A Contamination Control Strategy (CCS) outlines a means of identifying and analyzing risk, reviewing mitigating innovations and opportunities, and ultimately defining preventive and corrective action plans. By implementing a CCS, organizations can better address the diverse causes of compromised sterility and contamination.
Developing and delivering a robust and useful CCS requires input and support from a range of subject matter experts, including those in regulatory, manufacturing, quality assurance, quality control, and supporting departments.
Annex 1 of the 2022 EU GMP defines a CCS as:
Contamination Control Strategy (CCS) – A planned set of controls for microorganisms, pyrogens and particulates, derived from current product and process understanding that assures process performance and product quality. The controls can include parameters and attributes related to active substance, excipient and drug product materials and components, facility and equipment operating conditions, inprocess controls, finished product specifications, and the associated methods and frequency of monitoring and control.
Annex 1, 2022
A comprehensive Contamination Control Strategy should address environmental controls and optimize detection.
This requires manufacturers to implement Quality Risk Management (QRM) and Quality by Design into their production facility, personnel flows, equipment, and supporting processes to ensure reliability and inform the development of the CCS.
Historical data and trend analysis should also be reviewed and performed to appropriately measure and identify gaps, risks, and opportunities for risk mitigation.
These are all key factors for making a successful CCS, with its organization, design, procedures, and technological controls all considered most effective when they complement each other holistically.
Development of a Contamination Control Strategy
Annex 1 lists the elements that should be considered for the CCS:
“2.5 The development of the CCS requires detailed technical and process knowledge. Potential sources of contamination include microbial and cellular debris (e.g., pyrogens and endotoxins) as well as particulates (e.g., glass and other visible and subvisible particles).
Elements to be considered within a CCS should include (but are not limited to):
- Design of both the plant and processes, including the associated documentation.
- Premises and equipment.
- Personnel.
- Utilities.
- Raw material controls – including in-process controls.
- Product containers and closures.
- Vendor approval – such as key component suppliers, sterilization of components and single-use systems (SUS), and critical service providers.
- Management of outsourced activities and availability/transfer of critical information between parties, e.g., contract sterilization services.
- Process risk management.
- Process validation.
- Validation of sterilization processes.
- Preventative maintenance – maintaining equipment, utilities, and premises (planned and unplanned maintenance) to a standard that will ensure there is no additional risk of contamination.
- Cleaning and disinfection.
- Monitoring systems - including an assessment of the feasibility of the introduction of scientifically sound, alternative methods that optimize the detection of environmental contamination.
- Prevention mechanisms – trend analysis, detailed investigation, root cause determination, corrective and preventive actions (CAPA), and the need for comprehensive investigational tools.
- Continuous improvement based on information derived from the above.”
Manufacturers may interpret Annex 1’s definition of a CCS in two ways: as a method to implement or as an approach to verify its effectiveness.
Both of these interpretations involve a cyclical three-part process to assess, accept, and remediate the strategy, with the conclusion of each part feeding into the next in an ongoing loop.
It is important to note that the current state of the manufacturing process influences where to begin or review CCS development. There is no single, strict starting point for all applications.
For example, a manufacturer launching a new facility may start with the Assessment phase, identifying potential risks and gaps. Retrieving the required instrumentation and documentation for a comprehensive CCS would support the review of this strategy.
If the production or filling line is already established, subject matter experts involved in the CCS development may begin at the Acceptance stage, compiling historical data and trends and capturing information to show that there is a robust CCS in place.
Three steps for developing a CCS: Acceptance, assessment, and remediation
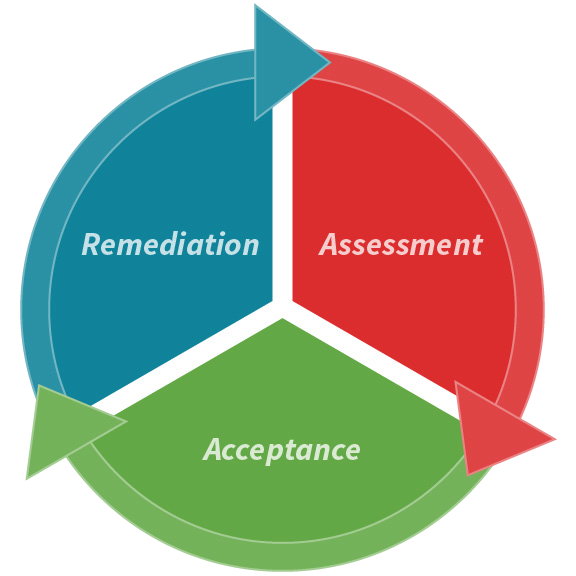
Figure 1. CCS development stages. Image Credit: Particle Measuring Systems
Assessment: Outline the CCS
CCS development must be underpinned by the need to maintain environmental control within sterile manufacturing principles.
