The release of the new EU GMP Annex 1 revision necessitates a review of current practices to ensure that the installed monitoring systems, chosen in accordance with Annex 1, comply with the necessary requirements. This article reviews the needs of Annex 1 with systems designs currently being installed.1
Cleanroom classification
The relevant classifications of pharmaceutical cleanrooms are grouped according to the particle concentration of the air that must conform to the cleanliness criteria for the manufacturing process being conducted.
The process of determining cleanroom class is based on statistically valid measurements and a function of the room’s filtration and operation status. Essentially, it is a calibration of the room to make sure it conforms to the target classification; it is not a primary function of the risk of application.2
There are three measurement phases that utilize particle counting strategies in cleanrooms:
- As built: an operational room with services connected and ready for function, but without production equipment or personnel within the facility.
- At rest: Services are fully connected, all the equipment is in place and operating as it should, but there are no personnel present.
- Operational: All equipment and personnel are present and working according to agreed procedures and protocols.
The airborne particle count test is conducted by counting particles at predetermined grid locations within the cleanroom. The test points should be spaced equally throughout the room and at operational heights to establish air quality and cleanliness in the work area.
Pharmaceutical cleanrooms are usually fully functional at Class 5 (most aseptic areas), Class 7 (surrounding areas), or Class 8 (support areas). Figure 1 offers a clear visual representation.
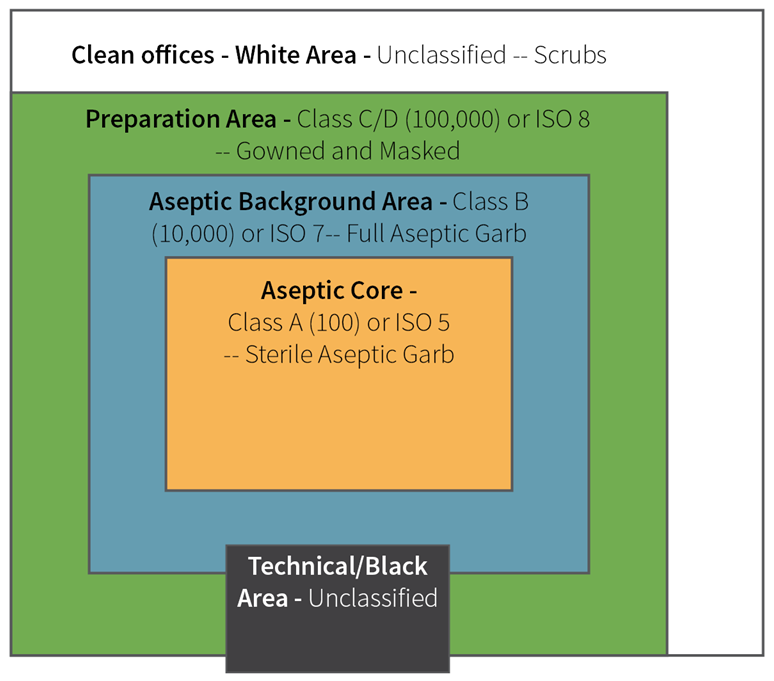
Figure 1. Diagram of a Typical ISO Rating in a Cleanroom. Image Credit: Particle Measuring Systems
Pharmaceutical cleanroom utilization
Cleanrooms are usually tested using a Light Scattering Aerosol Particle Counter (LSAPC) to determine compliance with cleanroom classification. The achieved classification dictates the activities that can be performed in that cleanroom or clean air device.
The FDA defines two areas:
- Critical areas contain products vulnerable to contamination when exposed; these areas are identified as Grade A (ISO5) within the Annex 1 document. To preserve product confidence, it is critical to perform aseptic operations in a controlled and well-maintained manner in compliance with the relevant category standards.
- Supporting clean areas are leveraged for all other activities that fall outside of the critical core; these are identified as grade B/C/D within the Annex 1 and are usually a lower risk to finished product contamination.
Once it has been confirmed that a cleanroom or clean air device meets certification cleanliness requirements, it must also be proven that control can be sustained throughout production periods.
The environment needs to be stringently monitored to offer full and consistent awareness of ongoing conditions, including the detection of periodic events, which could be catastrophic if left undetected. Continuous monitoring delivers a constant flow of information, yielding vast data quantities that can be used to observe developing trends.
