Abnormal Inflammation, Severe COVID-19
Evidence is accumulating that an aberrant inflammatory response is partly responsible for severe COVID-19. Therapy with dexamethasone, a potent corticosteroid, has proved successful in reducing mortality in severe COVID-19, probably due to its ability to suppress inflammation.
The current study uses single-cell RNA sequencing data from bronchoalveolar lavage fluid (BALF) and peripheral blood mononuclear cells (PBMCs) of patients with COVID-19 to understand the underlying disease process.
In both SARS and MERS, which are earlier outbreaks of respiratory infections caused by pathogenic coronaviruses, immunity was developed by CD4 memory T helper (Th) cells in the airway, mediated by interferon (IFN)-γ. The same type of response is also seen in SARS-CoV-2 disease in humans, and a sustained intense Th1 response is characteristically seen in severe COVID-19.
COVID-19 Patient Lungs Show Th1 Responses
To test the hypothesis that the exaggerated inflammation seen in this condition is heavily dependent on Th1 responses, the researchers focused on CD4 T cells within BALF.
They found that the percentage of CD4 cells within the total T cell population did not vary between patients and controls. However, in patients, the CD4 cells had a higher expression of over 300 genes and a subdued expression of over 130 genes. These genes are commonly expressed in cellular pathways involving IFN-γ and complement.
These CD4 cells were polarized towards Th1 lineage rather than Th2 or Th17. Interestingly, the same shift was not observed in PBMCs, which indicates a local phenomenon occurring within the Th cells specifically targeting the virus at the site of lung inflammation.
Complement and Immune Regulation
Prior research by the same team has shown that complement is among the most active pathways in CD4 T cells infiltrating the lung tissue, and that it is powerfully stimulated by SARS-CoV-2, especially the complement factor 3 (C3) in the lung epithelium. Other studies show that the lung tissue in COVID-19 is a hotbed of complement activity.
C3 is split into activation fragments, among which C3b binds to T cells via CD46 receptors. COVID-19 patients have CD4 T cells in their lungs bearing CD46, and when this is bound by C3b, an autocrine loop is set up, which results in a coordinated increase in Th1 differentiation and then a suppression of T cell activity, in synchrony with T cell receptor stimulation.
In terms of cytokines, when the binding of ligands activates T cells to the CD3 and CD46 receptors, they produce IFN-γ and subsequently IL-10, finally tuning out the former for a suppressive IL-10 response. This response is only seen in human CD4 T cells and not in mouse T cells.
Researchers say that this shift marks the change from effector T cell phenotype, which is required for viral clearance, into IL-10 secreting T cells that help to limit organ damage secondary to the inflammatory response and is a normal part of the T cell response.
In support of this explanation, non-IL-10-producing mice infected with pathogenic trypanosomes clear the parasites rapidly but die of organ dysfunction related to excessive inflammation mediated by Th1 cells.
The question then was whether the severe form of COVID-19 was related to the failure of Th1 suppression. BALF RNA-seq data shows that while Th1-related genes are upregulated in COVID-19 patients, IL-10 levels are fourfold lower, lending further support to the idea that this suppression is indeed not at work.
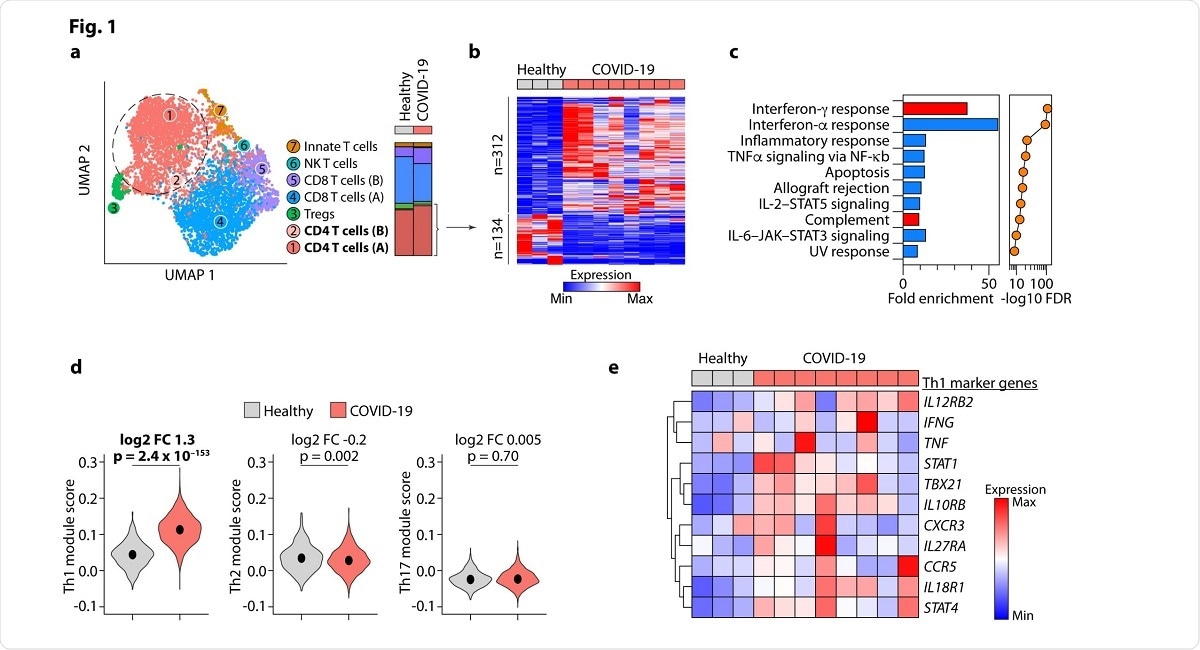
Image Credit: https://www.biorxiv.org/content/10.1101/2020.07.18.210161v1.full.pdf
Th1 and Vitamin D
The research also uncovered the fact that activated CD4 cells induced by CD3 and CD46 binding had about 2000 differentially expressed genes (DEGs) relating to transcription factors. CD46 induces 24 transcription factors, and one of these is the vitamin D receptor (VDR).
