Pharmaceutical companies create products in controlled conditions, such as cleanrooms, to ensure regulatory compliance and low risk production. Cleanrooms are rooms in which air filtration, air distribution, utilities, building materials, and equipment are maintained under controlled conditions to limit variability and particle contamination.
Unidirectional airflow is a typical contamination-management technique used in cleanrooms. A cleanroom with unidirectional airflow uses a controlled, steady airflow in parallel streamlines that move in one direction to reduce the potential movement of contaminants.
Unidirectional cleanrooms are often designed with laminar airflow hoods that direct the air. The airflow is intended to sweep particles from a potential contamination source away from the process before exhausting from the process area.
The continual laminar flow of clean air ensures that any potential contamination does not spread beyond a control area.
Unidirectional airflow design requirements
In unidirectional situations, there is an optimal airflow rate required to serve as sheathing air. This flow rate was previously defined as 90 feet per minute, which converts to approximately 0.45 m/ s.
Isokinetic sampling is required to successfully sample this air without interrupting the airflow patterns (and hence collecting the most accurate representation of the particles entrained in that airflow as possible). Isokinetic sampling operates on the following principles:
- The air entering the sampling probe has the same velocity as the surrounding undisturbed air.
- Particles that travel through a specific location and time are captured without disrupting their flow lines.
- The nozzle is designed to disturb the air minimally, allowing only the same particles to enter as would have passed through without it.
To reduce sample loss, the cleanroom’s airflow velocity must match the linear velocity into a particle counter's probe aperture as closely as possible. If the velocities do not match, mistakes occur (sub- and superisokinetic sampling).
Some variation is permitted because, in many environments, airflow is not always possible or steady at specified flow rates. This must be confirmed to preserve the desired effect of ensuring that the sheath air manages particles within a desired zone and exhausts them properly; this is accomplished by airflow visualization experiments.
When sampling with an incorrect velocity into the sampling probe, anisokinetic sample conditions (superisokinetic or sub-isokinetic) influence the data from a total count and particle distribution standpoint, since more or fewer particles might be collected under these conditions.
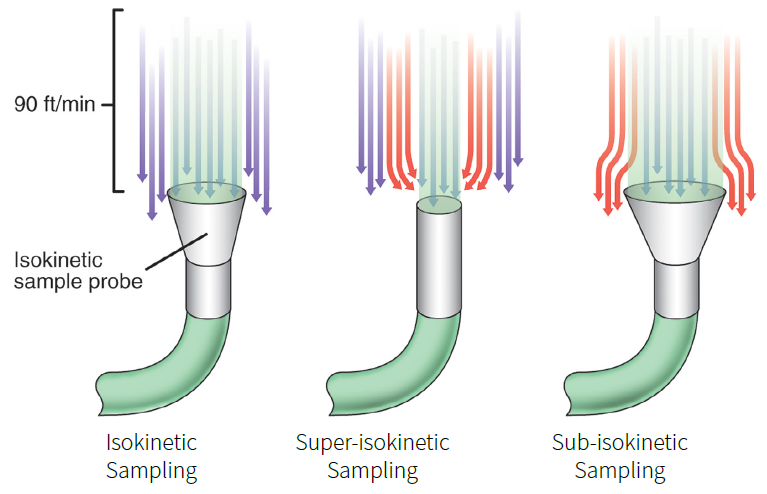
Figure 1. Isokinetic Sampling Diagram. Image Credit: Particle Measuring Systems
- Sub-isokinetic sampling: occurs when the air surrounding the sample probe has a higher velocity than the air entering it. In this example, a layer of air surrounds the probe, pushing flow lines away from it.
Heavy particles will continue to move in the straight-down, unidirectional flow line even if the flow line on which they were placed is part of the sheath layer, as their momentum takes them into the probe. This causes an oversampling of bigger particles.
- Super-isokinetic sampling: occurs when the air surrounding the sample probe has a lower velocity than the air entering it. In this scenario, flow lines are twisted to draw additional lines into the probe.
Heavy particles will continue to migrate in the straight-down, unidirectional flow line even after the flow line on which they were located is drawn into the probe, as their momentum carries them beyond the sample probe. This leads to an undersampling of bigger particles.
