-term trends are every three, six, or 12 months and may identify a progressive decline in the efficacy of installed controls.
These reporting and trending requirements feed into a site’s Contamination Control Strategy (CCS) and should be performed and documented regularly.
References
- Federal Drug Administration. General Principles of Validation, (1987).
- Federal Standard 209E Airborne Particulate Cleanliness Classes in Cleanrooms and Clean Zones. Available at: https://www.lascoservices.com/Federal_Standard_209.pdf.
- ISO (2016). Cleanrooms and associated controlled environments - Part 1: Classification of air cleanliness by particle concentration, ISO Standard No. 14644-1:2015. (online) ISO. Available at: https://www.iso.org/standard/53394.html.
- U.S. Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research (CDER,) Center for Biologics Evaluation and Research (CBER), Office of Regulatory Affairs (ORA). Guidance for Industry Sterile Drug Products Produced by Aseptic Processing - Current Good Manufacturing Practice, (2004).
Acknowledgments
Produced using materials originally written by Mark Hallworth from Particle Measuring Systems.
About Particle Measuring Systems
Particle Measuring Systems has 35 years of experience designing, manufacturing, and servicing microcontamination monitoring instrumentation and software used for detecting particles in air, liquid, and gas streams, as well as molecular contamination monitoring.
Specific applications include cleanroom monitoring, parenteral sampling, filter and in-line testing in deionized water and process chemicals, and point-of-use monitoring of inert gases and in-situ particle monitoring. Specialty monitoring includes parts cleanliness testing with a highly automated solution.
Particle measuring systems
Whether you want to protect a product or meet industry requirements, such as ISO 14644, USP 797, or GMP, Particle Measuring Systems has a large variety of particle counters and molecular monitors to meet your needs. With 35 years of experience, it has proven reliability to support your application.
Particle counters
Protect your product with our reliable particle counters. Particle Measuring Systems has airborne, portable, and liquid particle counters for a wide variety of applications, including DI water, chemicals, and cleanroom monitoring. Compare particle counters or learn how to monitor your cleanroom or product by reading our papers.
Molecular contamination monitors
Molecular contamination creates costly problems to high value products, production processes, and equipment surfaces. It has solutions for both Airborne Molecular contamination (AMC) and Surface Molecular contamination (SMC). With parts-per-trillion limits of detection, real-time sampling, NIST traceable calibrations, and various data analysis packages, you can monitor in confidence.
Gas detectors
If you need gas detectors for process control or continuous emissions monitoring, Particle Measuring Systems can help. Get real-time, reliable results with its ammonia, hydrogen fluoride, and chlorine detectors for worker protection, CEMs, and pollution monitoring.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.