Introduction
Advantages of contraceptive implants
Disadvantages of contraceptive implants
Minor side effects
Following insertion
Major complications of contraceptive implants
References
Millions of women have used the contraceptive implant, but its users' opinions on its advantages and adverse effects vary. In this article, we will investigate these advantages and disadvantages in detail.
Image Credit: WiP-Studio/Shutterstock.com
Advantages of contraceptive implants
The advantages of contraceptive implants include:
- High effectiveness of up to 99 percent within seven days of implant insertion
- Very inexpensive method of long-term contraception, comparable to intrauterine devices
- Safe in the majority of women
- Efficacy for three years without further intervention
- Easily removed in most cases to allow pregnancy to occur naturally
- Independent of user memory or schedule and sexual intercourse, as it provides continuous contraception
- Provides anonymity of use and is convenient to adopt
- Safe for use during lactation
- Relieves menorrhagia and dysmenorrhea in many cases
- Amenorrhea in one of every five women on the implant, which is often perceived to be a benefit
- Reduces the risk of pelvic inflammatory disease due to the thick cervical mucus, which prevents microbes from ascending from the vagina into the uterus
- Suitable in conditions that preclude the use of combined hormonal contraception
- Quick return of fertility within 21 days of implant removal
- Some women experience improvement in acne following the use of the implant
- Some protection against endometrial cancers
Disadvantages of contraceptive implants
Disadvantages of the contraceptive implant include:
- It offers no protection against sexually transmitted infections (STIs)
- Interaction with anticonvulsants, certain antibiotics, or St. John's wort may occur
- Immediate protection may not be available in all situations, and another mode of effective contraception must then be used for at least seven days following insertion
Minor side effects
These include:
1. Disruption of normal menstruation
The most common and troublesome adverse effects of contraceptive implants are alterations in the regular menstrual pattern. About 10 percent of women discontinue this method for this reason. Such changes have their onset within the first three months of implant insertion and indicate the future course of bleeding for the individual patient. For this reason, women who receive the implant should be warned what to expect and advised to come in for evaluation if abnormal bleeding patterns develop to rule out ectopic gestation, pregnancy, or disease conditions. Changes include:
- Amenorrhea
- Light or irregular bleeding
- Frequent bleeding episodes (more than five in 90 days)
- Continuous bleeding for weeks (more than 14 days)
- Occasionally menorrhagia
2. Acne in over 13 percent
3. Breast pain in 13 percent, as well as enlargement of the breast, nipple discharge, and pruritus vulvae
4. Abdominal pain 11 percent
5. Pharyngitis in 11 percent
6. Leukorrhea in 10 percent
Following insertion
- 5 percent of women may experience bruising and local irritation or pain, fibrosis, paresthesia, infection at the site, or scar formation
