Pharmaceutical products are manufactured to adhere to exacting standards of efficacy and quality. All aspects of quality are evaluated, taking into account the hazards connected with the delivery technique (injected, eaten, etc.) and the manner in which they were manufactured (aseptic, terminally sterilized, or under less regulated conditions).
This article examines two aspects of that process: the quality of the environment in which the product is created, and the rules governing particle concentration limitations that define what constitutes a controlled environment. It also addresses physical testing requirements (EN ISO 14644-1:2015)1 as well as the regulatory guidance standards (EU GMP Annex 1).2
ISO 14644-1
The ISO 14644 room classification suite of standards went into effect in 1999, with ISO 14644-1 establishing the procedure for classifying a room and specifying the maximum permitted particle concentration within a specific volume of air.
Although ISO 14644 has been widely used for cleanroom classification, there are discrepancies in routine monitoring, particularly between ISO 14644 and EU and WHO GMP. The certification state of the cleanroom must be determined prior to testing; ISO 14644-1 defines three states:
- As Built: a completed room with all services connected and functional, but without production equipment or staff within the facility.
- At Rest: all the services and equipment are operational, but no personnel are present.
- Operational: all equipment is installed and working properly, and a specified number of personnel are present and follow established procedures.
Table 1 defines and presents the cleanroom concentration limits for particles larger than a certain size.
Table 1. Airborne particulate cleanliness classes for cleanroom and clean zones (as indicated in ISO 14644). Source: Particle Measuring Systems
ISO 14644-1:2015
Classification
Number (N) |
Maximum Concentration Limits (Particles/m3) |
| 0.1 μm |
0.2 μm |
0.3 μm |
0.5 μm |
1.0 μm |
5.0 μm |
| ISO Class 1 |
10 |
|
|
|
|
|
| ISO Class 2 |
100 |
24 |
10 |
|
|
|
| ISO Class 3 |
1 000 |
237 |
102 |
35 |
|
|
| ISO Class 4 |
10 000 |
2 370 |
1 020 |
352 |
83 |
|
| ISO Class 5 |
100 000 |
23 700 |
10 200 |
3 520 |
832 |
|
| ISO Class 6 |
1 000 000 |
237 000 |
102 000 |
35 200 |
8 320 |
298 |
| ISO Class 7 |
|
|
|
352 000 |
83 200 |
2 930 |
| ISO Class 8 |
|
|
|
3 520 000 |
832 000 |
29 300 |
| ISO Class 9 |
|
|
|
35 200 000 |
8 320 000 |
293 000 |
These limitations have been defined in accordance with the standard’s calculation, using the following formula for the intermediate decimal classes:
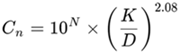
Where,
- Cn is the maximum allowed concentration (particles per cubic meter) of airborne particles equal to or larger than the considered particle size.
- N is the ISO categorization number, which cannot exceed nine or be less than one.
- D represents the particle size in micrometers that is not stated in Table E-1.
- K is a constant (0.1), expressed in micrometers.
The link between particle size and abundance within a population is thus a function of (1/D)2.08, and when plotted against concentration on a log/ log scale, the slope of the curve for each class is 2.08; this relationship is given in the table above.
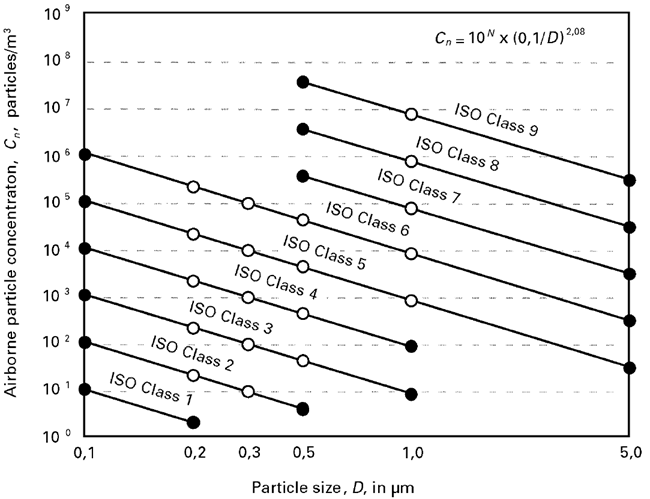
Figure 1. A graphical representation of ISO class concentrations limits for selected classes. Image Credit: Particle Measuring Systems
The designation for cleanroom or clean zone certification should additionally include the following features:
- The room categorization number, referred to as ‘ISO Class N’.
- Occupancy status
- The considered particle size. It is also feasible to certify a cleanroom in several sizes. In this scenario, the sample volume requirement for the greatest particle size is employed.
