In the pharmaceuticals sector, a primary objective of regulatory guidelines is to maintain control over contamination and preserve the sterility of products manufactured within a pharmaceutical cleanroom.
These controlled zones are designed to monitor and manage contamination levels, ensuring they meet specified cleanliness standards.1 To achieve this objective, consistent adherence to all GMP requirements is essential.
Microbiologically controlled environments are integral to various pharmaceutical industry applications, serving as a crucial tool for mitigating contamination risks in aseptic processing.
Products created in these settings include bulk sterile intermediates, finished sterile pharmaceuticals, excipients, and occasionally medical devices.
Consequently, any potential contamination of pharmaceutical products could pose significant risks to patients, who are the ultimate recipients. Therefore, ensuring product and patient safety is paramount
A key method for controlling contamination involves monitoring the environment for both viable and nonviable particles, ensuring all contamination levels remain within GMP parameters.
Over time, the approach to monitoring aseptic processes, as recommended by guidelines, has evolved. The Annex 1 revision published in August 2022 signifies a further shift towards a continuous monitoring methodology.
This implies that monitoring activities should be conducted throughout the entire duration of critical processes.2
As Annex 1 specifies regarding total particle monitoring:
9.16 For Grade A, particle monitoring should be undertaken for the full duration of critical processing, including equipment assembly.
9.17 The Grade A area should be monitored continuously (for particles ≥ 0.5 and ≥ 5 μm) and with a suitable sample flow rate (at least 28 liters (1 ft3) per minute) so that all interventions, transient events, and any system deterioration is captured.
The system should frequently correlate each individual sample result with alert levels and action limits at such a frequency that any potential excursion can be identified and responded to in a timely manner. Alarms should be triggered if alert levels are exceeded. Procedures should define the actions to be taken in response to alarms, including the consideration of additional microbial monitoring.
Regarding viable particle monitoring:
9.24 Continuous viable air monitoring in Grade A (e.g., air sampling or settle plates) should be undertaken for the full duration of critical processing, including equipment (aseptic set-up) assembly and critical processing.
A similar approach should be considered for Grade B cleanrooms based on the risk of impact on the aseptic processing. The monitoring should be performed in such a way that all interventions, transient events, and any system deterioration would be captured, and any risk caused by interventions of the monitoring operations is avoided.
As illustrated, Section 9 of Annex 1 addresses the monitoring of production processes and classified environments concerning the enumeration of viable and nonviable particles. The focus of Section 9 is routine monitoring, system design, the establishment of alarms, action limits, and the analysis of data trends.2
Previously, Annex 1 primarily regulated and discussed the qualification and classification of cleanrooms. In the new Annex, with its revised approach, the emphasis shifts to cleanrooms and process monitoring. Consequently, the concept of continuous monitoring in aseptic environments holds considerable importance.

Figure 1. Scheme of the Main Steps in the Pharmaceutical Industry. Image Credit: Particle Measuring Systems
Total particles continuous monitoring
Using calibrated particle counters, nonviable particle monitoring quantifies the number and size of particles in the air.5 Particles exhibit variations in size, shape, and density.
Their size is typically expressed as an equivalent particle diameter or the spherical diameter of a unit density particle that settles in the air at the same velocity as the particle in question.
Current regulations advise against using portable instruments for continuous particle surveillance within Grade A areas, as portable devices necessitate human intervention, a significant source of contamination.
Therefore, it is imperative to plan for the use of remote instruments for monitoring tasks. Remote instruments facilitate setup procedures (as detailed in the relevant section of Annex 1) and can be operated and resumed within an aseptic zone without human presence, which is identified as a major risk to product quality.
Remote instruments enable users to avoid interfering with aseptic filling operations, thanks to isokinetic probes.
Key advantages of remote instruments include:
- Measurement near the critical zone: aligned with FDA guidance (within 1 ft.)
- Situated just above the critical point, but not directly overhead to avoid influencing airflow or creating turbulence
- Verification that clean air is being supplied to the critical area, not just near the filters
Filling lines are a prime application for remote instruments. Within filling lines, typically situated in Grade A zones, monitoring can be performed at a minimum of three locations:
- Entry/accumulator
- Point of fill (fill needle)
- Stopper area/stopper bowl
Remote instruments also enhance the profitability of monitoring freeze-drying areas, as previously mentioned in earlier Annex 1 revisions, characterized as follows:
34. Prior to the completion of stoppering, transfer of partially closed containers, as used in freeze-drying should be done either in a Grade A environment with a Grade B background or in [a] sealed transfer tray in a Grade B environment.
116. Partially stoppered freeze-drying vials should be maintained under Grade A conditions at all times until the stopper is fully inserted.
Remote instruments and these regulations aid in controlling particle contamination, particularly in two critical areas:
- Transfer from filler to freeze dryer
- Lyo transfer carts – using WiFi communication
Alternatively, in blow-fill-seal operations, remote continuous monitoring significantly simplifies the control of the most delicate aspects, such as:
- Achieving a balance to ensure the area is clean while data quality is maintained
- Small area: precise point placement can be challenging
- Proximity to the filter may yield suboptimal data
- Avoiding plastic molding smoke near the fill point
- Consideration for monitoring incoming air pipes
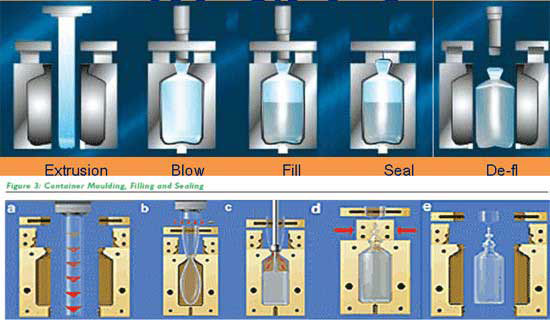
Figure 2. Blow-Fill-Seal Operations. Image Credit: Particle Measuring Systems
Regarding BioSafety cabinets and remote instruments:
- The number of monitoring points depends on the cabinet's size.
- Typically, one particle and one microbial point are sufficient.
- If there are two filter banks, consider two particle and two microbial points, or a point positioned centrally.
- Mount sensors below or to the side of the cabinet
- Route tubing through the side or back wall

Figure 3. BioSafety Cabinet. Image Credit: Particle Measuring Systems
Particular attention is also warranted for next-generation filling lines, such as isolators, where continuous particle control is now a standard expectation. Key advantages include:
- Isolator access is more challenging.
- Install ports can be installed to plumb transport tubing internally: bulkhead or tri-clamp sanitary fittings are commonly used.
- Mount sensors positioned below or to the side of the isolator: directly below is optimal for particle transport, though less convenient for sensor access.
It is important to note that any instruments used for contamination monitoring must be validated by the customer in accordance with the technical and environmental requirements.
The PMS Advisory team guides and assists customers in adopting the most effective scientific strategy for instrument validation (refer to the section "How can PMS help you with these crucial activities" within the paper "Validation And Qualification Approach Outlined In The New Annex 1 Revision").
Which instruments are capable of performing continuous particle monitoring?
Figure 4, presented below, offers a clear overview.
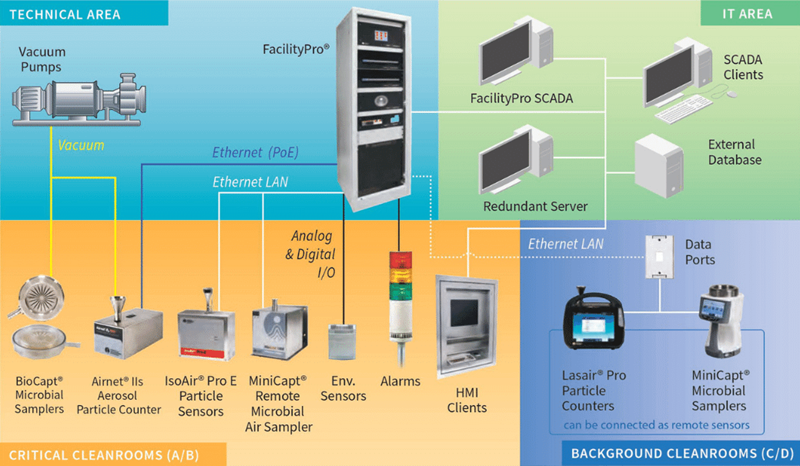
Figure 4. Fixed Sensors with Central Vacuum (PMS Product Schematic). Image Credit: Particle Measuring Systems
PMS provides numerous remote instruments, including:
- MiniCapt® Mobile Microbial Air Samplers (configurable/connectable as remote or portable)
- Lasair® Pro Particle Counters (configurable/connectable as remote or portable)
- BioCapt® Microbial Sensors (Single Use or Stainless Steel)
- MiniCapt® Pro Remote Microbial Air Samplers
- Airnet® II Aerosol Particle Counters
- IsoAir® Pro Particle Sensors
All these instruments and their associated data can be managed via FacilityPro® and interacted with on various PMS or external software platforms.
Viable continuous monitoring: Active air and passive air sampling
Continuous viable monitoring involves the detection and quantification of culturable microorganisms in the air. Sampling locations are determined based on a risk assessment (refer to the section "The importance of continuous monitoring and risk assessment technical support"), and sampling is performed using various methods, including:
- Active air sampling (e.g., BioCapt Single Use supplied by PMS)
- Passive air sampling (e.g., settle plates)
- Viable particle counting using fluorescence technology
Airborne biological particles, also known as bioaerosols, are a complex combination of various components, ranging from simple organic molecules in the nanometer range to viruses, bacteria, bacterial spores, mold spores, hyphae, and pollen. These components become airborne as individual particles or aggregates.
Scientific studies have indicated that organisms potentially impacting human health are typically in the size range of 4–20 μm. The following figure illustrates the Relative Particle size of microorganisms.1
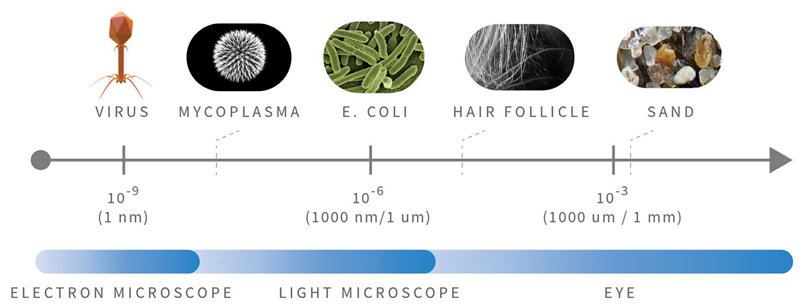
Figure 5. Relative Particle Sizes. Image Credit: Particle Measuring Systems
For viable monitoring, two continuous monitoring methods can be applied:
- Active air sampling
- Passive air sampling
Active air sampling
An active monitoring system comprises a pump that draws a specified volume of air at a particular rate and passes it over a dynamically and structurally designed plate, allowing microorganisms to deposit on the agar.
Where microorganisms are present, the plate will exhibit bacterial growth, thus indicating contamination. Air intake via this method can be conducted continuously with periodic plate changes, depending on the conditions and outcomes of the method's validation phase.
Particle Measuring Systems, as the developer and supplier of BioCapt Single Use (a viable active air sampler), has published a paper demonstrating the efficiency of BioCapt Single Use for up to four hours of continuous monitoring.
Further insights can be gained from the paper detailing a case study on the single-use device's efficacy under specific conditions: “Study of Single Use Device Performance After 4-Hour Continuous Sampling.
The sampling rate, duration, and type of sampling device can significantly influence the viability of the collected microorganisms. Impingement devices may not be suitable for sampling airborne viable particles due to their limited sampling volume, low sampling rate, and tendency to disrupt viable particle clumps.
Given the numerous and diverse microbial air sampling systems commercially available, the selection for a particular application should consider, at a minimum, the following factors:
- Type and size of viable particles to be sampled
- Sensitivity of the viable particles to the sampling procedure
- Expected concentration of viable particles
- Capability to detect high or low levels of bio-contamination
- Appropriate culture media
- Time and duration of sampling
- Ambient conditions of the sampled environment
- Disruption of unidirectional airflow by the sampling apparatus
- Sampler properties, such as:
-
- Appropriate suction flow rate for low levels of viable airborne particles
- Suitable impact/airflow velocity
- Collection accuracy and efficacy
- Ease of handling (weight, size) and operation (simplicity of use, auxiliary equipment, reliance on vacuum pumps, water, electricity, etc.)
- Ease of cleaning, disinfection, or sterilization
- Potential intrinsic addition of viable particles to the bio-contamination being measured. The exhaust air from the sampling apparatus should not contaminate the environment being sampled or be re-aspirated by the sampling device.
Passive air sampling
Passive air monitoring involves using settle plates exposed to the environment. The gravitational sedimentation of microorganisms allows their deposition and subsequent growth on the plates.
Due to particle deposition via gravitational sedimentation, turbulent airflow is the most suitable condition for particle collection on passive surfaces such as settle plates.
In Annex C of ISO 14698-1:2003, the definition of a settle plate states that passive microbial air sampling devices like settle plates do not measure the total quantity of viable particles in the air but rather the rate at which viable particles settle on surfaces.
This distinction between settle plates and viable active air sampling is significant and represents a limitation of passive air monitoring. Passive air monitoring yields different information that is less comprehensive in the overall context.
The settle plate method relies on the deposition of Molecular & Cellular Proteomics (MCPs) and microorganisms onto the agar plate surface. This method is strictly qualitative, with no possibility of determining collection efficiency.
It is not recommended for use in Grade A areas (ISO 5) where high air velocities and frequent air changes are common. These parameters significantly impede the settling of standalone particles, particles containing microorganisms, and standalone microorganisms.
Its application is more practical in static environments or areas with infrequent air changes. Continuous microbiological monitoring of air in critical areas using validated methodologies (such as active air samplers with recovery rates of ≥ 70 %) should be more widely adopted.
This contamination control strategy fulfills the regulatory requirement for enhanced process understanding and greater sterility assurance of released products.
Furthermore, validation presents a challenge with settle plates, as validation is limited to the characteristics of the soil. Validating an active air system is more straightforward, encompassing various aspects: biological recovery efficiency, dehydration, agar volume, and so forth.
Further details can be found in the paper: "Validation And Qualification Approach Outlined In The New Annex 1 Revision".
Particle Measuring Systems can assist customers in selecting the most appropriate method and devices based on their specific environment and process.
The importance of continuous monitoring and risk assessment technical support
In aseptic environments, it is essential to acknowledge that the presence of personnel can impact environmental contamination.
Where human intervention occurs, contamination cannot be entirely eliminated; GMP guidelines state that achieving zero microbial contamination is impossible when personnel are present in a pharmaceutical cleanroom.3
Given that environmental monitoring activities are conducted by operators, the monitoring process itself could inadvertently cause contamination or lead to false positive results (in the context of oRABS filling machines or Grade A environments).
Continuous monitoring is, therefore, one of the most effective approaches because it:
- Facilitates observation of the impact of all interventions and operations throughout the process
- Minimizes the need for human intervention, thereby reducing the risk of contamination and false positive results
It is crucial to consider the influence of human activity within the cleanroom, as it can affect the contamination of the environment, materials, or product. A continuous monitoring approach helps to capture any single particle variation during the process, and the resulting data can suggest potential causes for correction to mitigate contamination risks.
Similarly, continuous monitoring reduces human involvement as it eliminates the need to change plates or halt monitoring at specific intervals; it works from the first minute until the process concludes.4
To support the aforementioned points, adopting a risk assessment approach is vital for a comprehensive understanding of the optimal locations for sampling points and the primary areas requiring control from a risk perspective. 2
A risk assessment is defined by ICH Q9 as a systematic process for organizing information and data to inform risk-based decisions made as part of a risk management process.6
The 2022 revision of Annex 1 emphasizes the significance of risk management and risk assessments for maintaining quality and ensuring sterility.
A risk assessment is fundamental because decisions regarding sample point placement and the implementation of a monitoring plan must be documented and based on the generated risk assessment and historical/scientific data.
In Annex 1, paragraph 9.25 illustrates the utility of risk assessment in developing a sound and strategic monitoring plan:
-
- A risk assessment should evaluate the locations, type and frequency of personnel monitoring based on the activities performed and the proximity to critical zones. Monitoring should include sampling of personnel at periodic intervals during the process.
Sampling of personnel should be performed in such a way that it will not compromise the process. Consideration should be given to monitoring personnel following involvement in critical interventions (at a minimum glove but may require monitoring of areas of gown as applicable to the process) and on each exit from the Grade B cleanroom (gloves and gown).
Where monitoring of gloves is performed after critical interventions, the outer gloves should be replaced prior to continuation of activity. Where monitoring of gowns is required after critical interventions, the gown should be replaced before further activity in the cleanroom.
Summary
Key takeaways are listed below:
- Complete continuous process monitoring: Monitor the entire process continuously at all critical stages, commencing with equipment setup.
- Sampling points determined by risk assessment: Conduct a thorough risk assessment to identify sampling points for viable and nonviable counters.
- Provide concrete and effective data: The aim of continuous monitoring is to yield concrete and effective data, reducing the risk of false positives in terms of data quality and production efficiency.
- Monitoring must not disrupt the process: Continuous monitoring minimizes human intervention, thereby reducing contamination risk.
Conclusions and considerations
The critical importance of continuous monitoring in Grade A areas (and Grade B areas supporting Grade A) has been highlighted.
The use of instrumentation that does not require human intervention is highly recommended. The implementation of continuous microbiological monitoring instruments is considered to eliminate the most significant source of user contamination.
It is also feasible to explore the potential of replacing settle plates with continuous microbiological monitoring tools, given the differing outputs of each instrument. Risk assessments form the foundation for selecting monitoring points, are essential for process comprehension, and provide vital insights into the functionality of what is being monitored.
Particle Measuring Systems offers a range of products and consultancy services to address customer challenges in monitoring cleanrooms.
References
- Dalmaso, Dalmaso, G., Campanella, A. and Lazzeri, P. (2020). Continuous and Effective Microbiological Air Monitoring in Critical Environments: a Comparison of Analytical Methodologies. PDA Journal of Pharmaceutical Science and Technology, (online) 74(4), pp.446–455. Available at: https://eurekamag.com/research/069/817/069817485.php?srsltid=AfmBOop_Ju7tCsqStrz2wmmYZ3UOIeZ-wADmYhJvSevQd-vidfN-pZfT.
- European Commission (2022). The Rules Governing Medicinal Products in the European Union Volume 4 EU Guidelines for Good Manufacturing Practice for Medicinal Products for Human and Veterinary Use. Available at: https://health.ec.europa.eu/document/download/e05af55b-38e9-42bf-8495-194bbf0b9262_en?filename=20220825_gmp-an1_en_0.pdf.
- U.S. Pharmacopeial Convention (2025). General Chapter <1116> Microbiological Control and Monitoring of Aseptic Processing Environments. Available at: https://doi.usp.org/USPNF/USPNF_M99835_01_01.html.
- 1_GUID- B9A1739F-E171-4E11-A0C7-E43A318EA17F_1_en-US, General Chapter, <1116> Microbiological Control and Monitoring of Aseptic Processing Environments
- U.S. Pharmacopeial Convention (2024). ⟨1211⟩ Sterilization and Sterility Assurance of Compendial Articles. Available at: https://doi.usp.org/USPNF/USPNF_M99930_05_01.html.
- ICH. ICH Q9 Quality risk management. Available at: https://database.ich.org/sites/default/files/Q9_Guideline.pdf.
About Particle Measuring Systems
Particle Measuring Systems has 35 years of experience designing, manufacturing, and servicing microcontamination monitoring instrumentation and software used for detecting particles in air, liquid, and gas streams, as well as molecular contamination monitoring.
Specific applications include cleanroom monitoring, parenteral sampling, filter and in-line testing in deionized water and process chemicals, and point-of-use monitoring of inert gases and in-situ particle monitoring. Specialty monitoring includes parts cleanliness testing with a highly automated solution.
Particle measuring systems
Whether you want to protect a product or meet industry requirements, such as ISO 14644, USP 797, or GMP, Particle Measuring Systems has a large variety of particle counters and molecular monitors to meet your needs. With 35 years of experience, it has proven reliability to support your application.
Particle counters
Protect your product with our reliable particle counters. PMS has airborne, portable, and liquid particle counters for a wide variety of applications, including DI water, chemicals, and cleanroom monitoring. Compare particle counters or learn how to monitor your cleanroom or product by reading our papers.
Molecular contamination monitors
Molecular contamination causes costly problems for high-value products, production processes, and equipment surfaces. PMS has solutions for both Airborne Molecular contamination (AMC) and Surface Molecular contamination (SMC). With parts-per-trillion limits of detection, real-time sampling, NIST traceable calibrations, and various data analysis packages, you can monitor in confidence.
Gas detectors
If you need gas detectors for process control or continuous emissions monitoring, Particle Monitoring Systems can help. Get real-time, reliable results with its ammonia, hydrogen fluoride, and chlorine detectors for worker protection, CEMs, and pollution monitoring.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.