Manufacturing facilities should, therefore, implement a comprehensive environmental monitoring program that includes the monitoring of non-viable and viable airborne particulates, surface viable contamination, and – in aseptic areas – personnel.
These procedures and monitoring sample points should be targeted at specific locations on a frequent basis. Warning and alarm limits should be established in each area, and corrective actions should be in place if any of the areas deviate from the expected results.
When limits are exceeded, the necessary actions taken should include an investigation into the source of the issue, the potential impact on the product, and what measures should be put in place to prevent a recurrence.
Contamination control strategy
A contamination control strategy (CCS) will incorporate the environmental monitoring program and should be rolled out across the entire facility. The CCS should identify key control points in the context of a risk assessment and evaluate how effectively the controls and monitoring measures mitigate contamination-related risks.
A review of the CCS should be conducted periodically, more so during the early phases of implementation, and it should be revised to drive the continued development of the monitoring and control methods. Ultimately, this should be focused on improving the overall quality of the process.
Elements that should be incorporated as part of CCS will include:
- Design of the plant and processes
- Premises and equipment
- Personnel
- Utilities
- Raw material controls
- Product containers and closures
- Vendor approval: key suppliers
- Outsourced services, such as sterilization, ensure the process is operating correctly.
- Process risk assessments
- Process validation
- Preventive maintenance
- Cleaning and disinfection
- Monitoring systems: the introduction of scientifically sound, modern methods that optimize the detection of environmental contamination
- Prevention: trending, investigation, corrective, and preventive actions (CAPA)
Conclusion
The revisions made to Annex 1 (2022) support the already existing requirements for monitoring protocols, which, in pharmaceutical aseptic production environments, are already well-established practices.
The inclusion of data and formal risk assessment within CCS facilitates a more extensive detailing of the continuous systems installed under previous regulations. Additional emphasis highlights how to determine and establish the sample locations and appropriate techniques based on risk, and review data to support product release.
This emphasis improves strategies and documentation requirements compared with the traditional expectations of a continuous system.
References
- European Commission (2022). The Rules Governing Medicinal Products in the European Union Volume 4 EU Guidelines for Good Manufacturing Practice for Medicinal Products for Human and Veterinary Use: Annex 1, Manufacture of Sterile Medicinal Products, European Commission. Available at: https://health.ec.europa.eu/medicinal-products/eudralex/eudralex-volume-4_en.
- International Standards Organization. Cleanrooms and associated controlled environments – Part 1: Classification of air cleanliness by particle concentration, ISO Standard No. 14644-1:2015 (2015).
Acknowledgments
Produced using materials originally authored by Mark Hallworth from Particle Measuring Systems.
About Particle Measuring Systems
Particle Measuring Systems has 35 years of experience designing, manufacturing, and servicing microcontamination monitoring instrumentation and software used for detecting particles in air, liquid, and gas streams, as well as molecular contamination monitoring.
Specific applications include cleanroom monitoring, parenteral sampling, filter and in-line testing in deionized water and process chemicals, and point-of-use monitoring of inert gases and in-situ particle monitoring. Specialty monitoring includes parts cleanliness testing with a highly automated solution.
Particle measuring systems
Whether you want to protect a product or meet industry requirements, such as ISO 14644, USP 797, or GMP, Particle Measuring Systems has a large variety of particle counters and molecular monitors to meet your needs. With 35 years of experience, it has proven reliability to support your application.
Particle counters
Protect your product with reliable particle counters. PMS has airborne, portable, and liquid particle counters for a wide variety of applications, including DI water, chemicals, and cleanroom monitoring. Compare particle counters or learn how to monitor your cleanroom or product by reading our papers.
Molecular contamination monitors
Molecular contamination causes costly problems for high-value products, production processes, and equipment surfaces. PMS has solutions for both Airborne Molecular contamination (AMC) and Surface Molecular contamination (SMC). With parts-per-trillion limits of detection, real-time sampling, NIST traceable calibrations, and various data analysis packages, you can monitor in confidence.
Gas detectors
If you need gas detectors for process control or continuous emissions monitoring, Particle Monitoring Systems can help. Get real-time, reliable results with its ammonia, hydrogen fluoride, and chlorine detectors for worker protection, CEMs, and pollution monitoring.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.