Scientific fundamentals, regulations, production principles, codes and standards of recognized authorities, and the latest technological innovations must be reviewed and referenced to ensure that CCS development is compliant.
Applying the essential principles of QRM remains the best way to achieve a full assessment. Any changes or the introduction of new elements to the CCS should be defined and outlined, beginning with the preparation of a risk assessment.
ICH Guidance Q9 (Figure 2) suggests that subject matter experts investigate the potential of risks or any gaps in both product and patient safety. Experts should also develop methods to mitigate or eliminate high risks.
Methods require evidence-based support to verify that risk or gaps have been reduced (or eliminated) and that control of the environment has improved. Implemented risk mitigation methods may be performed once, periodically, or on an ongoing basis.
It is also essential to define and utilize Standard Operating Procedures (SOPs) with periodic reviews. This should be accompanied by system qualifications and the validation of production processes, including cleaning, decontamination, sterilization, and environmental monitoring.
Follow-up compliance and staff training are also important. Documents relevant to risk mitigation methods are identified and collected to complete the assessment stage. Historical data and trends should be analyzed to gauge the success of any corrective actions and identify any required improvements.
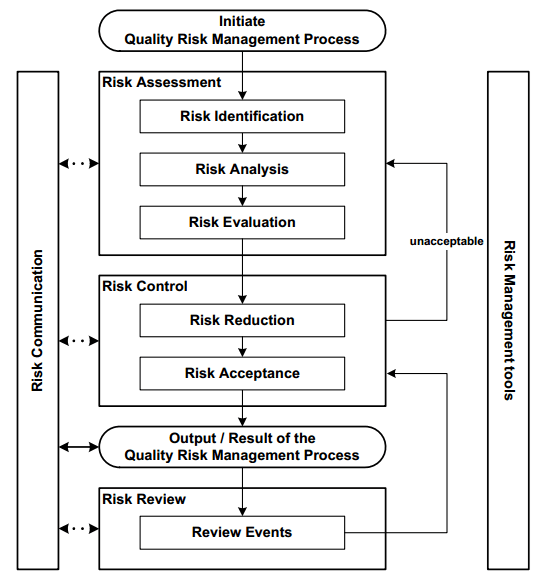
Figure 2. Overview of quality risk management (QRM) process. Image Credit: Particle Measuring Systems
Acceptance: Compilation of CCS documentation
Compiling documentation for CCS into a single document is a laborious task, but an essential one, as a lack of organized, relevant documents can present a number of challenges.
It can make it difficult to identify and properly understand contamination risks and existing risk-mitigating procedures. This will make it harder to determine the remediation process changes required.
Manufacturers may also find it difficult to track and analyze historical data, further complicating the task of identifying patterns and trends.
A lack of centrally organized documentation could lead to compliance issues with industry regulations or standards, or make it difficult to demonstrate that the company has adopted sufficient measures to protect its personnel, environment, and products.
Documentation should include comprehensive information on the risk assessments, standards, and protocols in place to prevent and mitigate contamination risks. This may include information on staff training and qualifications, equipment design and maintenance, and the facility’s cleaning and sanitation procedures.
Documentation should also detail any testing and monitoring done to detect and identify potential sources of contamination.
Remediation: CCS evaluation
The Annex 1 document, released in 2022, highlights the value of adopting a holistic approach to measure the prevention and control of contamination.
2.6 The CCS should consider all aspects of contamination control with ongoing and periodic review resulting in updates within the pharmaceutical quality system as appropriate. Changes to the systems in place should be assessed for any impact on the CCS before and after implementation.
Annex 1, 2022
Manufacturers should perform a CCS evaluation after reviewing data collected via controls. This analysis determines whether the program is sufficiently preventing contamination and whether the residual risk falls within an acceptable range, as defined by regulatory bodies such as the FDA or EU GMP.
If this residual risk is found to be outside of the acceptable range, it will be necessary to edit the CCS and apply upgrades to processes where required.
CCS Remediation’s occurrence and frequency will be contingent upon a range of variables, including the type and complexity of the facility’s equipment and processes, the level of risk associated with the facility and its operations, any history of contamination incidents, and any relevant regulatory and legal requirements.
Facilities implementing new QMS or equipment may have specific frequency parameters, with high personnel turnover, deviations, the environment, external audits, and inspections contributing to the necessary review frequency.
Technical controls
Risk Communication and Management Tools are underpinned by technical controls. Properly implemented, maintained, and joined-up equipment, systems, and procedures are required in order to prevent, detect, and minimize the spread of contaminants.
