A study conducted by German researchers has shown that the neurological symptoms observed in some patients with coronavirus disease 2019 (COVID-19) may be caused by autoantibodies targeting the brain.
The team says that although some autoantibodies still need to be identified, they may at least partly explain the multiple organ disease that can occur in COVID-19 patients and help to inform immunotherapy decisions in certain cases.
A pre-print version of the paper is available on the server medRxiv*, while the article undergoes peer review.
Neurological symptoms in COVID-19
In some cases, patients with COVID-19 develop neurological symptoms, which may range from mild such as a reduced sense of smell to severe symptoms such as myoclonus (sudden involuntary muscle spasms).
Researchers have debated whether such symptoms may be caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infecting the brain.
However, in most patients who develop neurological symptoms, SARS-CoV-2 is not detected in the cerebrospinal fluid (CSF). This suggests that other cellular or humoral mechanisms may be involved, including autoimmunity.
Previous studies have suggested that certain viruses can lead to autoimmune encephalitis, with examples including HIV, adenovirus, and hepatitis C.
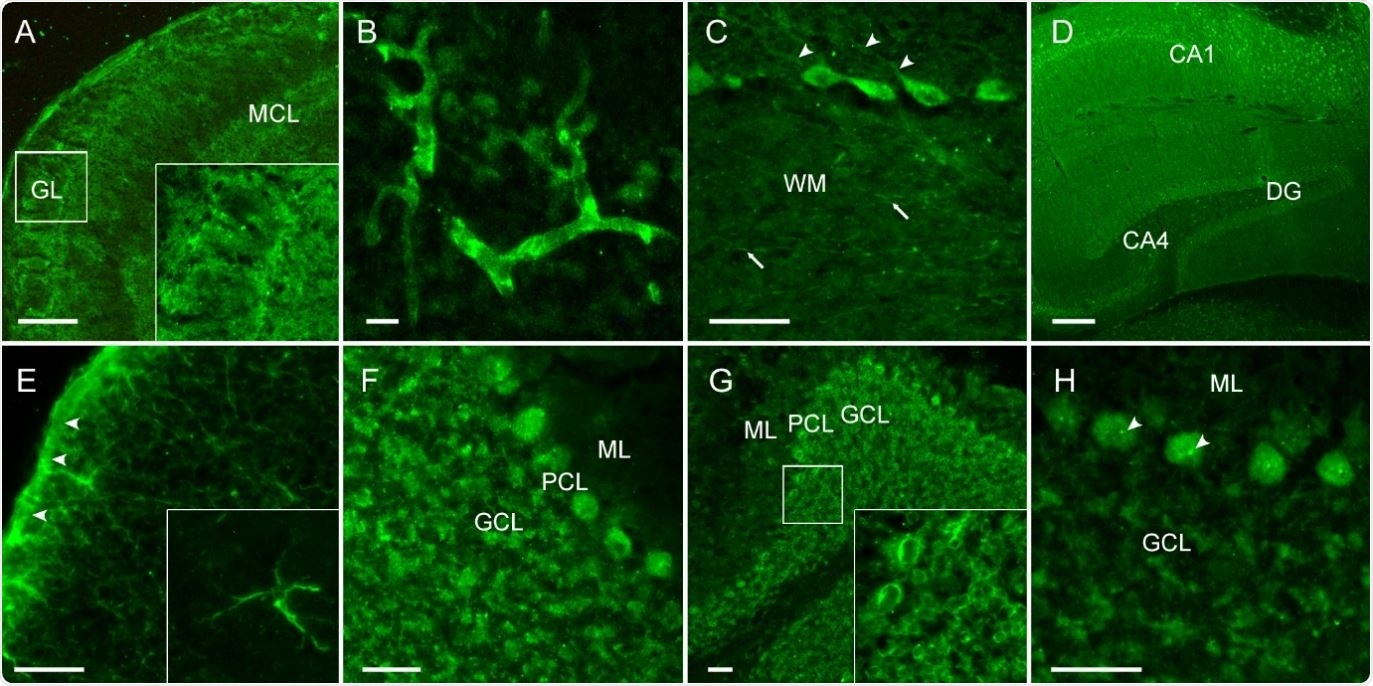
CSF of COVID-19 patients shows strong IgG autoreactivity on unfixed mouse brain sections. Representative images of indirect immunofluorescence demonstrate autoantibody binding to circumscribed anatomical structures including (A) neuropil of the olfactory bulb, (B) medium-sized vessels in the brain, (C) proximal dendrites of Purkinje neurons (arrowheads) and myelinated fibers (arrows) in the cerebellum, (D) neuropil in the hippocampus, (E) glia limitans (arrowheads) and astrocytes (enlarged box) throughout the brain. Several autoantibodies target intracellular antigens, such as (F) densely clustered intraneuronal epitopes, (G) perinuclear antigens or (H) nucleoli (arrowheads) as part of an anti-nuclear antibody response.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
What did the current study involve?