Example
As an example, unidirectional airflow device x operates in an ISO Class 5 clean zone at 0.5 µm (3520 n/ m3). The clean zone must be tested to validate the statement; the ISO 14644-1 standard outlines each of the component processes required to demonstrate compliance.
Assume we have a clean-air device intended for use in an aseptic processing environment. In the operating state, this area must meet ISO Class 5 at 0.5 µm (≡ 10,000/ ft3): how can the classification be determined?
The space is 12 meters by five meters (60 m2), with a worktable in the center.
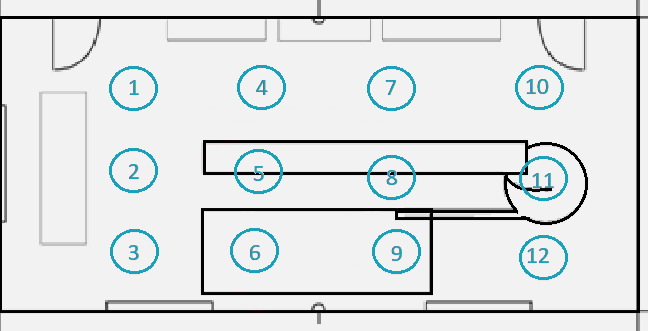
Figure 2. Diagram showing the classification locations within the clean zone. Image Credit: Particle Measuring Systems
Step 1. Calculate the maximum permitted particle concentration
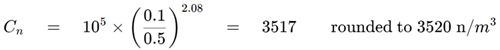
Figure 3. Maximum permitted particle concentration calculation. Image Credit: Particle Measuring Systems
Step 2. Determine the number of sample locations from the table below
Table 2. Number of sample locations required. Source: Particle Measuring Systems
| Area of Zone (m2) |
ISO 14644-1:1999 |
ISO 14644-1:2015 |
| 2 |
2 |
1 |
| 4 |
2 |
2 |
| 6 |
3 |
3 |
| 8 |
3 |
4 |
| 10 |
4 |
5 |
| 24 |
5 |
6 |
| 28 |
6 |
7 |
| 32 |
6 |
8 |
| 36 |
6 |
9 |
| 52 |
8 |
10 |
| 56 |
8 |
11 |
| 64 |
8 |
12 |
| 68 |
9 |
13 |
| 72 |
9 |
14 |
| 76 |
9 |
15 |
| 104 |
11 |
16 |
| 108 |
11 |
17 |
Step 3. Calculate the sample volume
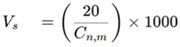

Based on ISO 14644-1:2015, this formula yields a sample volume of 5.68 L, and for a conventional particle counter with a flow rate of 28.3 L/min, a one-minute sample would exceed the minimum sample volume requirement. To meet the standard, we will take a one-minute sample at each of the twelve locations.
Step 4. Take measurements at each location (one minute per sample) and record the results
Table 3. Illustration of locations within the example clean zone and measurement results. Source: Particle Measuring Systems
| Location |
Number/m3 |
| 1 |
708 |
| 2 |
885 |
| 3 |
1522 |
| 4 |
2336 |
| 5 |
3363 |
| 6 |
2584 |
| 7 |
2301 |
| 8 |
2089 |
| 9 |
1344 |
| 10 |
2013 |
| 11 |
1756 |
| 12 |
1897 |
Step 5. Define report
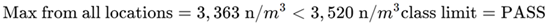
This area meets ISO Class 5 clean zone specifications at 0.5 µm, making it suitable for its intended function. Room classification must be repeated at a frequency specified by ISO 14644-2.3
EU GMP Annex 1
The European Union GMP advice for sterile manufacturing was changed in 2003 to reflect the transition from many cleanroom standards to a single, unified cleanroom standard, ISO4644-1.4 The front page notes this:
“Annex 1 of the EC Guide to Good Manufacturing Practice (GMP) provides supplementary guidance on the application of the principles and guidelines of GMP to sterile medicinal products. The guidance includes recommendations on environmental cleanliness standards for clean rooms. The guidance has been reviewed in the light of the international standard EN/ISO 14644-1 and amended in the interests of harmonisation but taking into account specific concerns unique to the production of sterile medicinal products.”
Specifically, the method by which a cleanroom was certified had to follow the principles and format of the ISO14644-1 guidance, but the ISO standard was amended with regard to sterile pharmaceutical items. To that purpose, a table of cleanroom certification values - roughly equivalent to ISO 14644-1 - was created.
For clarity, a series of comments was added alongside the table; however, the first of these created some misunderstanding. This uncertainty was resolved with the 2008 introduction of the EU GMP Annex 1, which clearly states the three processes that must be completed.5
Each cleanroom and clean air device should be classified first, followed by monitoring to ensure that conditions are maintained in relation to product quality, and the data gathered from said monitoring should be examined in light of the risk to finished product quality.
