2. What should I know before I use COSENTYX?
Do not use if you have ever had an allergic reaction to secukinumab or any of the
ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines,
or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section
2. What should I know before I use COSENTYX? in the full CMI.
3. What if I am taking other medicines?
4. How do I use COSENTYX?
Your healthcare provider will prescribe the dose of COSENTYX that is right for you.
Use COSENTYX exactly as prescribed.
Do not try to inject COSENTYX yourself until you or your caregiver has been shown
how by your healthcare provider.
If you have forgotten to inject a dose of COSENTYX, do not take a double dose to make
up for the dose you missed.
5. What should I know while using COSENTYX?
|
Things you should do
|
Discontinue treatment and tell your doctor or pharmacist immediately if you get any
signs or symptoms of a potentially serious infection or an allergic reaction during
treatment with COSENTYX.
Tell your doctor you are taking COSENTYX before a vaccination as live vaccines are
not suitable for you.
Tell your doctor if you have ever had hepatitis B
|
|
Things you should not do
|
Do not stop taking your medicine or change dosage without checking with your doctor
first.
Do not use this medicine if the liquid contains easily visible particles, is cloudy
or is distinctly brown.
Do not use if the outer box or blister seals are broken, or if it has been dropped
with the cap removed.
|
|
Driving or using machines
|
Be careful before you drive or use any machines or tools until you know how COSENTYX
affects you.
|
|
Looking after your medicine
|
Keep it in a refrigerator at 2°C to 8°C. Do not freeze it.
If necessary, COSENTYX can be left out of the refrigerator for a single period of
up to 4 days at room temperature, not above 30°C. Do not put back in the refrigerator
after it has reached room temperature. Discard any unused product.
|
6. Are there any side effects?
All medicines can have side effects although not everybody gets them. The most common
side effects of COSENTYX include cold symptoms, diarrhea, and upper respiratory infections.
COSENTYX may cause serious allergic reactions.
Tell your doctor or pharmacist as soon as possible if you do not feel well while you
are taking COSENTYX.
This medicine is subject to additional monitoring due to approval of an extension
of indications. This will allow quick identification of new safety information. You
can help by reporting any side effects you may get. You can report side effects to
your doctor, or directly at www.tga.gov.au/reporting-problems .
Active ingredient(s):
secukinumab
Full Consumer Medicine Information (CMI)
This leaflet provides important information about using COSENTYX. You should also speak to your doctor or pharmacist if you would like further information
or if you have any concerns or questions about using COSENTYX.
Where to find information in this leaflet:
1. Why am I using COSENTYX?
COSENTYX contains the active ingredient secukinumab. COSENTYX is a fully-human monoclonal antibody and belongs to a group of medicines
called interleukin (IL) inhibitors. Monoclonal antibodies are proteins that recognise
and attach specifically to certain proteins in the body. COSENTYX works by stopping
the activity of the IL-17A protein which is present at increased levels in the following
conditions:
Plaque psoriasis
Psoriatic arthritis
Axial spondyloarthritis, including ankylosing spondylitis (axial spondyloarthritis
with radiographic damage) and non-radiographic axial spondyloarthritis (axial spondyloarthritis
without radiographic damage)
Juvenile idiopathic arthritis (JIA), including Enthesitis Related Arthritis (ERA)
and Juvenile Psoriatic Arthritis (JPsA)
Hidradenitis suppurativa
Plaque psoriasis
COSENTYX is used to treat a skin condition called 'moderate to severe plaque psoriasis'
in patients 6 years and older.
Plaque psoriasis causes inflammation of the skin.
Psoriatic arthritis
COSENTYX is used to treat a condition called 'psoriatic arthritis'. The condition
is an inflammatory disease of the joints, often accompanied by psoriasis.
Axial spondyloarthritis, including ankylosing spondylitis (axial spondyloarthritis
with radiographic damage) and non-radiographic axial spondyloarthritis (axial spondyloarthritis
without radiographic damage)
COSENTYX is used to treat a condition called 'ankylosing spondylitis' and ‘non-radiographic
axial spondyloarthritis’ in adults. These conditions are inflammatory diseases primarily
affecting the spine which causes inflammation of the spinal joints.
