2. What should I know before I use BUDENOFALK foam?
Do not use if you have ever had an allergic reaction to BUDENOFALK foam or any of
the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines,
or are pregnant or plan to become pregnant or are breastfeeding. For more information, see Section
2. What should I know before I use BUDENOFALK foam? in the full CMI.
3. What if I am taking other medicines?
4. How do I use BUDENOFALK foam?
The recommended dose is one actuation daily, corresponding to 2 mg budesonide. BUDENOFALK
foam can be applied in the morning or evening.
This medicine may only be used rectally, so it has to be inserted through the anus.
Do NOT take it by mouth.
More instructions can be found in Section
4. How do I use BUDENOFALK foam? in the full CMI.
5. What should I know while using BUDENOFALK foam?
|
Things you should do
|
Remind any doctor, dentist or pharmacist you visit that you are using BUDENOFALK foam.
If you become pregnant while using this medicine, tell your doctor immediately.
|
|
Things you should not do
|
Do not use BUDENOFALK foam to treat any other complaints unless your doctor tells
you to.
Do not give your medicine to anyone else, even if they have the same condition as
you.
Do not stop using BUDENOFALK foam or change the dosage without checking with your
doctor.
|
|
Driving or using machines
|
This medicine is not expected to affect your ability to drive a car or operate machinery.
|
|
Looking after your medicine
|
Keep BUDENOFALK foam in the original packaging until it is time to use it.
Keep your BUDENOFALK foam in a cool dry place where the temperature stays below 25°C.
Store the pack upright.
Do not refrigerate or freeze. Protect from direct sunlight.
Keep away from flames or sparks. Contains flammable gas.
Keep it where children cannot reach it.
Use within 4 weeks of first opening.
A locked cupboard at least one-and-a-half metres above the ground is a good place
to store medicines.
|
6. Are there any side effects?
All medicines can have side effects. If they do occur, they are usually minor and
temporary. The most common side effects using BUDENOFALK foam are: Burning or pain
in the rectum, indigestion, irritable stomach (dyspepsia), increased risk of infection,
muscle and joint pain, muscle weakness, muscle twitching, brittle bones (osteoporosis),
mood changes, such as depression, irritability or euphoria.
For more information, including what to do if you have any side effects, see Section
6. Are there any side effects? in the full CMI.
Active ingredient: budesonide
Full Consumer Medicine Information (CMI)
This leaflet provides important information about using BUDENOFALK foam. You should also speak to your doctor or pharmacist if you would like further information
or if you have any concerns or questions about using BUDENOFALK foam.
Where to find information in this leaflet:
1. Why am I using BUDENOFALK foam?
BUDENOFALK foam contains the active ingredient, budesonide. Budesonide belongs to
a group of medications called corticosteroids.
BUDENOFALK foam is used to treat ulcerative colitis (inflammation) of the rectum (back
passage) and the lower part of the large bowel.
2. What should I know before I use BUDENOFALK foam?
Warnings
Do not use BUDENOFALK foam if:
you are allergic to budesonide, or any of the ingredients listed at the end of this
leaflet.
Always check the ingredients to make sure you can use this medicine.
Do not use this medicine if you suffer from a severe liver disease (liver cirrhosis).
Do not use this medicine after the expiry date printed on the pack or if the packaging
is torn or shows signs of tampering when it is dispensed to you.
Check with your doctor if you:
Tell your doctor if you have any other conditions and taking medicines
liver disease
lung disease (tuberculosis)
high blood pressure
diabetes, when the level of sugar in the blood is too high
disease which causes bones to become less dense, gradually making them weaker, more
brittle and likely to break (osteoporosis)
ulcer in stomach or duodenum
glaucoma (high pressure in the eye)
cataracts
family history of diabetes or glaucoma
any stresses
any other disease where use of corticosteroids may have unwanted effects.
you have an infection. The symptoms of some infections can be atypical or less pronounced.
have been exposed to chicken pox, measles or shingles infections. These illnesses
may become more severe when you take BUDENOFALK.
have not yet had measles.
need to be vaccinated, please speak to your doctor first.
are due to have an operation, please tell your surgeon and anaesthetist that you are
using BUDENOFALK foam.
have been treated with a systemically acting corticosteroid preparation (i.e. one
which is absorbed into the blood circulation and acting outside the gastrointestinal
tract) before starting treatment with BUDENOFALK foam, your symptoms may reappear
when the medicine is changed. If this happens, contact your doctor.
experience blurred vision or other visual disturbances.
There is insufficient information to recommend BUDENOFALK foam for use in children
or adolescents.
BUDENOFALK foam contains propylene glycol which may cause skin irritation in some
people.
BUDENOFALK foam also contains cetyl alcohol and cetostearyl alcohol (component of
emulsifying wax), which may cause local skin reactions (e.g. contact dermatitis).
During treatment, you may be at risk of developing certain side effects. It is important
you understand these risks and how to monitor for them. See additional information
under Section
6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Your doctor will discuss the risks and benefits of using BUDENOFALK foam if you are
pregnant or breastfeeding.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any
medicines, vitamins or supplements that you buy without a prescription from your pharmacy,
supermarket or health food shop.
