The evolution of quality assurance systems within pharmaceutical entities has accelerated more than any other area over the past 15 years.
During this time, a specialized group focused on traditional compliance expanded to encompass quality specialists in domains, such as validation, product authorization, operations, sterility assurance, and other niche areas.
The underlying objective of this expansion was to foster greater integration quality linkage between production and engineering. Gradually, quality became integral to every phase of pharmaceutical production, shifting from a segmented approach to a more unified one. A new challenge now looms.
The advent of the fourth industrial revolution (Pharma 4.0™ 1) ushers in the era of the "Intelligent Facility," where digital technologies and automated processes will converge to facilitate highly complex applications and product lifecycles.
Within this transformed landscape, quality specialists will confront the necessity of redefining their roles and restructuring their organizations' quality frameworks to align with the principles of Data Quality.
The ICH vision for pharma
Since 1990, the International Council for Harmonization (ICH) has strived to establish a globally consistent approach to ensure the development and registration of safe, effective, and high-calibre medicines in the most efficient way.
Between 2005 and 2008, through the release of guidelines Q9 and Q10, the council outlined the nature and operational aspects of quality systems essential for ensuring the utmost quality in drug products.
Over these years, it's evident that most global pharmaceutical companies have adapted their quality systems to conform to ICH guidelines, thereby amplifying the council's influence within these organizations.
Concurrently, the core value and challenge presented by the ICH lies in applying scientific and risk-based quality system tenets across the entire product lifecycle.
Emerging terminology
The ICH Q9 and Q10 guidelines represented a significant advancement, marking what could be termed the peak era of blockbuster pharmaceuticals, or “Pharma 2.0”.
This period was defined by large-scale manufacturing of uniform products over extended durations. Some of the defining characteristics of these Pharma 2.0 organizations were the use of a hierarchical management structure rooted in the experience of decision makers, and a siloed operational model.
Following the ICH guidelines, quality departments began to dismantle this mentality, making the quality function so pervasive that it appeared to operate seamlessly. The pivotal figure in this evolving, less rigid structure was the quality specialist. Functioning as subject matter experts, they were tasked with synthesizing field-derived information with their own quality expertise to inform decisions grounded in scientific principles
Presently, the landscape is rapidly changing. The already swift transition of conventional pharmaceuticals toward biotechnology and genetics has been expedited by the COVID-19 pandemic, market shifts, and technological progress.
These factors are propelling the pharmaceutical sector into a new industrial epoch, or, as designated by ISPE, Pharma 4.0™.
As with any new era, specific keywords are crucial for comprehending the critical distinctions from prior periods. For the fourth industrial revolution, digital maturity, automation, and holistics are the most essential terms to grasp.
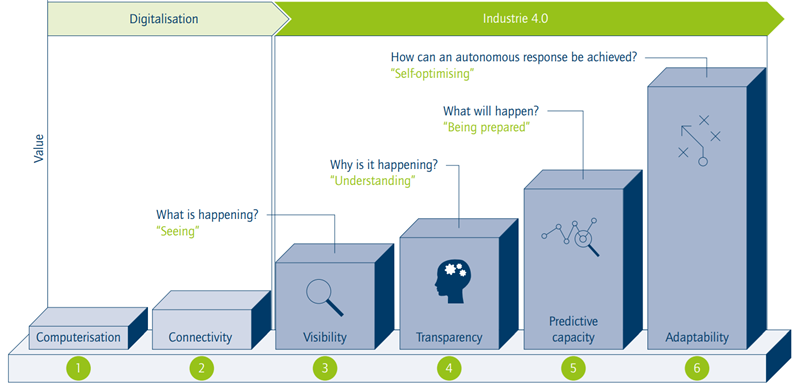
Figure 1. Acatech Digital Maturity Stages. Image Credit: FIR a V. at RWTH Aachen University
From digital maturity to the data quality concept
A significant indicator of the fourth industrial revolution in Pharma is the emergence of smart facilities. After years of conceptual development, these technologically enabled sites are becoming a reality. Broadly, a facility is deemed "smart" when it meets all the Acatech digital maturity benchmarks:
- Stage 1: Computerization
- Stage 2: Connectivity
- Stage 3: Visibility
- Stage 4: Transparency
- Stage 5: Predictive Capacity
- Stage 6: Adaptability
Achieving Stage 1 signifies a transition from paper-based to data-centric operations, while establishing Connectivity (Stage 2) involves moving from standalone devices to integrated systems where data can be shared across various departments.