Juvenile idiopathic arthritis, including Enthesitis Related (ERA) and Juvenile Psoriatic
Arthritis (JPsA)
These conditions are inflammatory diseases affecting the joints and the places where
tendons join the bone. In JPsA patients 2 years and older and ERA patients 4 years
and older, COSENTYX reduces the symptoms of these diseases and improves their ability
to do normal daily activities.
In patients with plaque psoriasis, psoriatic arthritis, and axial spondyloarthritis
the body produces increased amounts of a protein called IL-17A. This may lead to symptoms
such as itching, pain, scaling in psoriasis, swollen and tender joints in psoriatic
arthritis, and pain in the spine in axial spondyloarthritis.
Hidradenitis suppurativa
COSENTYX is used to treat a condition called hidradenitis suppurativa, also sometimes
called acne inversa or Verneuil’s disease. This condition is a chronic and painful
inflammatory skin disease. Symptoms may include tender nodules (lumps) and abscesses
(boils) that may leak pus. It commonly affects specific areas of the skin, such as
under the breasts, the armpits, inner thighs, groin and buttocks. Scarring may also
occur in affected areas. COSENTYX can reduce the number of nodules and abscesses you
have and may reduce the pain that is often associated with the disease.
COSENTYX is used in adults with hidradenitis suppurativa and can be used alone or
with antibiotics.
2. What should I know before I use COSENTYX?
Warnings
Do not use COSENTYX if:
you are allergic to secukinumab, or any of the ingredients listed at the end of this
leaflet.Always check the ingredients to make sure you can use this medicine. Some
of the symptoms of an allergic reaction may include:
shortness of breath, wheezing or difficulty breathing or swallowing
swelling of the face, lips, tongue or other parts of the body
rash, itching or hives on the skin
If you think you may be allergic, ask your doctor for advice before using COSENTYX.
Do not take COSENTYX if you have an active infection which your doctor thinks is important.
Do not take COSENTYX if there are visible signs of deterioration. If it has expired
or is damaged, return it to your pharmacist for disposal.
Check with your doctor if you:
are not sure whether you should start taking this medicine
have tuberculosis
have ever had hepatitis B
have ever been diagnosed with inflammatory bowel disease (Crohn's disease or ulcerative
colitis)
had a recent vaccination or if you will receive a live vaccine during treatment with
COSENTYX
ever had an allergic reaction to latex
Tell your doctor or pharmacist immediately if you get any of these symptoms during
treatment with Cosentyx:
If you develop itchy rash, dry skin, dry patches, inflamed skin, coin-shaped spots
on the skin [dermatitis (including eczema)].
If you develop severe inflammation of the entire skin surface (dermatitis exfoliative).
During treatment, you may be at risk of developing certain side effects. It is important
you understand these risks and how to monitor for them. See additional information
under Section
6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant. COSENTYX
is not recommended during pregnancy unless the benefits clearly outweigh the potential
risks.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Special populations
COSENTYX is not recommended for children under 6 years of age with plaque psoriasis
because it has not been studied in this age group.
Cosentyx is not recommended for children under 4 years of age with Enthesitis Related
Arthritis (ERA) and for children under 2 years of age with Juvenile Psoriatic Arthritis
(JPsA) categories of Juvenile Idiopathic Arthritis (JIA) because it has not been studied
in this age group.
COSENTYX is not recommended for children and adolescents (under 18 years of age) in
other indications because it has not been studied in this age group.
COSENTYX may be used by people aged 65 years and over.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any
medicines, vitamins or supplements that you buy without a prescription from your pharmacy,
supermarket or health food shop.
Tell your doctor or pharmacist if you:
are taking a blood thinning medicine called warfarin.
are taking, have recently taken or might take any other medicines.
have recently had or are going to have a vaccination. You should not receive certain
types of vaccines (live vaccines) while using COSENTYX. It is recommended that children
and adolescents receive all age-appropriate vaccines according to current guidelines
before using COSENTYX.
Such medicines may interfere with COSENTYX and affect how it works. You may need different amounts of your medicines, or you may need to take different
medicines.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins
or supplements you are taking and if these affect COSENTYX. Your doctor and pharmacist have more information on medicines to be careful with
or avoid while taking this medicine.
4. How do I use COSENTYX?
How much to give / use
Your doctor will decide how much COSENTYX you need.