Some medicines may interfere with BUDENOFALK foam and affect how it works.
cardiac glycosides such as digoxin, medicines used to treat heart conditions
diuretics, medicines used to treat excess fluid in your body
ketoconazole and itraconazole, medicines used to treat fungal infections
antibiotics such as clarithromycin and rifampicin, medicines used to treat infections
ritonavir and cobicistat medicines used for treating HIV infections
carbamazepine, medicine used for treating epilepsy
rifampicin, medicine used to treat tuberculosis
contraceptive pill
cholestyramine, medicine used to reduce cholesterol level
cimetidine, medicine to reduce stomach acid.
These medicines may be affected by BUDENOFALK foam or they may affect how well BUDENOFALK
foam works. You may need different doses of your medicines or you may need to take
different medicines. Your doctor or pharmacist will advise you if this is required.
Avoid drinking grapefruit juice while you are using BUDENOFALK foam as this can alter
its effects.
Your doctor or pharmacist have more information on medicines to be careful with or
to avoid while using BUDENOFALK foam.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins
or supplements you are using and if these affect BUDENOFALK foam.
4. How do I use BUDENOFALK foam?
How much to use
Follow all directions given to you by your doctor or pharmacist carefully.
They may differ from the information contained in this leaflet.
Adults and the elderly: Apply one actuation daily, corresponding to 2 mg budesonide. BUDENOFALK foam can
be used in the morning or evening.
One BUDENOFALK foam contains 14 doses (14 applications or "actuations") or 2 weeks
of dosing, based on a standard 2 mg/day dosing regimen.
In the product pack, the individual applicators are inserted into purpose-designed
plastic tray. To remove an applicator, push down firmly on the tray with one hand,
while pulling out the applicator with the other hand.
When to use
BUDENOFALK foam should be administered about the same time each day. Using it at the
same time each day will have the best effect. It will help you remember when to use
it.
How to use
Picture of the can
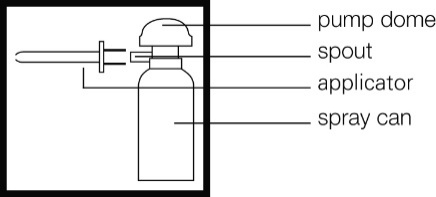
This medicine may only be used rectally, so it has to be inserted via the anus. Do
NOT take it by mouth.
If possible, go to the toilet and empty your bowels before using the foam.
1. Wash your hands thoroughly with soap and water.
2. For your comfort, ensure that BUDENOFALK foam should always be stored at room temperature
before use.
3. Push the applicator firmly onto the spout of the spray can.
4. Shake the can for 15 seconds to mix the contents.
5. Each time you use a new can, remove the safety tab from under the pump dome.
6. Twist the dome on top of the canister until the semi-circular gap underneath it is
in line with the applicator.
The can is now ready for use.
7. Place your forefinger on top of the pump dome. Turn the can upside down. Note that
the can will only work properly when held with the pump dome pointing down and as
vertically as possible.
8. Place one foot on a stool or chair. Alternatively, lie on your side with the lower
leg stretched out and the upper leg bent at an angle.
9. Insert the applicator into the rectum as far as possible. Push down the pump dome
fully once, hold and then release it very slowly. As soon as you release the pump
dome, the foam will be actuated from the can. Leave the applicator in this position
for 10 to 15 seconds before withdrawing it from your rectum to allow the full dose
to be released.
This will ensure that the entire dose is delivered to the rectum and will also reduce
the risk any irritation around your anus if the applicator is removed too quickly
while the foam is still being released.
10. After administering the foam remove the applicator from the spout of the can and dispose
of it as domestic waste in the plastic bag provided.
11. Wash your hands thoroughly and try not to empty your bowels again for as long as possible.
12. Always use a new applicator for each dose.
You may experience a little discomfort and a feeling of urgency to empty your bowels
immediately after foam insertion. This is normal and expected, especially when you
first start using BUDENOFALK foam due to the inflammation present within the bowel
related to your ulcerative colitis. Try to resist this urge to empty your bowels for
as long as possible. This feeling will subside over the next few days as treatment
continues and the inflammation decreases.
The following QR code links to a patient instructions for use video, which covers
the same dosing procedure detailed above:

How long to use
The duration of treatment will be decided by your doctor. In general, the inflammation
and associated symptoms should subside after 6 to 8 weeks.
BUDENOFALK helps control your condition but does not cure it. Therefore, you must
continue to use BUDENOFALK for as long as your doctor tells you to.
If you forget to use
If you missed a dose and it is almost time for your next dose, skip the dose you missed
and administer your next dose when you are meant to.
Otherwise, dose as soon as you can and then go back to taking your medicine at the
time in the day you would normally administer it.
Do not use a double dose to make up for the dose that you missed.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering when to use your medicine, ask your pharmacist for
some hints.