Regulations and tolerance
After determining the acceptable airflow range for a given location, isokinetic sampling should be performed there. The EU GMP Annex 1 recommends:
4.30 The speed of air supplied by unidirectional airflow systems should be clearly justified in the qualification protocol including the location for air speed measurement. Air speed should be designed, measured, and maintained to ensure that appropriate unidirectional air movement provides protection of the product and open components at the working position (e.g., where high- risk operations occur and where product and/or components are exposed). Unidirectional airflow systems should provide a homogeneous air speed in a range of 0.36 - 0.54 m/s (guidance value) at the working position, unless otherwise scientifically justified in the CCS. Airflow visualization studies should correlate with the air speed measurement.
EU GMP Annex 1 Regulatory Advice
The sample probe used in unidirectional environments is designed for an air flow rate of 0.45 m/ s. However, with airflows ranging from 0.36 to 0.54 m/ s, can the probe meet isokinetic criteria at the extremities of the range?
Belyaev and Levin (Journal of Aerosol Science, Volume 5, Issue 4, August 1974) suggested that a five percent sampling error around isokinetic sampling is acceptable. This allowance is reliable throughout a wide range of airflow velocities and instrument flow rates encountered in cleanroom and clean air device applications.
The tolerance is determined by the way that particles of various sizes migrate (Cunningham slip factor) within the air-stream lines during a change in direction caused by either super-isokinetic or sub-isokinetic sampling. When the limits for the flow rates allowed by EU GMP Annex 1 (2023) are computed, the following findings are obtained.
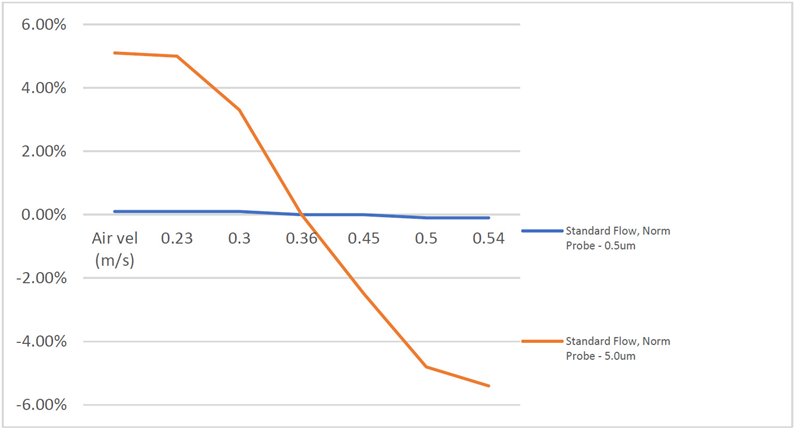
Figure 2. Anisokinetic Sampling Tolerances. Image Credit: Particle Measuring Systems
Conclusion
If a five percent tolerance for sample error is added, which is a suggested industry allowance, the range of air flow rates that can be achieved using a typical probe design is 0.22 to 0.54 m/ s, which is within the range of flow rates authorized under Annex 1 standards for isokinetic sampling.
Acknowledgments
Produced using materials originally written by Mark Hallworth from Particle Measuring Systems.
About Particle Measuring Systems
Particle Measuring Systems has 35 years of experience designing, manufacturing, and servicing microcontamination monitoring instrumentation and software used for detecting particles in air, liquid, and gas streams, as well as molecular contamination monitoring.
Specific applications include cleanroom monitoring, parenteral sampling, filter and in-line testing in deionized water and process chemicals, and point-of-use monitoring of inert gases and in-situ particle monitoring. Specialty monitoring includes parts cleanliness testing with a highly automated solution.
Particle measuring systems
Whether you want to protect a product or meet industry requirements, such as ISO 14644, USP 797, or GMP, Particle Measuring Systems has a large variety of particle counters and molecular monitors to meet your needs. With 35 years of experience, it has proven reliability to support your application.
Particle counters
Protect your product with our reliable particle counters. PMS has airborne, portable, and liquid particle counters for a wide variety of applications, including DI water, chemicals, and cleanroom monitoring. Compare particle counters or learn how to monitor your cleanroom or product by reading our papers.
Molecular contamination monitors
Molecular contamination causes costly problems for high-value products, production processes, and equipment surfaces. PMS has solutions for both Airborne Molecular contamination (AMC) and Surface Molecular contamination (SMC). With parts-per-trillion limits of detection, real-time sampling, NIST traceable calibrations, and various data analysis packages, you can monitor in confidence.
Gas detectors
If you need gas detectors for process control or continuous emissions monitoring, Particle Monitoring Systems can help. Get real-time, reliable results with its ammonia, hydrogen fluoride, and chlorine detectors for worker protection, CEMs, and pollution monitoring.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.