Plaque psoriasis
In adults, the recommended dose is 300 mg by subcutaneous injection with initial loading
dose at Weeks 0, 1, 2, 3, and 4 followed by the same dose every month. If you weigh
90 kgs or higher, further adjustments to your dose may be recommended by your doctor.
Each 300 mg dose is given as one subcutaneous injection of 300 mg or as two sub-cutaneous
injections of 150 mg.
In children 6 years and older, the recommended dose is based on body weight and is
given by subcutaneous injection with initial dosing at Weeks 0, 1, 2, 3, and 4 followed
by the same dose every month.
For children receiving the 75mg dose, the 75mg/0.5mL pre-filled syringe should be
used.
Psoriatic arthritis
The recommended dose is 150 mg by subcutaneous injection with initial dosing at Weeks
0, 1, 2, 3, and 4 followed by the same dose every month. Based on your response, your
doctor may increase the dose to 300 mg.
For psoriatic arthritis patients who also have moderate to severe plaque psoriasis,
your doctor may adjust the dose recommendation as needed.
For patients who did not respond well to medicines called tumour necrosis factor (TNF),
the recommended dose is 300 mg by subcutaneous injection with initial dosing at Weeks
0, 1, 2, 3, and 4 followed by the same dose every month. Each 300 mg dose is given
as one subcutaneous injection of 300 mg or as two subcutaneous injections of 150 mg.
COSENTYX may be administered with or without methotrexate.
Axial Spondyloarthritis
Ankylosing spondylitis
The recommended dose is 150 mg by subcutaneous injection with initial dosing at Weeks
0, 1, 2, 3, and 4 followed by the same dose every month. Based on your response, your
doctor may increase the dose to 300 mg. Each 300 mg dose is given as one subcutaneous
injection of 300 mg or as two subcutaneous injections of 150 mg.
Non-radiographic axial spondyloarthritis
With a loading dose: The recommended dose is 150 mg by subcutaneous injection with
initial dosing at Weeks 0, 1, 2, 3, and 4 followed by the same dose every month.
Without a loading dose: The recommended dose is 150 mg by subcutaneous injection every
month.
Juvenile idiopathic arthritis
Enthesitis Related Arthritis (ERA) in children 4 years and older
The recommended dose is based on body weight and is given by subcutaneous injection
with initial dosing at Weeks 0, 1, 2, 3, and 4 followed by the same dose every month.
For patients weighing < 50 kg the dose is 75 mg. For patients weighing ≥ 50 kg the
dose is 150 mg.
Juvenile Psoriatic Arthritis (JPsA) in children 2 years and older
The recommended dose is based on body weight and is given by subcutaneous injection
with initial dosing at Weeks 0, 1, 2, 3, and 4 followed by the same dose every month.
For patients weighing < 50 kg the dose is 75 mg. For patients weighing ≥ 50 kg the
dose is 150 mg.
Hidradenitis Suppurativa
The recommended dose is 300 mg by subcutaneous injection with initial dosing at Weeks
0, 1, 2, 3, and 4 followed by the same dose every month. Further adjustments to your
dose may be recommended by your doctor. Each 300 mg dose is given as one subcutaneous
injection of 300 mg or as two subcutaneous injections of 150 mg.
Do not exceed the recommended dose.
Follow the instructions provided and use COSENTYX until your doctor tells you to stop.
How to give COSENTYX
Follow all directions given to you by your doctor, nurse or pharmacist carefully.
They may differ from the information contained in this leaflet.
Always use COSENTYX as your doctor has told you. You should check with your doctor,
nurse or pharmacist if you are not sure.
COSENTYX is intended for subcutaneous use. This means that it is injected into the
fatty tissue just under the skin.
The injection may be given by your doctor or nurse or caregiver or you may be taught
how to inject yourself with the medicine.
You and your doctor should decide if you should inject COSENTYX yourself.
After proper training in subcutaneous injection technique, patients ≥ 12 years old
may self-inject COSENTYX or, for all patients ≥ 6 years old, the injection may be
given by a caregiver if your doctor determines that it is appropriate.
It is important not to try to inject yourself until you have been trained by your
doctor, nurse or pharmacist. A caregiver may also give you your COSENTYX injection
after proper training.
If you do not understand the instructions on the label ask your doctor or pharmacist
for help.
Choosing the injection sites
The injection sites are where the skin will be pierced to administer the subcutaneous
injection.