- Weight gain: 15 percent of women with the implant gain significant weight, averaging approximately 3 pounds after one year and almost 4 pounds after two years. The removal rate for this indication is less than 3 percent
- Vaginal dryness: Almost 15 percent of women using the implant complained of vaginal dryness
- Headaches occur in a quarter of all women with the implant
- Influenza-like symptoms, dizziness, dysmenorrhea, back pain, emotional instability, nausea, mood swings, and hypersensitivity reactions have also been reported
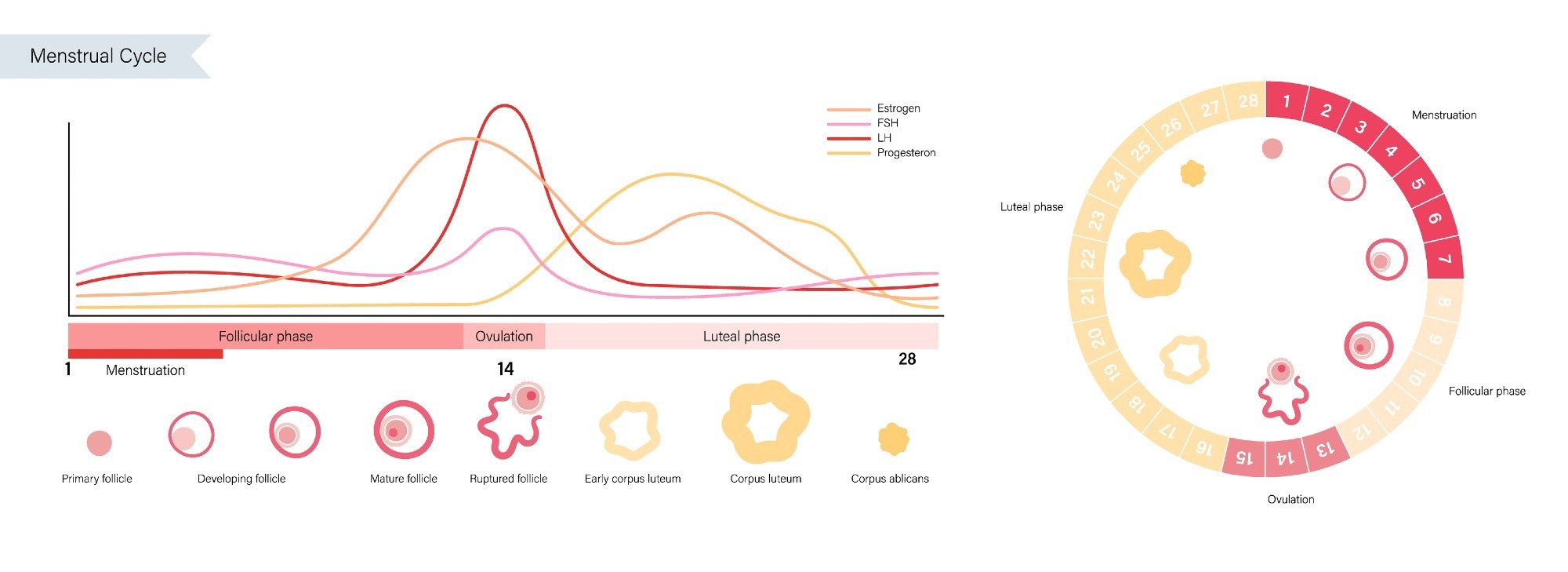
Image Credit:
Major complications of contraceptive implants
Insertion complications
As the implant is inserted in the arm, the implant can migrate from the insertion site. This is sometimes the result of implanting it deeper than the subcutaneous plane.
Migration
Migration may occur in the blood vessels of the arm, and some reports indicate migration into the pulmonary artery. In such cases, inadvertent intravascular insertion may have occurred. Such cases may require deep surgery and endovascular removal.
Hypertension
Hormonal contraception is contraindicated in women who have a history of hypertension, especially with complications, but progestin-only implants may be used if the hypertension is well controlled and under close monitoring. Removal is mandated if sustained or uncontrolled rises in blood pressure occur during the implant period.
Non-palpable implants
These are not ideal and require to be located as soon as possible by imaging. Early removal should be scheduled once the location is known - exploration without knowing where the implant can be dangerous and should never be attempted. Failure to remove a misplaced implant may result in prolonged subfertility, ectopic gestation, or adverse events related to progestin administration.
Anaphylactic reactions
Known to occur, including angioedema of the skin and subcutaneous tissue.
Ectopic pregnancies
Often occur if women conceive while the implant is in situ (which is extremely rare).
Vascular events
Combined oral contraceptives have been associated with a higher risk of vascular thrombosis and infarctions, including retinal artery thrombosis. However, contraceptive implants contain only progestin. It is currently unknown whether they carry the same increased risk, but arterial or venous thromboses and venous thromboembolism have been reported, including pulmonary, deep leg vein, myocardial, and cerebrovascular arterial or venous thrombotic events.
A history of such events is a contraindication to using contraceptive implants. Their occurrence necessitates immediate removal.
Use in the postpartum period
It is recommended that the implant be inserted 21 days after childbirth at the earliest to reduce the risk of thromboembolism associated with pregnancy and delivery.
Ovarian cysts
The prolonged release of low-dose progestin normally suppresses follicular development in the ovaries. However, if a follicle escapes this inhibition, it may continue to grow till it is beyond the size of a normal mature follicle. This may form a follicular cyst. While they typically disappear on their own, surgical removal is rarely indicated.
