PLEASE NOTE:
THIS ARTICLE CONTAINS GRAPHIC IMAGES OF PATIENT WOUNDS.

What is ‘flesh-eating’ disease and how many people does it affect?
Untreated, the mortality probably exceeds 70%. Flesh-eating is a description of Toxic Inflammatory Cellulitis which usually starts by bacterial subcutaneous infection followed by freezing in phase II which is the normal inflammatory response phase of wound healing.
The natural process of wound healing is:
- Phase I is wounding
- Phase II is inflammatory response
- Phase III is regeneration of cellular healing
- Phase IV is remodeling
If frozen in phase II, the body’s cellular toxins break down tissue and the destruction continues until intervention occurs. We have learned that this can be changed by debridement and, as we tried recently, with irrigation with NeutroPhase®. This product, was developed by NovaBay Pharmaceuticals. NovaBay has shown in the laboratory studies that NeutroPhase directly neutralizes tested toxins. Usually skin grafting is necessary in large wounds with tissue loss. If caught early, small incisions for debridement followed by instillation of NeutroPhase and the wound vac can halt the inflammatory process and not allow progression to the next phase.
How serious is the condition if left untreated?
When adequately and timely treated we have had neither fatalities nor amputations. This includes antibiotics, debridement and Negative Pressure Wound therapy. Toxic Shock is another symptom that in the past has had a mortality of 50%. With the treatment I’ve described in our most recent publication in the journal wounds, we have had 2 patients who survived Toxic Shock with no residual impact.
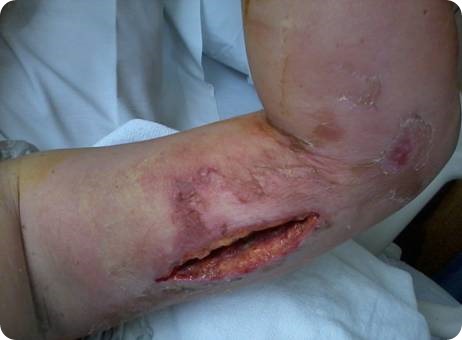
What treatments are currently available for ‘flesh-eating’ disease?
The current standard treatment has been antibiotics and debridement of the necrotic tissue as well as symptomatic therapy for shock. Based on experience, I now treat patients as I described here.
Please can you outline the novel anti-microbial product which you have been investigating? How was this developed?
The first case treated with Negative Pressure Wound Therapy and NeutroPhase after surgical debridement was noted to work after all bacterial infection was gone. As I mentioned, NovaBay performed some laboratory testing to explore why NeutroPhase seemed to have helped this patient who still had toxic inflammatory cellulitis after the bacterial infection was cleared. Novabay’s laboratory studies showed neutralization in vitro of several of the isolated toxins after exposure to NeutroPhase. This is what the recent paper in the medical journal Wounds describes.
By what mechanism is this thought to work?
In the laboratory, NeutroPhase was shown to directly neutralize the bacterial toxins, which in vivo means their inflammatory potential was eliminated/minimized.
What stage of development is the product currently at and what further research is required?
NovaBay has obtained 510(k)-clearance by the FDA for NeutroPhase as a wound cleanser. The product is on the market and is available via prescription. Regarding using this cleanser as a treatment for Toxic Inflammatory Cellulitis, it would be useful if it could be tested by other medical centres as well.
%20resized_thumb.jpg)
How do you think ‘flesh-eating’ disease treatments will develop in the future?
I am hopeful more cases can be treated with the regimen as described here and in the example we published in Wounds. Early recognition of Toxic Inflammatory Cellulitis (Necrotizing fasciitis or flesh eating bacteria or gas gangrene) and the use of this treatment regimen could dramatically change the standard of care.
Where can readers find more information?
National Necrotizing Fasciitis Foundation in New York or literature search for Necrotizing Fasciitis is the most rapid way.
About Dr. John Crew
 John R Crew, MD began practicing vascular surgery in San Francisco in 1964 and has resided in San Francisco since that time. He has done all major vascular surgical procedures, peripheral angioplasty, transmyocardial laser revascularization, made retractors in his garage, helped to develop numerous devices, products, procedures and techniques. He started a noninvasive vascular laboratory when everyone thought it foolish. He published the first paper on carotid surgery without angiography, considered unthinkable then but is now an acceptable way to proceed. He is constantly on the lookout for ways to improve patient care. He had a vision for a Wound Care Center and pushed to make it a reality. The clinic is now busy 5 days a week. Dr Crew sings at hospital Christmas parties, gives lectures to education, and even writes songs about wounds. He always has a smile, does not rant, does not swear, does not raise his voice, is kind to everyone he passes, creates solutions to problems, has the gift of teaching and makes learning enjoyable. He can grasp a concept, mold it, improve on it, make it work, and pass it on to others. He is a team player from the housekeeping staff, to nurses, physicians, and CEOs.
John R Crew, MD began practicing vascular surgery in San Francisco in 1964 and has resided in San Francisco since that time. He has done all major vascular surgical procedures, peripheral angioplasty, transmyocardial laser revascularization, made retractors in his garage, helped to develop numerous devices, products, procedures and techniques. He started a noninvasive vascular laboratory when everyone thought it foolish. He published the first paper on carotid surgery without angiography, considered unthinkable then but is now an acceptable way to proceed. He is constantly on the lookout for ways to improve patient care. He had a vision for a Wound Care Center and pushed to make it a reality. The clinic is now busy 5 days a week. Dr Crew sings at hospital Christmas parties, gives lectures to education, and even writes songs about wounds. He always has a smile, does not rant, does not swear, does not raise his voice, is kind to everyone he passes, creates solutions to problems, has the gift of teaching and makes learning enjoyable. He can grasp a concept, mold it, improve on it, make it work, and pass it on to others. He is a team player from the housekeeping staff, to nurses, physicians, and CEOs.
He spends time with patients and families, giving them his wisdom, expertise, warmth and spirit. He will celebrate, laugh, cry, and pray with patients, families and friends. He is able to touch, physically, emotionally, and spiritually.
Dr Crew has served almost 50 years and retirement is closing in but not just yet. He is a man of great faith and great love of God. He sang bass in a Christian quartet for 50 years. He has 3 children and 7 grandchildren. He has been married to his college sweetheart for almost 60 years. He loves his work and it shows.