Pharmaceutical products are manufactured in a controlled environment to meet regulatory agency requirements. Cleanrooms are one example of a controlled environment: they are employed to lessen the risk of contamination and the variability of potential production environments.
Cleanrooms can be regulated to meet specific standards, since they are controlled environments. GMP regulations require these environments to be meticulously monitored to ensure that there is a developed and consistent awareness of current environmental conditions for both viable and non-viable contamination.
Cleanrooms are the key starting points for controlling contamination. Cleanrooms are defined as rooms in which air filtration and distribution, construction materials, utilities, and equipment are controlled. Operational procedures are defined and regulated to ensure airborne particle concentrations meet suitable classifications for particulate cleanliness.
There are two key basic types of cleanrooms:
Turbulent dilution of contamination
A turbulent airflow cleanroom employs the exchange rate of filtered air to dilute contamination to an acceptable level. HEPA-filtered air is delivered via a central system (through diffusion panels in the ceiling) at a rate of 10-40+ room volumes per hour (Air Exchange Rate).
This value depends on the operations and number of personnel within each area. To maintain comfort, the return air is recirculated, with a percentage of fresh air added.
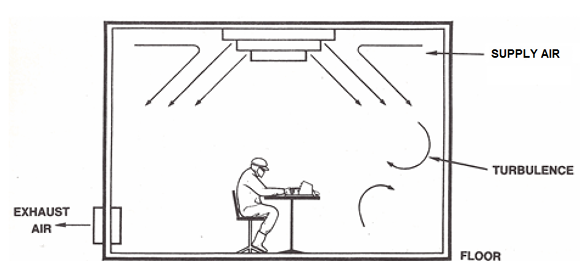
Figure 1. Turbulent Dilution of Contamination. Image Credit: Particle Measuring Systems
Unidirectional air wash
Unidirectional air cleanrooms use airflow velocity as a shroud, preventing extraneous particles from a potential contaminant source from affecting adjacent areas. If particles are generated by the process or operators, the airflow is also designed to sweep them away from the critical zone; this can be confirmed by the smoke airflow visualization test. Flow rates are optimally 0.36-0.54 m/s, though other airflow velocities can be used.
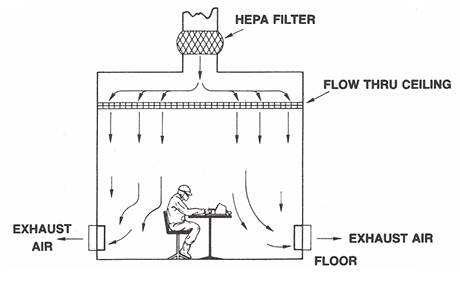
Figure 2. Unidirectional Air Wash. Image Credit: Particle Measuring Systems
In a cleanroom, operations often take place on HEPA-filtered benches, which allow containers, products, and processing equipment to be manually manipulated. Flow direction within such benches can be horizontal or vertical.
Horizontal flow moves directly toward the operator and sweeps any operator-borne contaminants out of the critical zone. Air flows down over the process when vertical flow is employed, ensuring a shroud is maintained around the critical areas.
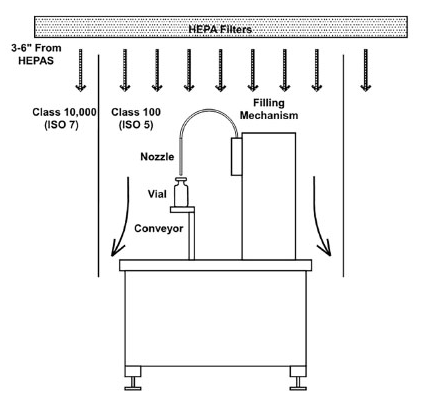
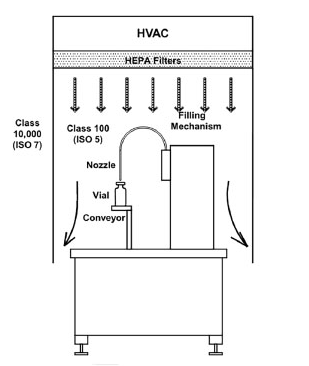
Figure 3. Filling Mechanism Diagrams. Image Credit: Particle Measuring Systems
Vertical flows also allow fill-and-finish equipment to be automated and enclosed within a designed space. The filling machine can be opened for access from above and to utilize the unidirectional airflow within the room to ensure that the process is isolated from outside activities within the room’s general environment.
Restricting access to the critical processing zone can be achieved using fixed or flexible curtains, providing a degree of isolation between the critical area and the general room environment, allowing operators to work on the process if necessary.
A dedicated filter can alternatively be used to deliver filtered air only to the processing equipment. This enables the background areas to have a lower grade of air and a higher degree of separation between the critical and background spaces.
This design can be scaled up to create restricted access barrier protection (RABS), in which the critical zone can be accessed only through gloved access ports. Full isolation can be achieved using fully closed isolator systems, which also enable enclosed sanitation and sterilization processing.
The nature of the activities and the cleanroom used will also affect the type of environmental monitoring required. With easier operator access comes a greater risk of contamination, because personnel are the greatest contributors of airborne contaminants in cleanroom settings.
Acknowledgments
Produced using materials originally authored by Mark Hallworth from Particle Measuring Systems
About Particle Measuring Systems
Particle Measuring Systems has 35 years of experience designing, manufacturing, and servicing microcontamination monitoring instrumentation and software used for detecting particles in air, liquid, and gas streams as well as molecular contamination monitoring.
Specific applications include cleanroom monitoring, parenteral sampling, filter and in-line testing in deionized water and process chemicals, and point-of-use monitoring of inert gases and in-situ particle monitoring. Specialty monitoring includes parts cleanliness testing with a highly automated solution.
Particle measuring systems
Whether you want to protect a product or meet industry requirements, such as ISO 14644, USP 797, or GMP, Particle Measuring Systems offers a wide range of particle counters and molecular monitors to meet your needs. With 35 years of experience, it has the proven reliability to support your application.
Particle counters
Protect your product with our reliable particle counters. Particle Measuring Systems has airborne, portable, and liquid particle counters for a wide variety of applications, including DI water, chemicals, and cleanroom monitoring. Compare particle counters or learn how to monitor your cleanroom or product by reading our papers.
Molecular contamination monitors
Molecular contamination causes costly problems for high-value products, production processes, and equipment surfaces. It has solutions for both Airborne Molecular contamination (AMC) and Surface Molecular contamination (SMC). With parts-per-trillion limits of detection, real-time sampling, NIST traceable calibrations, and various data analysis packages, you can monitor in confidence.
Gas detectors
If you need gas detectors for process control or continuous emissions monitoring, Particle Measuring Systems can help. Get real-time, reliable results with its ammonia, hydrogen fluoride, and chlorine detectors for worker protection, CEMs, and pollution monitoring.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.