The latter has been identified as one of the most highly expressed transcription factors regulating the DEGs expressed by CD4 T cells in BALF of patients vs. healthy donors.
Moreover, another enzyme called CYP27B1 is also expressed at high levels, and this is also called vitamin D 1α hydroxylase, the final activator of vitamin D for its physiological role in the body.
The ability of complement to induce the expression of both VDR and the Vitamin D activating enzyme by T cells via CD46 binding is probably indicative of an autocrine or paracrine loop, which means that T cells activate VDR and respond to it.
Vitamin D and Cytokine Responses
Vitamin D has numerous functions in the body, including immune regulation in specific cell populations and particular contexts. Its deficiency is linked to a higher prevalence of infectious and autoimmune diseases, all of which are linked by the key presence of vitamin D.
The study showed that vitamin D upregulates VDR, induces genes such as CTLA4 (which modifies C3 within the cell to bind CD46), CD38 and CYP24A1, and suppresses both type 1 and type 3 cytokines. It also induces IL-10 and IL-6, with other genes such as JUN, BACH2 and STAT3. It can also induce the differentiation of powerful suppressive cells called type 1 regulatory T cells.
In the present study, the most prominent effect of vitamin D was a dose-dependent increase in cytokine gene expression by Th cells, especially IL-6, which usually promotes inflammation. Vitamin D caused repression of IFN-γ and IL-17 and induction of IL-10 and IL-6. This is the first time that vitamin D has been shown to induce IL-6.
IL-6 Promotes IL-10 in the Presence of Vitamin D
IL-6 and IL-10 showed a strong correlation, indicating that one may be dependent on the other. Experiments showed that IL-6 receptor-mediated signaling by IL-6 leads to IL-10 production by Th cells.
When the IL-6R blocking antibody tocilizumab was added, the IL-10 production fell significantly. However, if IL-6 is provided to Th cells without vitamin D, the result is the production of the pro-inflammatory IL-17 instead. Therefore, IL-6 may be shifted to an anti-inflammatory role in the presence of vitamin D by the latter’s ability to induce IL-10.
Vitamin D and other Transcription Factors
Cytokine signaling pathways often depend on phosphorylation, and so the current experiment was extended to study the presence and impact of this process.
The researchers found that vitamin D induces other transcription factors such as STAT3 and also its phosphorylation via IL-6. The phosphorylation of STAT3 enhances IL-10 production. When tocilizumab was added to block the IL-6 receptor, STAT3 was induced but not phosphorylated.
Vitamin D modifies the size of super-enhancer peaks via epigenetic changes. Super-enhancers are domains of regulatory enhancing genes that are needed to regulate many essential genes. In this way, it alters the expression of BACH2, STAT3 as well as IL-10.
Vitamin D causes the production and phosphorylation of a protein called c-JUN without the help of IL-6. This is involved in DNA transcription and probably takes part in gene regulation by vitamin D as well. BACH2 is another vital transcription factor that regulates immune function, and its deficiency is linked to autoimmune disease in humans.
Vitamin D induces BACH2 expression in Th cells, and this molecule mediates much of its immunoregulatory function. Among the most commonly BACH2-induced genes is that encoding the IL-6 receptor. Therefore, a normal BACH2 level is required for the regulation of genes induced by vitamin D. When deficient, this prevents the induction of IL-6R and the STAT-3 signaling that causes IL-10 production.
Vitamin D and Steroids
Finally, many of the genes regulated by vitamin D are also common to corticosteroid activity. Therefore, dexamethasone might owe its beneficial effect partly to its enhancement of vitamin D effects via the upregulation of VDR.
In patients with severe COVID-19, addition of VitD to other immunomodulatory agents might be beneficial.”
High doses of steroids could be avoided by adding vitamin D as well, which helps to reverse the downregulation of vitamin D-mediated immunomodulation in severe COVID-19.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources