Phase-contrast X-ray imaging differs from conventional X-rays because it uses a different physical principle.
X-rays are effectively electromagnetic waves of very high frequencies. Waves do a number of different things when they go through matter and we’re interested in the different speeds they take when traveling through different materials.
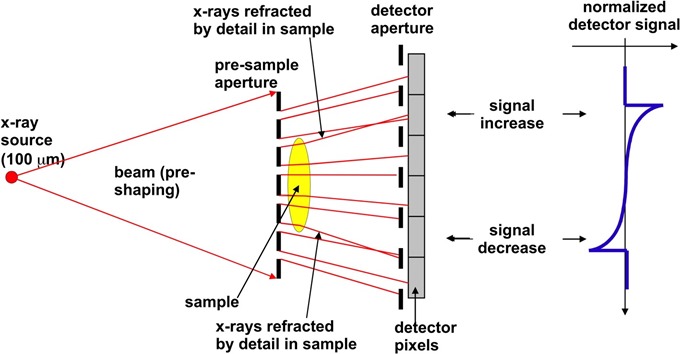
Credit: Advanced X-Ray Imaging (AXIm) Group, UCL
If you have a different speed when you travel through a different material, or different tissue type, then this leads to a phase shift. This is when the waveform gets distorted. It's difficult to pick up these mechanisms, but there is one manifestation called refraction.
When the waveform gets distorted, locally, you have a small deviation in the direction of X-rays, which is something that you might be able to pick up with the right sort of set-up. This deviation is effectively x-ray refraction, in a similar way to what happens with visible light.
A classic example of visible light refraction is the straw that you see bent in a glass of water. The same thing happens to X-rays, only at a much smaller angle. We're talking about angles in the order of micro-radians. A micro-radian is the angle subtended by a millimeter placed a kilometer away, so it's a very small angle indeed. If you manage to have a system that can pick up these very small angular deviations, then you can do x-ray phase-contrast.
The reason we want to do it is that these phenomena are much stronger than those produced by conventional x-ray attenuation, which means that the contrast in the images is very significantly increased and you can see things that, up to this point, you might have previously considered to be invisible.
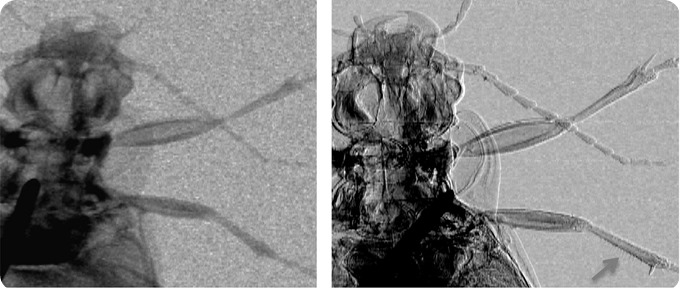
Attentuation (left) and phase (right) images of a ground beetle. The arrow in the bottom right corner of the latter image shows how thin hairs on the insect’s leg are visualized thanks to phase effects. Credit: Advanced X-Ray Imaging (AXIm) Group, UCL
Could phase-contrast X-ray imaging help to identify tumors in living tissue earlier?
Since Röntgen’s discovery over a hundred and twenty years ago, people have been exploiting the different attenuation of materials to create X-ray images. However, whenever you have materials (or tissues in medical imaging) with a very similar attenuation coefficient, then the contrast in your image tends to disappear.
Likewise, if you have, for example, a more significant difference but the object you are looking at is extremely thin, then again this leads to very small contrast in the image. So this is where phase-contrast can help. Since, as we said before, the mechanism is much stronger, you can generate contrast from features which are very similar in terms of X-ray attenuation characteristics.
That means that you can generate image contrast from things that you wouldn't normally be able to see. For example, tumors at the very early stages of formation or very faint tumors which, at the moment, are difficult to see. So in that sense, if you implement it clinically, picking up tumors earlier and seeing smaller tumors should be possible.
How long has this technique been around for and why hasn’t it been used in healthcare so far?
The first evidence for the use of X-ray phase-contrast was published in 1965 in a very dedicated experiment. Then everything went silent for about 30 years until the mid-90's where a number of third generation synchrotron facilities were built and that's when people started to look into this phenomenon in much more detail, realizing the massive potential it would have in medicine, but also outside medicine.
Initially people thought that you needed to have a synchrotron to be able to explore this phenomenon, these very faint phase changes or small angular deviations, and it was only more recently that techniques have developed that enable you to do similar things without having to use a synchrotron.
Indeed, there is one example of in vivo use of phase contrast imaging using a synchrotron and that is the synchrotron I come from at the Elettra light source. At Elettra there is a system that can and does perform phase contrast mammography in vivo on living patients. That is one of the last things I contributed to when I was still working in Trieste before I moved to UCL.
That’s also when I realized that bringing the patients into the synchrotron was not exactly what I wanted to do and I was much more interested in the opposite approach, which was taking the technology out of the synchrotron and adapt it so that it can be implemented with conventional X-ray sources because this is what is needed to transfer it into the clinic.
Since then, we've been developing approaches to try to achieve this, we had a breakthrough a few years ago and now we think we can do it.
What needs to be done before phase-contrast X-ray imaging could be put into clinical practice?
One of the projects we're working on at the moment is clinical, however it won't be in vivo. In collaboration with Nikon Metrology UK we have funding from the Welcome Trust to develop an intra-operative specimen imaging system that will try to determine the degree of involvement of tumor margins in breast-conservative operations.
At the moment, most breast operations are breast-conservation surgeries instead of full mastectomies, which means, instead of removing the entire breast, only the tumor is removed, so that the impact and the required plastic surgery that has to follow a full mastectomy is avoided.
The problem is that you have to make sure that the margins of the lump of tissue that you are removing are not involved. Meaning that the tumor is fully contained within this lump of tissue that you have removed.
The machine that we are developing should enable the surgeon to scan the tissue sample there and then, while the women is still on the operating table, and determine whether the margins are involved or not. If they are involved, he or she will then be in a position to continue the operation and remove more tissue . If they're not involved they can be confident that the operation has been completed successfully.
At the moment there is a high rate of re-operations, which means women have to be called back to have a second surgery after it has been found out at histology that the margins of the lump of tissue were involved.
Currently you have to use histology in order to confirm the involvement of the margins of the tissue lump and this may take a couple of weeks. It cannot be done there and then.
The impact we are trying to have is significant: being able to determine margin involvement there and then so that only a single operation is required. We aim to significantly reduce the number of re-operations that are needed in the future.
This will be our first clinical translation, even though we wouldn't be looking at imaging in vivo patients. The next step will be to scale the technology up to then target proper in vivo diagnostic applications.

Credit: Advanced X-Ray Imaging (AXIm) Group, UCL
What hurdles will need to be overcome?
Most of the hurdles are of an engineering nature. In order to make a system sensitive to this phase-based X-ray imaging method, we use X-ray masks. We have masks which are typically small squares, about 6 x 6cm and 10 x 10 cm, and bigger masks need to be created in order to be able to image larger samples.
You might need more flux to speed up the acquisition and sophisticated alignment mechanisms to position the masks in the right way, but I don't think there is any significant inherent physical principle that prevents this from happening. It's mostly engineering.
What impact do you think this technology will have on healthcare?
It has the potential to transform healthcare. One important application would be mammography. However, there is a very wide, diverse range of applications where this could be used in medical applications. The first thing we have looked at are tumors, because that is an area where early detection significantly increases the chances of curing that tumor, but there are other diseases as well. For example, osteoporosis, osteoarthritis, and many other types of medical problems could be targeted; we have started to explore application in cartilage imaging, because cartilage is invisible to conventional X-rays and at the moment you have to use MRI, so that's another idea that could be explored.
