Tuberculosis is the leading infectious killer in the world, far above AIDS. It kills about 1.6 million of the 10 million it affects every year. About 30,000 patients worldwide now have XDR tuberculosis (that fails to respond to any of the four antibiotic categories in use at present).
On August 14, 2019, the US Food and Drug Administration (FDA) approved pretomanid, the latest of three drugs used in the new BPaL regimen that cured up to 90% of XDR tuberculosis with 5 pills a day, within just 6 months, in the Nix-TB trial in South Africa. This gives hope to “hopeless” tuberculosis patients. Meanwhile, a new paper published in the New England Journal of Medicine on August 15, 2019 also throws new light on the immense and growing significance of nonprofit organizations in the development of new antibiotics to treat resistant infections.
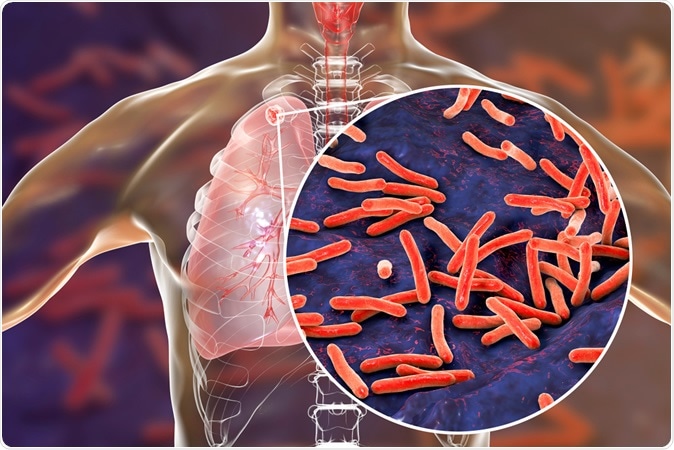
Secondary tuberculosis in lungs and close-up view of Mycobacterium tuberculosis bacteria, 3D illustration Credit: Kateryna Kon / Shutterstock
Current therapy for XDR tuberculosis requires up to 40 pills to be taken every day for up to 2 years, but still cures only 34% of these patients. The rest often waste away often even before a diagnosis is made. In some countries, older regimens are in place, relying on daily injectable antibiotics which can cause renal failure, deafness and psychosis.
The three drugs used here are pretomanid, bedaquiline and linezolid. These three drugs are all mycobactericidal, meaning they kill the tuberculosis bacilli. Most other drug regimens use only two killers with one bacteriostatic drug that stops the bacterial multiplication but allows it to live in an inactive state.
Pretomanid is the latest to gain approval. The other two drugs were already approved. Some groups argued for further pretomanid testing before approval. They seem to have been overruled by others who say a trial is not only unethical but impractical. This argument holds water since the only control group that can reasonably be set up would have to be given a far inferior treatment protocol. The FDA approved the newest drug in this regimen on August 14, 2019, paving the way for its universal adoption by the World Health Organization (WHO) and other global health initiatives.
The deadliest strain of tuberculosis yet known appeared first in a small South African town in 2006, killing 52 out of 53 patients, typically within a month of diagnosis. Most of them were young and had not been treated for tuberculosis before. Many of these were, in fact, health workers. The conclusion was clear: the infection had spread from other patients rather than developing in the index group.
A few months later, the infection was reported in 40 South African hospitals, triggering off worldwide testing. This revealed the presence of the killer strain in 28 countries, with two of every three cases being in China, India and Russia. It is 25 times more common in HIV patients, but many affected individuals have never had HIV.
For the next seven years, XDR tuberculosis was the focus of desperate medical battles, being treated with every kind of drug conceivably useful in this disease. Yet, 80% of patients continued to succumb to the inexorable illness, or to the adverse reactions caused by the drugs.
Now, the Nix-TB trial has shown a cure rate of over 90%. However, the use of linezolid (previously used to counter severe hospital-acquired infections) is associated with neuropathy in the feet, or bone marrow suppression. A new trial called ZeNix is now underway to find the right dose of this harsh drug.
The TB Alliance, a New York-based nonprofit, was responsible for bringing out pretomanid. This brought another relevant issue to light: new antibiotic development may well be taken over by nonprofits, to allow the development of new drugs which can then be distributed in an affordable and timely manner.
But why turn to nonprofits rather than innovation-driven entrepreneurial companies? The NEJM paper published on August 15, 2019, gives some compelling reasons.
Penicillin was the first antibiotic, reducing the death rate due to infectious diseases by 80%. However, new and emerging strains of bacteria that are resistant to one or multiple antibiotics are posing a new danger to infection control, causing some scientists to warn of a coming “post-antibiotic era’ in which untreatable infection will again ravage the world. New antibiotics against these resistant organisms are urgently required, but drug companies are often uninterested in this area due to the huge expense of new antibiotic development compared to their small returns.
This is partly because most antibiotics are used for a very short time (days or weeks) and cost little to the patient. In contrast, drugs for chronic medical conditions such as high blood pressure command large returns on investment because of their long-term use and high retail prices.
Economic incentives such as funding by government agencies and the formation of partnerships between public and private firms have succeeded in tripling the rate of new antibiotic development. The FDA saw a three-fold rise in new antibiotic approvals over the last 6 years, which indicates a welcome change from the 90% fall over the preceding 30 years (from 1983 to 2012).
However, few of these drugs are useful in treating extremely drug-resistant (XDR) infections caused mostly by mycobacteria and gram-negative bacilli. Many of them are directed against the same pathogen, lowering their utility. Add to this the fact that antibiotics against XDR infections already have a very small target.
Each new antibiotic released over the last 10 years has therefore found a still smaller share of the already fragmented market, causing poor sales. This has, in fact, driven Achaogen, a company which had a drug newly approved in 2018, to bankruptcy.
Along with stricter antibiotic regulation and shorter treatment regimens, these factors have made revenue returns on investment (estimated to be $1 to $2 billion per approved antibiotic) so low as to be impractical for most drug companies. However, without such development, 10 million people could die of drug-resistant infections by 2050, according to UN estimates.
This is why some health economists believe that private entrepreneurial development of antibiotics is no longer the answer. New antibiotics that are most crucial to current needs, and which actually improve patient outcomes, must be developed, and this is possible only with a new development system that centers around nonprofits. Nonprofits don’t need to please shareholders. They can keep their drug prices as low as they want. They can concentrate on unmet needs. They would be able to break even on investment even when peak sales of new drugs are low, and could regulate the use of the new drugs to limit the emergence of resistance.
Various models of highly successful nonprofit involvement in this field exist, some striking examples being the TB Alliance and Medicines for Malaria Venture. Tuberculosis is one of the neglected but extremely needy areas of new antibiotic development. This disease ravages low-income regions of the world, which means new drugs will not reap much profit. Thus Helen Boucher, director of the Tufts Center for Integrated Management of Antimicrobial Resistance, says, “There isn't a market to sell [a TB drug] to make money, so it was imperative that a nonprofit take that on.”
Mel Spigelman, president and chief executive of TB Alliance, says, “We can have a huge impact on the lives of people who are afflicted and also take a major step ultimately toward, really, the eradication of a disease like TB."