The initial phase of the Pharma 4.0 shift is Visibility (Stage 3), which is the ability to observe real-time events through data streams from sensors within the digital infrastructure established in prior stages.
It is evident that a substantial portion of the pharmaceutical industry is currently at this stage. Here, key performance indicators (KPIs) are gathered and accessible online.
Nevertheless, as noted previously, it still relies on quality specialists to make decisions informed by a combination of experience and scientific reasoning, employing historical data analysis performed retrospectively.
The digital representation of processes created in the first three stages is complemented by "automation" delivered by the subsequent three stages.
Transparency (Stage 4) is defined by the capability to identify and interpret interactions within the digital representation as they occur, enabling the determination of true root causes (deductive reasoning).
Predictive Capacity (Stage 5) is vital for automated decision-making. In Stage 4, systems begin to analyze trends, projecting the digital representation into the future to implement appropriate corrective actions promptly.
The sixth and final stage is Adaptability, which is the automated capacity to adjust processes based on corrected outcomes without human intervention, despite evolving inputs and conditions.
Not all pharmaceutical companies are required to achieve every stage.
For instance, entities producing high-volume consumer goods may not inherently need to progress through the digital maturity stages.
Conversely, adopting this comprehensive approach will be indispensable for the sustainability of businesses involved with high-tech products, advanced therapies, and premium-value offerings.
Moreover, it is important to recognize that an organization can meet all five Acatech Digital Maturity stages simply by possessing the capacity to implement them. However, it is the responsibility of quality specialists and system proprietors to translate that potential into actuality.
"Holistic" signifies a comprehensive perspective, encompassing all influencing factors and potential contexts. Holism in scientific inquiry represents an approach that prioritizes the study of intricate systems.
Systems are viewed as unified wholes, where individual components are best understood within their context and in relation to each other and the overall structure.
This approach to decision-making in pharmaceutical production can be termed "Data Quality." It pertains to the extensive data lifecycle through the digital maturity stages, with its ultimate goal of converting collected data into informed decisions (CAPA).
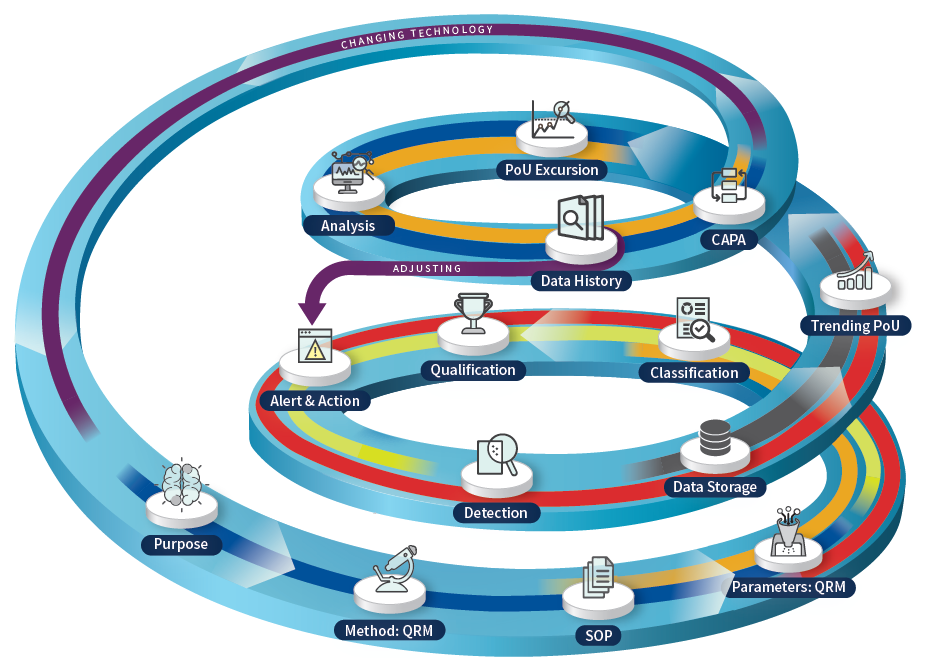
Figure 2. Data Quality Spiral. Image Credit: Particle Measuring Systems
The evolving challenge
Systems will advance to autonomously correct, anticipate, and adapt existing processes, and in a short time, they will supersede current decision-making methodologies.
A few key questions emerge: what will become of the quality specialist as envisioned by the ICH? Is Data Quality alone sufficient to guarantee the quality of pharmaceutical products?
As with any industrial transformation, technological advancement alone is insufficient to redefine an era; cultural progress must be equally impactful.
Cultural shifts can manifest as top-down initiatives, driven by senior management, or as bottom-up movements. In both scenarios, shifts indicate the presence of a segmented organization requiring evolution.
In Pharma 4.0, following the integration of digitalization and automation, companies will no longer require organizations structured with distinct levels and departments.
For the first time in the industry's history, both top-down and bottom-up cultural changes are happening concurrently. Personnel at all tiers must acquire diverse skills within a fluid, process-centric organizational framework.
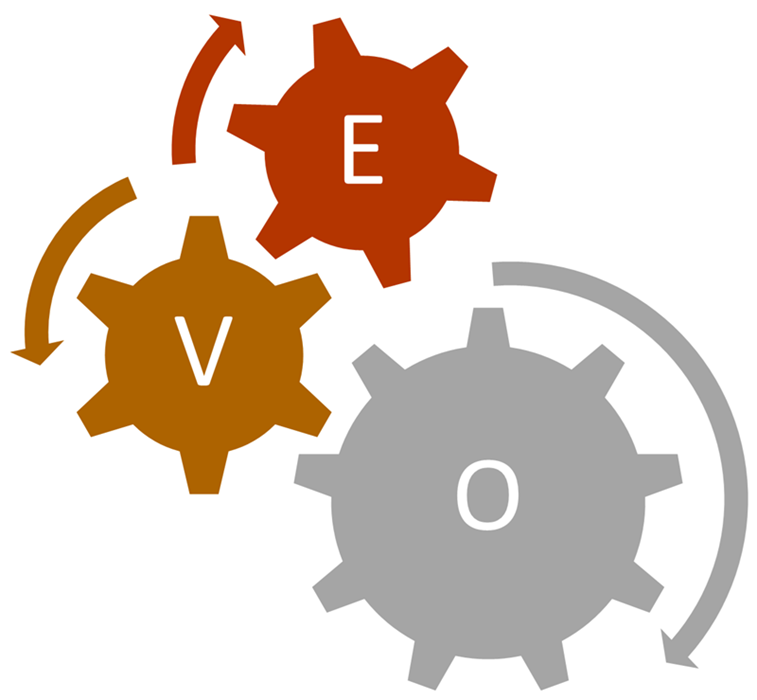
Figure 3. Empowered Value-Driven Organization (EVO). Image Credit: Particle Measuring Systems
This principle also applies to the quality unit and its experts. Due to the increased speed and accuracy of quality information dissemination, there will be limited time to seek advice and approvals.
A comprehensive understanding of quality concepts, alongside process, manufacturing, and statistical knowledge, will be essential skills for emerging experts. The quality department within Pharma 4.0 will operate as an Empowered Value-driven Organization (EVO), where each expert is equipped with data quality insights, enabling them to make decisions independent of a hierarchical structure.
The Automation stages (transparency, predictive capacity, and adaptability) ensure these decisions are made to achieve objectives such as enhanced system reliability, improved product quality, and reduced documentation requirements.
Indeed, recent market analyses suggest that automation is poised to replace a significant portion of "predictable tasks," while simultaneously increasing the demand for specialists capable of managing decision-making processes for "unpredictable" elements.
The scale of the required increase in specialized personnel is substantial. Particle Measuring Systems projects a global growth of 150 %, involving hundreds of millions of individuals worldwide who will need to upskill in conjunction with this new industrial revolution.
Concluding thoughts
In the era of the fourth industrial revolution, data quality serves as the fundamental bedrock upon which pharmaceutical companies must build their production capabilities.