Christiana Franke (Charité – Universitätsmedizin Berlin) and colleagues recently tested whether autoantibodies were present in eleven patients with severe COVID-19 and unexplained neurological symptoms.
Between March and May 2020, the team assessed a large panel of antineuronal and anti-glial autoantibodies using serum and CSF samples from intensive care patients with symptoms including delirium, epileptic seizure, dystonia, and myoclonus.
The team reports that no SARS-CoV-2 was detected in any of the CSF samples.
CSF inflammation was indicated in most patients, and all had increased CSF levels of neurofilament light chain (a biomarker for axonal damage).
“Elevated NFL levels might reflect direct tissue destruction from viral replication or from inflammatory damage,” writes the researchers. “Whether this is a transient elevation or a continuous transformation into a degenerative phenotype is yet to be determined.”
All patients had anti-neuronal antibodies in their serum or autoantibodies in their CSF that target surface antigens known to be involved in central nervous system disease.
Among these antigens were well-established proteins such as the NMDA receptor and myelin antigens.
The researchers say that one patient with a high level of serum IgG autoantibodies against NMDA receptors required cardiopulmonary resuscitation. The raised IgG level may have reflected NMDA receptor encephalitis, which often causes arrhythmia and autonomic dysfunctions, they add.
Strong IgG binding in specific brain sections
To test CSF for anti-neuronal autoantibodies not routinely included in clinical assays, the team performed indirect immunofluorescence on mouse brain sections. Staining patterns showed that most patients exhibited strong immunoglobulin G (IgG) binding. The patterns showed specificity for the hippocampus, olfactory bulb, vessel endothelium, astrocytic proteins, and the neuropil of basal ganglia.
“The high frequency of CSF anti-neuronal and anti-glial autoantibodies is remarkable, as is the confinement to specific immunofluorescence patterns,” writes Franke and colleagues.
“Although more than one patient each had IgG autoantibodies targeting neuropil, astrocytes or medium-sized blood vessels, it will require larger patient cohorts for linking a given autoantibody pattern to clinical symptoms.”
What are the study implications?
According to the authors, recent studies have demonstrated that some viral infections can lead to secondary autoimmune encephalitis, including HIV, hepatitis C Epstein-Barr virus, human herpes virus, enterovirus, and adenovirus.
“The present findings suggest that SARS-CoV-2 is no exception to this general principle.”
The researchers say the retrospective nature of the study means the findings could not be used to guide decisions about immunotherapy for patients. However, improvement has been reported for some COVID-19 patients with encephalitis after they received steroids. This suggests that patients with CSF autoantibody-positive COVID-19 may benefit from immunotherapy in the future.
“The high frequency of autoantibodies targeting the brain in the absence of other explanations suggests a causal relationship to clinical symptoms,” writes Franke and colleagues.
“While several underlying autoantigens still await identification in future studies, the presence of autoantibodies may explain some aspects of multiorgan disease in COVID-19 and can guide immunotherapy in selected cases,” concludes the team.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Journal references:
- Preliminary scientific report.
Franke C, et al. High frequency of cerebrospinal fluid autoantibodies in COVID-19 patients with neurological symptoms. medRxiv 2020. doi: https://doi.org/10.1101/2020.07.01.20143214
- Peer reviewed and published scientific report.
Franke, Christiana, Caroline Ferse, Jakob Kreye, S. Momsen Reincke, Elisa Sanchez-Sendin, Andrea Rocco, Mirja Steinbrenner, et al. 2021. “High Frequency of Cerebrospinal Fluid Autoantibodies in COVID-19 Patients with Neurological Symptoms.” Brain, Behavior, and Immunity 93 (March): 415–19. https://doi.org/10.1016/j.bbi.2020.12.022. https://www.sciencedirect.com/science/article/pii/S088915912032465X.