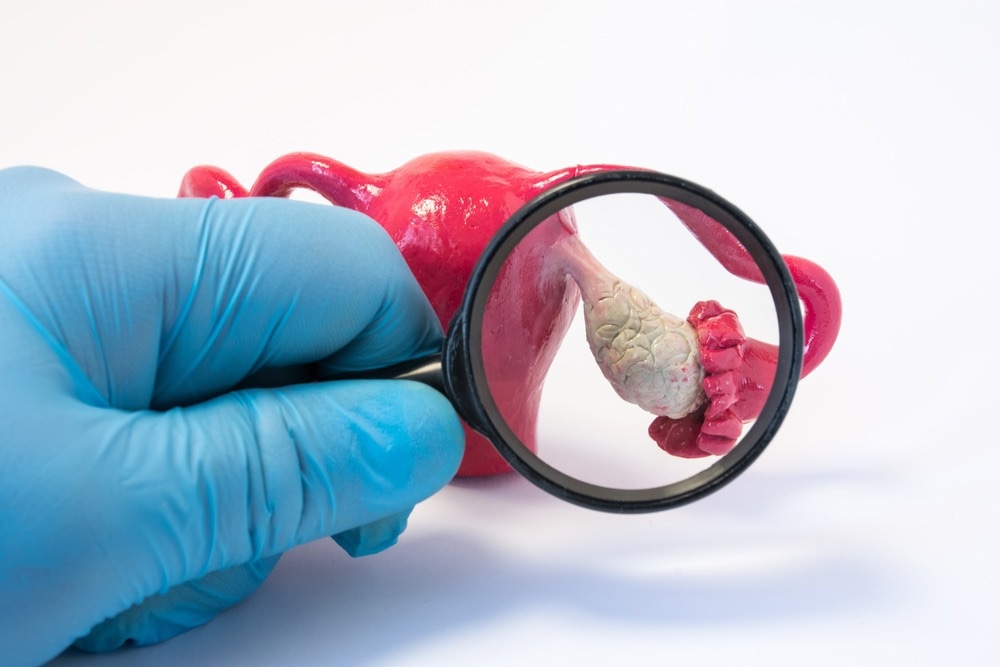
Image Credit: Shidlovski/Shutterstock.com
Reproductive organ cancers
Hormonal contraception is contraindicated in women with a history of breast cancer because of the known sensitivity of these tumors to sex hormones. While progestin-only implants are unlikely to produce the same kind of cancerous change, women with implants with a family history of breast growth or cancers should be closely followed up.
Liver disease
Liver function tests should be repeated if they are initially abnormal. The occurrence of jaundice usually indicates the removal of the implant. Hepatic adenomas may occur with combined oral contraceptives, but the risk with progestin-only implants is unknown. Conversely, liver damage may prolong the time of active progestin release from the implant, and therefore the presence of active hepatitis or liver carcinoma is an absolute contraindication for the insertion of the implant.
Gallbladder disease
The risk of cholecystitis or gallstones following hormonal contraception is established, though small, but similar risks among progestin-only implant users have not yet been shown or disproved.
Effects on carbohydrate and lipid metabolism
Progestin-only implants can cause insulin resistance, resulting in mild hyperglycemia, though their significance is unknown. For this reason, prediabetic or diabetic women using the implant should be followed carefully. It also causes hyperlipidemia in some cases.
Depressed mood
Women with a history of low mood should be adequately assessed before the use of this method, and its removal is seriously advised if depression occurs.
Rapid resumption of ovulation
The decline in progestin levels to undetectable levels occurs within a week of removal, and pregnancies have been reported to occur within one to two weeks of removal. Therefore, other contraceptive measures should be adopted immediately if the woman does not want to conceive.
Fluid retention
The use of hormonal contraceptives is associated with fluid retention to some extent, and therefore, they may not be suitable for use in women with heart or renal conditions, which could become worse with increased fluid retention. Careful monitoring is required in such situations.
Broken or bent implant
Complicated insertion or removal may result in the presence of a fragment or bent rod, which is associated with a somewhat higher rate of progestin release.
Nervous system and psychological disorders
These links to progestin implants are rare but may include seizures, migraines, sleepiness, loss of libido, and anxiety.
References:
- Reproductive Health | HHS Office of Population Affairs (2022). Available at: https://opa.hhs.gov/reproductive-health?pregnancy-prevention/hormonal-methods/implant/index.html (Accessed: 16 August 2022).
- Women's sexual and reproductive health - Better Health Channel (2022). Available at: https://www.betterhealth.vic.gov.au/campaigns/womens-sexual-and-reproductive-health (Accessed: 16 August 2022).
Further Reading
Last Updated: Sep 11, 2023