This will be achieved through a profound integration of all manufacturing stages, which are currently losing their distinct boundaries. On one hand, digitalization and automation will supplant work categories we can define as "predictable."
On the other hand, the vast volume of data generated, which forms the basis for decision-making, necessitates multi-skilled experts for its interpretation.
Advanced proficiencies in statistics, process engineering, contamination control, and microbiology are illustrative examples of the knowledge base that will be required.
The role of these new quality specialists will become more pivotal than in the past. The fluid organizational structure fostered by the data quality paradigm will transform traditional departments into EVOs, where critical decisions will originate from quality specialists without recourse to a supervisory hierarchy.
References
- ICH. ICH Harmonized Tripartite Guideline – Quality Risk Management Q9. Available at: http://people.duke.edu/~ghturner/ICH-Guidelines/E9Step4.doc.
- ICH (2008). ICH Harmonized Tripartite Guideline – Pharmaceutical Quality System Q10. Available at: https://database.ich.org/sites/default/files/Q10%20Guideline.pdf.
- Al-Hafez N., et al. (2021). ISPE | International Society for Pharmaceutical Engineering. Holistic Control Strategy: From ICH Quality Guidelines to Pharma 4.0™. Available at: https://ispe.org/pharmaceutical-engineering/march-april-2021/holistic-control-strategy-ich-quality-guidelines-pharma.
- Wyn S., Canterbury J. (2020). ISPE | International Society for Pharmaceutical Engineering. (2020). A GAMP® Approach to Robotic Process Automation. Available at: https://ispe.org/pharmaceutical-engineering/gamp-approach-robotic-process-automation.
- Heesakkers H., et al. (2020). ISPE | International Society for Pharmaceutical Engineering. Applying Holistic Control Strategy in Pharma 4.0™. Available at: https://ispe.org/pharmaceutical-engineering/january-february-2020/applying-holistic-control-strategy-pharma-40.
- Schuh, G., et al. (2017). acatech STUDY Industrie 4.0 Maturity Index. Available at: https://fundacioperlaindustria.org/wp-content/uploads/2021/07/2020.acatech_STUDIE_Maturity_Index.Update.pdf.
- Tilley J., et al. (2019).Industrial robotics Insights into the sector’s future growth dynamics. Available at: https://www.mckinsey.com/~/media/mckinsey/industries/advanced%20electronics/our%20insights/growth%20dynamics%20in%20industrial%20robotics/industrial-robotics-insights-into-the-sectors-future-growth-dynamics.pdf.
About Particle Measuring Systems
Particle Measuring Systems has 35 years of experience designing, manufacturing, and servicing microcontamination monitoring instrumentation and software used for detecting particles in air, liquid, and gas streams, as well as molecular contamination monitoring.
Specific applications include cleanroom monitoring, parenteral sampling, filter and in-line testing in deionized water and process chemicals, and point-of-use monitoring of inert gases and in-situ particle monitoring. Specialty monitoring includes parts cleanliness testing with a highly automated solution.
Particle measuring systems
Whether you want to protect a product or meet industry requirements, such as ISO 14644, USP 797, or GMP, Particle Measuring Systems has a large variety of particle counters and molecular monitors to meet your needs. With 35 years of experience, it has proven reliability to support your application.
Particle counters
Protect your product with our reliable particle counters. PMS has airborne, portable, and liquid particle counters for a wide variety of applications, including DI water, chemicals, and cleanroom monitoring. Compare particle counters or learn how to monitor your cleanroom or product by reading our papers.
Molecular contamination monitors
Molecular contamination causes costly problems for high-value products, production processes, and equipment surfaces. PMS has solutions for both Airborne Molecular contamination (AMC) and Surface Molecular contamination (SMC). With parts-per-trillion limits of detection, real-time sampling, NIST traceable calibrations, and various data analysis packages, you can monitor in confidence.
Gas detectors
If you need gas detectors for process control or continuous emissions monitoring, Particle Monitoring Systems can help. Get real-time, reliable results with its ammonia, hydrogen fluoride, and chlorine detectors for worker protection, CEMs, and pollution monitoring.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.