If you use too much
To date, no cases of overdosage with budesonide are known. In view of the properties
of budesonide contained in BUDENOFALK foam, an overdose resulting in toxic damage
is extremely unlikely.
For information on the management of overdose, contact the Poisons Information Centre
on 13 11 26 (Australia).
5. What should I know while using BUDENOFALK foam?
Things you should do
If you are about to start on any new medicine, remind your doctor and pharmacist that
you are also using BUDENOFALK foam.
Tell any other doctors, dentists, and pharmacists who treat you that you are using
this medicine.
If you are going to have surgery, tell the surgeon or anaesthetist that you are using
BUDENOFALK foam.
It may affect other medicines used during surgery.
If you become pregnant while using this medicine, tell your doctor immediately.
Things you should not do
Do not use BUDENOFALK foam to treat any other complaints unless your doctor tells
you to.
Do not give your medicine to anyone else, even if they have the same condition as
you.
Do not stop using BUDENOFALK foam or change the dosage without checking with your
doctor.
Driving or using machines
This medicine is not expected to affect your ability to drive a car or operate machinery.
Looking after your medicine
Keep BUDENOFALK foam in its original packaging until it is time to use it.
Keep your BUDENOFALK foam in a cool place where the temperature stays below 25°C.
Store the pack upright.
Do not refrigerate or freeze.
Protect from direct sunlight.
Keep away from flames or sparks. Contains flammable gas.
Use within 4 weeks of first opening.
Do not store BUDENOFALK foam or any other medicine in the bathroom or near a sink.
Do not leave it on a windowsill or in the car.
Heat and dampness can destroy some medicines.
Keep it where children cannot reach it.
A locked cupboard at least one-and-a-half metres above the ground is a good place
to store medicines.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy
for safe disposal.
Do not use this medicine after the expiry date.
Do not pierce or burn the can even when empty.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of
them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, speak to your doctor or pharmacist
if you have any further questions about side effects.
Less serious side effects
Serious side effect
The following side effects have been reported with medicines which are in the same
class as BUDENOFALK foam (corticosteroids). These side effects are typical for systematically
acting preparations (i.e. absorbed into the blood circulation and acting outside the
gastrointestinal tract). As BUDENOFALK foam acts locally, the risk of these class
effects occurring with this medicine is generally expected to be lower than with the
systemically acting corticosteroids.
Possible class effects of systemically acting corticosteroids are:
Cushing’s syndrome – e.g. with roundness of the face, weight gain, reduced glucose
tolerance, high blood sugar, high blood pressure, fluid retention in the tissues (e.g.
swollen legs), increased excretion of potassium (hypokalaemia), irregular periods
in women, unwanted body hair in women, impotence, abnormal laboratory findings (reduced
adrenal function), red stripes on the skin (stretch marks), acne.
increased risk of infection
mood changes such as depression, irritation or euphoria
Tiredness, malaise
blurred vision (e.g. glaucoma and cataract)
increased risk of blood clotting, disease of the blood vessels (associated with stopping
steroid use after long term therapy)
stomach complaints, gastric ulcers, pancreatitis and constipation
muscle pain and bone weakness (osteoporosis), loss of bone and cartilage (aseptic
bone necrosis)
rash from hypersensitivity reactions (allergic exanthema), formation of red stripes
(striations) and bleeding in the skin, delayed wound healing, local skin reactions
(such as contact dermatitis)
isolated cases: increased brain pressure with possible additional swelling of the
optic disk in adolescents.
Some of these unwanted effects were only reported after long-term use of oral budesonide.
These side effects are typical for systemic corticosteroids. Data shows that the frequency
of systemic adverse reactions is lower with BUDENOFALK foam due to its local action.
Tell your doctor or pharmacist if you notice anything else that may be making you
feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can
report side effects to the Therapeutic Goods Administration online at
www.tga.gov.au/reporting-problems . By reporting side effects, you can help provide more information on the safety of
this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop
using any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What BUDENOFALK foam contains
|
Active ingredient
(main ingredient)
|
Budesonide 2 mg
|
|
Other ingredients
(inactive ingredients)
|
emulsifying wax
cetyl alcohol
purified water
disodium edetate
steareth-10
propylene glycol
citric acid monohydrate, butane, isobutane and propane as propellants.
|
Do not use this medicine if you are allergic to any of these ingredients.
After administration of 14 spray actuations (14 daily doses), a residual amount of
foam will remain in the can. This is present in the can to ensure that 2 mg of budesonide
is contained in all 14 doses. Throw away the can with any residual contents after
application of the 14 daily doses.
What BUDENOFALK foam looks like
BUDENOFALK foam available in the following packs:
Two pressurised containers (14 daily doses each can), with 28 applicators and 28 plastic
bags.
Who distributes BUDENOFALK foam
Dr Falk Pharma Australia Pty Ltd,
9 Help Street
Chatswood, NSW 2067
Australian Registration Number: AUST R 179575
BUDENOFALK® is a registered trademark of Dr. Falk Pharma GmbH, Germany.
This leaflet was prepared in February 2025.
