By Jeyashree Sundaram, MBA
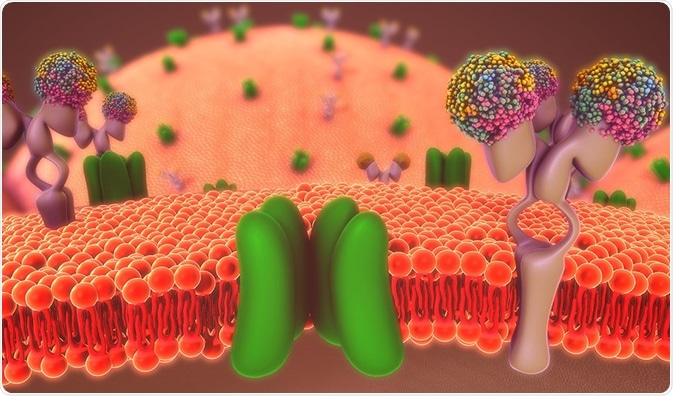
Phosphatidylinositol 4,5-bisphosphate (PIP2) is an essential lipid involved in metabolic processes. It is integral to the cell membrane of all animal and plant cells and acts as a second messenger in a variety of signaling pathways.
Credit: sciencepics/Shutterstock
PIP2 is an important phosphoinositide that is present in around 1% of the plasma membrane. It is also involved in signal transduction of intracellular and extracellular components.
There are two important types of receptors whose availability is identified and transduced. They are the G protein coupled receptors (GPCRs) and receptor tyrosine kinases. These two receptors activate the isoforms of phospholipase C through various mechanisms.
Intracellular mediators for PIP2 pathway
One of the most essential pathways of intracellular transduction involves the PIP2 molecule.
The muscarinic receptor is a G-protein-coupled receptor that stimulates an enzyme known as phospholipase C. Hydrolysis of PIP2 by phospholipase C produces intracellular mediators such as IP3 (inositol triphosphate) and DAG (diacylglycerol).
These intracellular enzymes perform as downstream signaling components that generate and amplify the signals originated from the binding of ligand molecules. The targets of this intracellular signaling pathway include transcription factors that help in the control of gene expression.
Stimulation by extracellular signals
Acetylcholine is an extracellular chemical signal that performs physiological actions such as opening of ligand-gated ion channels, synaptic signaling molecule, and for stimulation of GPCR.
An individual cell contains various type of receptors, for example, the acetylcholine receptors present in the skeletal muscle known as nicotinic receptors and muscarinic receptors of cardiac muscles.
G protein coupled receptors
The functions of G protein in PIP2-mediated signal transduction take place in three stages: (i) identification of G proteins, (ii) comparing the detected G proteins with their thoroughly distinguished equivalents in animals, and (iii) analysis of G protein involvement in the turnover of inositol phospholipid.
GPCRs are the seven membrane-spanning domains whose cytoplasmic loops and tails bind the heterotrimeric G protein complexes.
Through ligand binding, guanosine diphosphate bound by an alpha subunit is interchanged for guanosine triphosphate, causing the G-protein complex to disintegrate.
The alpha and gamma subunits are comprised of covalently bound fatty acids that are covalently bound that keep them secured with the membrane.
Upon ligand binding and segregation, the alpha subunit is no longer connected to the membrane and can now bind a various number of partners inside the cytoplasm.
After binding and stimulation by downstream activators, the beta and gamma subunits remain together and form a signal. A set of regulatory proteins that bind the cytoplasm of the GPCR known as arrestins suppress the GPCR pathway.
Activation of phospholipase C
Phospholipids serve as a source for signaling molecules after being split into four lipases, namely, phospholipase A, phospholipase B, phospholipase C, and phospholipase D.
Various forms of phospholipase enzymes with different substrates are found in eukaryotes to generate a wide range of extracellular and intracellular signaling molecules.
The general role of phospholipase C is to split PIP2, to generate DAG and IP3.
- IP3 and its control of Ca2+
Inositol triphosphate is a second messenger thathelps in transmission of chemical signals such as growth factors, neurotransmitters, hypertrophic stimuli called Angiotensin-II, and hormones for the transduction networks of the cell.
The basic structure of the IP3 receptor is comprised of three domains: an IP3 binding domain near the amino terminus, a coupling domain in the center of the molecule, and a transmembrane domain near the carboxyl terminus.
IP3 is a negatively charged water-soluble molecule that can rapidly diffuse into cytosol to bind with IP3 receptor; it is opened to release Ca2+ out of endoplasmic reticulum.
The production of IP3 is therefore capable of coupling the activated receptor in the plasma membrane to the Ca2+ ions released from an intracellular store.
Various cellular responses can depend on this pathway, which includes contraction of smooth muscle and secretion of enzymes by the acinar cells of the pancreas.
Additionally, calcium ions are capable of initiating or inhibiting signaling pathways in the cells. Hence, stimulation of IP3 signaling cascade controls the enzymatic activity within eukaryotic cells.
Hydrolysis of PIP2 produces a hydrophobic molecule known as diglycerides or diacylglycerol (DAG). After IP3 is formed, DAG is retained in the cell membrane. The DAG pathway is a message generating pathway that is involved in the activation of enzymes and in turn produces various biological events, including transcription of DNA.
Similar to other lipids, DAG also diffuses through the membrane surface where it can interact with some other enzyme called protein kinase C and hence activating them.
There is a wide range of transmembrane receptors that transduce extracellular signals through the activation of Bruton's tyrosine kinase. This mechanism activates phospholipase C-gamma 2 (PLCg2) that results in the generation of DAG and IP3.
Ligation taking place outside the cell can activate the receptors, leading to phosphorylation and cross phosphorylation of PLCg2, which is then active.
The metabolism of DAG can form arachidonic acid and 2-arachidonyl glycerol, an endogenous agonist for the cannabinoid receptors of the central nervous system.
Calcium and IP3 in Signaling Pathways
Credit: tacitserendipity/Shutterstock
Further Reading
Last Updated: Aug 24, 2023