Wherever you use X-rays, you could have an improvement by making your X-ray systems more sensitive with phase.
Could the technology be used in other fields?
Absolutely. We have a project looking at security scans. In a series of proof-of-concept experiments, the technology was proven to be able to pick up threat devices better, identify explosives or pick up small components of a detonator device, for example.
Other obvious applications would be in material science, e.g. non-destructive testing, industrial testing, quality control on objects or materials.
One area which we think is very interesting is composite materials, the use of which is seeing constant increase, and which are difficult to test. We have proven that x-ray phase contrast imaging can pick up damage and defects even if they are very small or have just started to develop.
Another example is use in pre-clinical imaging for drug development studies, the list is very long.
How could phase-contrast X-ray imaging be used in pre-clinical studies?
In pre-clinical imaging, many studies of drug development or new treatment development are carried out on small animal models, typically mice and rats. This involves a lot of scans on animals, which is carried out using a small-animal CT scanner or a pre-clinical CT scanner and sometimes other techniques like MRI or methods based on visible light.
MRI is a fantastic method, but it takes lengthy acquisitions if you want to have a high resolution and it's very expensive. On the other hand, pre-clinical CT at the moment is limited by lack of soft tissue contrast.
Ideally, you would like to run longitudinal studies, which means imaging the same animal multiple times, but at the moment you cannot do this because the radiation dose that you deliver to the animal with current pre-clinical CT scanners is so high that it is even a struggle to carry out more than one or a few scans on the same animal.
Therefore, we hope to increase the sensitivity to soft tissues and reduce the dose. This is an area we continue to research and hope to eventually, significantly reduce the dose delivered in a single scan. This will allow studies to be more effective because you will be able to image the soft tissue and have many more scans of the same animal.
I think this is transformative in two ways. One is that it means a much better understanding of disease progression and development because you can carry out longitudinal studies. At the moment we use a number of animals and then sacrifice them at different time points, which means that there is an inter-animal variability which is something you cannot control very easily.
In addition to this, I think it's very important in terms of reducing the number of animals required by research. Instead of having to sacrifice a group of animals, you can do multiple studies on the same animal and hence you can scale down the total number of animals required by a study by a factor of 10 for example.
Are there any other types of X-ray imaging that could be potentially used in healthcare that have not been explored yet?
There are other approaches that are being explored, which are possibly more suitable for niche applications.
One very powerful method is X-ray fluorescence, which has elemental discrimination capabilities and people are using this research to look for traces of heavy metals in brain tissue, which could be one of the reasons why then people develop neurodegenerative diseases.
Another technique that has been of interest is X-ray diffraction, which has sensitivity to molecular organization of tissues and could be used for identifying some types of diseases.
Where can readers find more information?
About Professor Alessandro Olivo
 Alessandro (“Sandro”) Olivo is Professor of Applied Physics with the Department of Medical Physics and Biomedical Engineering at UCL, and the spokesperson of the Advanced X-Ray Imaging (AXIm) Group (https://www.ucl.ac.uk/medphys/research/axim).
Alessandro (“Sandro”) Olivo is Professor of Applied Physics with the Department of Medical Physics and Biomedical Engineering at UCL, and the spokesperson of the Advanced X-Ray Imaging (AXIm) Group (https://www.ucl.ac.uk/medphys/research/axim).
He graduated summa cum laude from the University of Trieste in 1995, did a PhD at the same university after a short stint as a hospital physicist, and moved to UCL in 2005 thanks to a EU-funded Marie Curie Fellowship.
Overall he has worked in x-ray imaging for 20 years, and is considered a pioneer in the field of X-ray phase contrast imaging, having co-designed the in vivo mammography system operational at the Trieste synchrotron and invented the “edge-illumination” and “coded-aperture” methods.
He has been awarded close to £11M in research funding, published 184 papers, and given 94 conference talks/seminars, 59 of which were invited. Articles on his work appeared, among others, in Nature, Scientific American, Physics Today, Physics World, the BMJ.