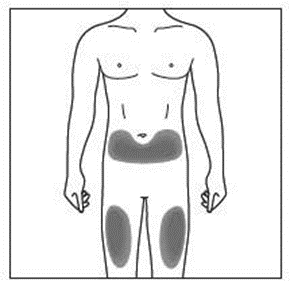
The recommended site is the front of your thighs. You may also use the lower abdomen,
but not the area five centimetres around the navel (belly button). Choose a different site
each time you give yourself an injection.
If a caregiver is giving you the injection, the outer upper arms may also be used
(Shown in grey in the diagram for illustrative purposes).
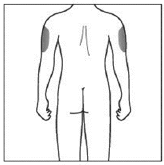
Do not inject into areas where the skin is tender, bruised, red, scaly or hard. Avoid
areas with scars or stretch marks

Read the Instructions for Use on how to use COSENTYX all the way through before removing
COSENTYX from the refrigerator. These instructions are to help you to inject correctly.
These instructions are also available via the following hyperlinks:
How long to use COSENTYX
This is a long-term treatment. Keep using this medicine for as long as your doctor
tells you.
Your doctor will regularly monitor your condition to check that the treatment is having
the desired effect.
If you forget to use COSENTYX
If you have forgotten to inject a dose of COSENTYX, inject the next dose as soon as
you remember. Then talk to your doctor to discuss when you should inject the next
dose.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering to have your medicine, ask your pharmacist for some
hints.
Do not take a double dose to make up for the dose you missed.
If you use too much COSENTYX
If you think that you have used too much COSENTYX, you may need urgent medical attention.
You should immediately:
phone the Poisons Information Centre
(by calling 13 11 26), or
contact your doctor, or
go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using COSENTYX?
Things you should do
Discontinue treatment and tell your doctor or pharmacist immediately if you get any
of these symptoms during treatment with COSENTYX.
Call your doctor straight away if you have:
Signs or symptoms of a potentially serious infection. These may include:
fever, flu-like symptoms, night sweats
feeling tired or short of breath; cough which will not go away
warm, red and painful skin, or a painful skin rash with blisters
burning when passing urine
Signs or symptoms of an allergic reaction. These may include:
difficulty breathing or swallowing
low blood pressure, which can cause dizziness or light-headedness
swelling of the face, lips, mouth or throat
severe itching of the skin, with a red rash or raised bumps
Keep all of your doctor's appointments so that your progress can be checked. Your doctor will do tests from time to time to make sure the medicine is working
and to prevent unwanted side effects. Your doctor will decide if and when you may
restart the treatment.
If you need to be vaccinated, tell your doctor you are taking COSENTYX before you
have the vaccination. Live vaccines will not be suitable for you.
If you are about to be started on any new medicine, remind your doctor and pharmacist
that you are taking COSENTYX.
Remind any doctor, dentist or pharmacist you visit that you are using COSENTYX.
Things you should not do
Do not stop taking your medicine or change dosage without checking with your doctor
first.
Never leave the prefilled pen lying around where others might tamper with it.
Do not open the sealed box until you are ready to use COSENTYX.
Do not use this medicine if the liquid contains easily visible particles, is cloudy
or is distinctly brown.
Do not use the prefilled pen if either the seal on the outer box or the seal of the
blister are broken. It may not be safe for you to use.
Do not shake the prefilled pen.
Do not use the pen if it has been dropped with the cap removed.
Do not take it to treat any other complaints unless your doctor tells you to.
Do not give this medicine to anyone else, even if they have the same condition as
you.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how COSENTYX
affects you.
No studies on the effects on the ability to drive and use of machines have been performed
on COSENTYX.
Looking after your medicine
Keep it in a refrigerator at 2°C to 8°C. Do not freeze it.
Store it in the carton in order to protect it from light. Follow the instructions
in the carton on how to take care of your medicine properly.
If necessary, COSENTYX can be left out of the refrigerator for a single period of
up to 4 days at room temperature, not above 30°C.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do
not store it:
in the bathroom or near a sink, or
in the car or on window sills.
A locked section of the refrigerator, at least one-and-a-half metres above the ground,
is a good place to store COSENTYX.
Keep it where young children cannot reach it.
When to discard your medicine
Do not put back in the refrigerator after it has reached room temperature. Discard
any unused product.