Table 4. EU GMP Annex 1:2008 room classification table. Source: Particle Measuring Systems
| EU GMP Grade |
At Rest |
In Operation |
| Maximum number of particles permitted/m3 equal to or greater than the tabulated size |
|
0.5 μm |
5.0 μm |
0.5 μm |
5.0 μm |
| A |
3520 |
20 |
3520 |
20 |
| B |
3520 |
29 |
352,000 |
2900 |
| C |
352,000 |
2,900 |
3,520,000 |
29,000 |
| D |
3,520,000 |
29,000 |
Not Defined |
Not Defined |
As mentioned above, the restrictions in the EU GMP table differ slightly from those in the ISO 14644 standard.
To perform the required certification, it is necessary to understand the workings of ISO 14644-1, how to certify a cleanroom in accordance with that standard, the rules governing the number of sample points, the location of sample points, the volume of sample to be taken at each location, and the statistical analysis of cleanroom data that must be followed.
Rather than using the table for the categorization limitations recommended in ISO 14644-1, the above table should be used, which is printed in the new guideline document.
The GMP also defines other expectations. Expectations for Grade A samples include a 1 m3 sample volume per location and a minimum sample tubing length due to the high precipitation of 5 µm particles in transport tubing. Ideally, no sample tubing should be used.
Cleanroom recertification should adhere to the ISO 14644-2 guidelines: once each year for ISO Grade 6 and above, and once every six months for ISO Grade 5. Concessions are allowed for enlarging ISO Grade 5 areas if a continuous monitoring system is in place. Certification is best performed during media fills or simulated filling runs.
References
- ISO (2016). Cleanrooms and associated controlled environments - Part 1: Classification of air cleanliness by particle concentration, ISO Standard No. 14644-1:2015. (online) ISO. Available at: https://www.iso.org/standard/53394.html.
- European Commission (2022). The Rules Governing Medicinal Products in the European Union Volume 4 EU Guidelines for Good Manufacturing Practice for Medicinal Products for Human and Veterinary Use: Annex 1, Manufacture of Sterile Medicinal Products, Annex 1. (online) Available at: https://health.ec.europa.eu/latest-updates/revision-manufacture-sterile-medicinal-products-2022-08-25_en.
- ISO (2015). Cleanrooms and associated controlled environments - Part 2: Monitoring to provide evidence of cleanroom performance related to air cleanliness by particle concentration, ISO Standard No. 14644-2:2015. (online) ISO. Available at: https://www.iso.org/standard/53393.html.
- European Commission. EC Guide to Good Manufacturing Practice Revision to Annex 1: Manufacture of Sterile Medicinal Products, Annex 1 (2003).
- European Commission, EudraLex. The Rules Governing Medicinal Products in the European Union Volume 4 EU Guidelines for Good Manufacturing Practice for Medicinal Products for Human and Veterinary Use: Annex 1, Manufacture of Sterile Medicinal Products (corrected version), Annex 1 (2008).
Acknowledgments
Produced using materials originally authored by Mark Hallowrth from the Life Sciences division of Particle Measuring Systems.
About Particle Measuring Systems
Particle Measuring Systems has 35 years of experience designing, manufacturing, and servicing microcontamination monitoring instrumentation and software used for detecting particles in air, liquid, and gas streams, as well as molecular contamination monitoring.
Specific applications include cleanroom monitoring, parenteral sampling, filter and in-line testing in deionized water and process chemicals, and point-of-use monitoring of inert gases and in-situ particle monitoring. Specialty monitoring includes parts cleanliness testing with a highly automated solution.
Particle measuring systems
Whether you want to protect a product or meet industry requirements, such as ISO 14644, USP 797, or GMP, Particle Measuring Systems offers a wide range of particle counters and molecular monitors to meet your needs. With 35 years of experience, it has proven reliability to support your application.
Particle counters
Protect your product with our reliable particle counters. PMS has airborne, portable, and liquid particle counters for a wide variety of applications, including DI water, chemicals, and cleanroom monitoring. Compare particle counters or learn how to monitor your cleanroom or product by reading our papers.
Molecular contamination monitors
Molecular contamination causes costly problems for high-value products, production processes, and equipment surfaces. PMS has solutions for both Airborne Molecular contamination (AMC) and Surface Molecular contamination (SMC). With parts-per-trillion limits of detection, real-time sampling, NIST traceable calibrations, and various data analysis packages, you can monitor in confidence.
Gas detectors
If you need gas detectors for process control or continuous emissions monitoring, Particle Monitoring Systems can help. Get real-time, reliable results with its ammonia, hydrogen fluoride, and chlorine detectors for worker protection, CEMs, and pollution monitoring.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.