These technical controls for a CCS in sterile manufacturing may include:
- Ventilation and air filtration systems are designed to maintain the appropriate flow rate in the facility and control the spread of contaminants in the air.
- Cleanroom technology, with specialized equipment and construction techniques designed to ensure a controlled environment with minimal contaminants.
- The facility's design and layout minimize risk by separating clean and dirty areas and implementing barriers to prevent cross-contamination.
- Process controls, with dedicated procedures and equipment designed to control the spread of contaminants during manufacturing, for example, using specialized packaging and sealing methods, isolating certain processes, and implementing stringent hygiene protocols for personnel.
- Material controls with specialized procedures for logging, handling, storage, and disposal of raw materials, active ingredients, and finished products to minimize the risk of contamination.
- Monitoring and testing for contaminants in the facility via surface sampling, air sampling, and environmental monitoring to understand and minimize risk.
- Maintenance and decontamination, with specialized procedures implemented to prevent contamination and protocols in place to remove and avoid the buildup of contaminants and residue on facilities and equipment.
- Automation streamlines monitoring, recording, and control of process parameters via automated systems to help limit human error and ensure consistent performance.
Conclusion
A Contamination Control Strategy (CCS) in biomedical and pharmaceutical manufacturing settings is a set of procedures and protocols designed to prevent, control, and monitor the spread of contaminants throughout the production process. A robust, regularly reviewed CCS is key to ensuring the safety, efficacy, and purity of products.
A CCS for pharmaceutical manufacturing should include a range of measures, including the implementation of:
- Stringent hygiene and sanitation protocols for equipment, facilities, and personnel
- Access restrictions for specific areas of the facility to minimize contamination risk
- Appropriate handling, storage, and disposal procedures for supplies and hazardous byproducts
- Regular inspections and testing to detect and identify potential contamination sources
- Emergency response plans for potential contamination incidents
- Employee and contractor training on contamination control procedures
- A QMS designed to ensure compliance with standards and regulations, specifically GMP
- Regular reviews and updates to the CCS to ensure its ongoing effectiveness
- Specific procedures for handling active ingredients, raw materials, and finished products
- Strict protocols for the cleaning and maintenance of facilities and equipment
CCS in pharmaceutical manufacturing is also closely monitored by regulatory bodies like the FDA and WHO, meaning that failure to comply can lead to considerable penalties and potential product recalls.
References
- European Commission (2022) The rules governing medicinal products in the European Union, Volume 4: EU guidelines for good manufacturing practice for medicinal products for human and veterinary use. Annex 1: Manufacture of sterile medicinal products. C 5938 final, 22 August.
- International Council for Harmonisation (ICH) (2005) ICH guideline Q9 on quality risk management. Step 4: GMP-related documents.
- U.S. Food and Drug Administration (2004) Guidance for industry: Sterile drug products produced by aseptic processing – current good manufacturing practice.
Acknowledgments
Produced using materials originally authored by Ugonna Omeronyé from Particle Measuring Systems.
About Particle Measuring Systems
Particle Measuring Systems has 35 years experience designing, manufacturing, and servicing microcontamination monitoring instrumentation and software used for detecting particles in air, liquid, and gas stream as well as molecular contamination monitoring.
Specific applications include cleanroom monitoring, parenteral sampling, filter and in-line testing in deionized water and process chemicals, and point-of-use monitoring of inert gases and in-situ particle monitoring. Specialty monitoring includes parts cleanliness testing with a highly automated solution.
Particle measuring systems
Whether you want to protect product or meet industry requirements, such as ISO 14644, USP 797, or GMP, Particle Measuring Systems has a large variety of particle counters and molecular monitors to meet your needs. With 35 years of experience, it has proven reliable for application support.
Particle counters
Protect your product with its reliable particle counters. We have airborne, portable, and liquid particle counters for a wide variety of applications, including DI water, chemicals, and cleanroom monitoring. Compare particle counters or learn how to monitor your cleanroom or product by reading our papers.
Molecular contamination monitors
Molecular contamination creates costly problems for high-value products, production processes, and equipment surfaces. PMS has solutions for both Airborne Molecular contamination (AMC) and Surface Molecular contamination (SMC). With parts-per-trillion limits of detection, real-time sampling, NIST traceable calibrations, and various data analysis packages, you can monitor in confidence.
Gas detectors
If you need gas detectors for process control or continuous emissions monitoring, PMS can help. Get real-time, reliable results with its ammonia, hydrogen fluoride, and chlorine detectors for worker protection, CEMs, and pollution monitoring.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.