Dispose of the used COSENTYX prefilled pens immediately after use in a sharps container.
The COSENTYX prefilled pen should never be re-used.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy
for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects although not everybody gets them. If you do experience
any side effects, most of them are minor and temporary. However, some side effects
may need medical attention.
Tell your doctor or pharmacist as soon as possible if you do not feel well while you
are taking COSENTYX.
See the information below and, if you need to, ask your doctor or pharmacist if you
have any further questions about side effects.
Less serious side effects
|
Less serious side effects
|
What to do
|
|
Infection and infestation related such as sore throat, stuffy nose, oral thrush, cold
sores, infection of the external ear, discharge from the eye with itching, redness
and swelling, and athlete's foot
Allergic reactions related such as runny nose, hives, headache, nausea, and itchy
rash
Small, itchy blisters on the palms of hands, soles of feet and edges of the fingers
and toes (dyshidrotic eczema)
inflammation of small blood vessels, which can lead to a skin rash with small red
or purple bumps (vasculitis)
Diarrhoea
Painful period
Nausea, diarrhea, vomiting, abdominal pain and fever (symptoms of inflammatory bowel
disease)
|
Speak to your doctor if you have any of these less serious side effects and they worry
you.
|
Serious side effects
|
Serious side effects
|
What to do
|
|
Severe allergic reaction with shock (anaphylactic shock)
Signs of low levels of white blood cells such as fever, sore throat or mouth ulcers
due to infections (neutropenia)
Symptoms of inflammatory bowel disease such as nausea, diarrhoea, vomiting, abdominal
pain and fever. New cases of inflammatory bowel disease or "flare-ups" can happen with COSENTYX, and
can sometimes be serious. If you have inflammatory bowel disease (ulcerative colitis or Crohn's disease), tell
your doctor if you have worsening symptoms during treatment with COSENTYX or develop
new symptoms of stomach pain or diarrhoea.
Severe inflammation of the entire skin surface (dermatitis exfoliative)
Itchy rash, dry skin, dry patches, inflamed skin, coin-shaped spots on the skin [dermatitis
(including eczema)]
Swelling of the face, lips, tongue or throat (angioedema)
|
Call your doctor straight away, or go straight to the Emergency Department at your
nearest hospital if you notice any of these serious side effects.
|
The frequency of some side effects cannot be estimated from available data
Fungal infections of the skin and mucous membranes (thrush)
Inflammation of small blood vessels, which can lead to a skin rash with small red
or purple bumps (hypersensitivity vasculitis)
Painful swelling and skin ulceration (pyoderma gangrenosum)
COSENTYX may affect the results of some blood tests, including:
slight increase in blood cholesterol and blood fat (triglycerides)
elevated liver enzymes
In clinical trials, major adverse cardiovascular events were rarely observed with
COSENTYX.
Ask your doctor or pharmacist to answer any questions you may have. Tell your doctor
or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can
report side effects to the Therapeutic Goods Administration online at
www.tga.gov.au/reporting-problems . By reporting side effects, you can help provide more information on the safety of
this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop
taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What COSENTYX contains
|
Active ingredient
|
secukinumab
|
|
Other ingredients
|
trehalose dihydrate
histidine / histidine hydrochloride monohydrate
methionine
polysorbate 80
water for injection
|
|
Potential allergens
|
This medicine does not contain sucrose, lactose, gluten, tartrazine or any other azo
dyes.
|
Do not take this medicine if you are allergic to any of these ingredients.
What COSENTYX looks like
COSENTYX solution for injection is a clear liquid. Its colour may vary from colourless
to slightly yellow.
COSENTYX 75 mg/0.5 mL solution for injection is available in packs containing 1 single-use
pre-filled syringe (AUST R 353254).
COSENTYX 150 mg/1 mL solution for injection is available in packs containing 1 or
2 single-use pre-filled pens (AUST R 218800).
COSENTYX 300 mg/2 mL solution for injection is available in packs containing 1 single-use
pre-filled pen (AUST R 353266).
Who distributes COSENTYX
Novartis Pharmaceuticals Australia Pty Limited
ABN 18 004 244 160
54 Waterloo Road
Macquarie Park NSW 2113
Telephone 1 800 671 203
® = Registered Trademark
This leaflet was prepared in March 2025.
(cos050325c based on PI cos050325i)