Researchers in the U.S. have shown both preventive and treatment effects of an antibody cocktail in animal models of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) - the agent that causes coronavirus disease 2019 (COVID-19).
Christos Kyratsous and colleagues from Regeneron Pharmaceuticals, New York, had previously identified that a combination of the antibodies REGN10933 and REGN10987, which targets the SARS-CoV-2 spike protein, potently neutralizes the virus in vitro. The spike protein is the main surface structure the virus uses to bind to human host cell membranes and gain viral entry. The research is published on the preprint server bioRxiv* in August 2020.
Now, teaming up with collaborators at Texas Biomedical Research Institute in San Antonio and BIOQUAL in Rockville, Maryland, the researchers have demonstrated the efficacy of the antibody cocktail in two animal models: the rhesus macaque and the golden hamster.
The authors say their findings provide evidence that this REGN-CoV-2 therapy, which is currently being tested in clinical trials of COVID-19, may provide significant clinical benefit in both the prevention and treatment setting.
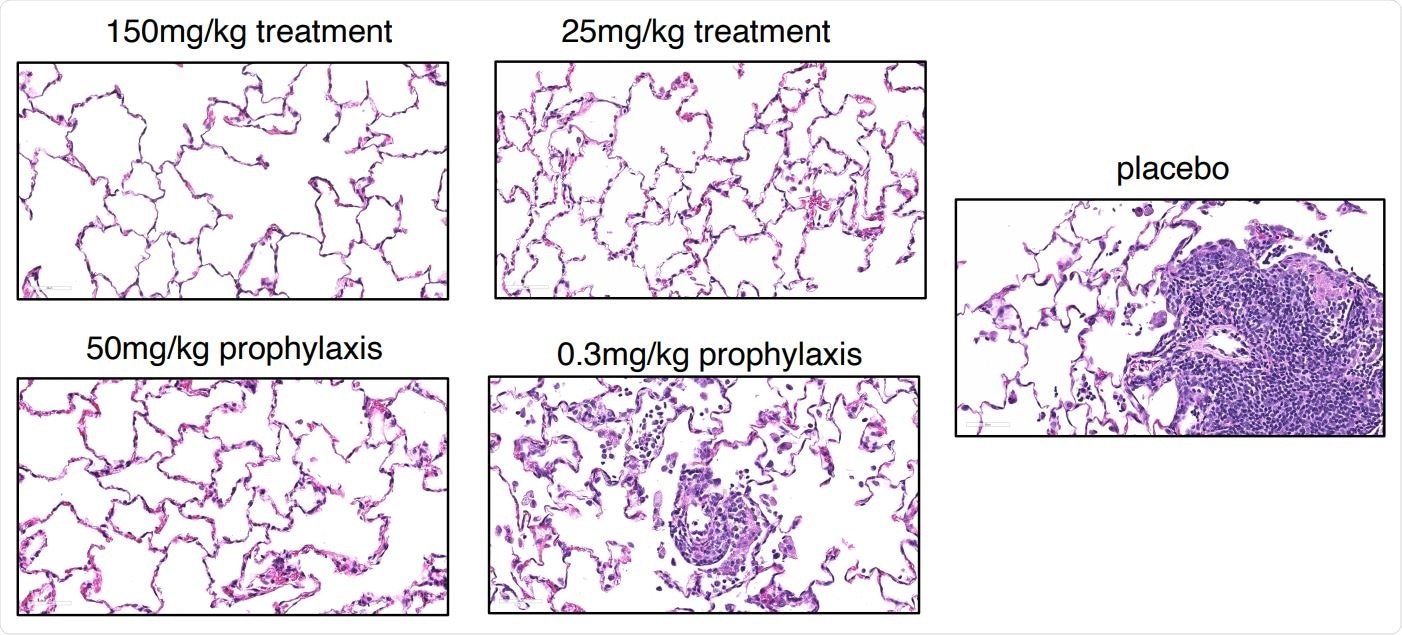
Prophylactic and therapeutic efficacy of REGN-COV2 in the rhesus macaque model of SARSCoV-2 infection (NHP Study #2). Representative images of histopathology in lungs of treated and placebo animals.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
No relevant animal model has yet been established
Since the COVID-19 outbreak began in Wuhan, China, late last year, researchers worldwide have been urgently seeking effective strategies to prevent and treat the disease.
Various studies have reported on monoclonal antibodies that potently neutralize the SARS-CoV-2 spike protein, but in vivo evaluation is only in the early stages and has mainly been focused on prevention.
Furthermore, since animal models of SARS-CoV-2 infection are still in development, no one model has yet been established as more relevant to disease in humans.
The rhesus macaque, which is widely used in preclinical efficacy studies, only exhibits a mild form of disease following SARS-CoV-2 infection. In contrast, the golden hamster exhibits a much more severe form, characterized by significant weight loss and severe lung pathology.
“Indeed, based on the extremely diverse manifestations of COVID-19 in humans, multiple animal models may be needed to mimic various settings of human infection,” write Kyratsous and colleagues.
Combining the models to capture diverse pathology of SARS-CoV-2 infection
Now, Baum and the team have used these two animal models to compare the efficacy of the REGN-CoV-2 cocktail in diverse disease settings for a more detailed understanding of how monoclonal antibodies might reduce viral load and pathology in humans.
To assess the prophylactic effect of the antibody combination in the context of mild disease, rhesus macaques received 50 mg/kg of REGN-CoV-2 prior to challenge with 1x10^5 PFU (plaque-forming unit) of SARS-CoV-2 three days later. Nasopharyngeal (NP) swabs and bronchoalveolar lavage (BAL) fluid were collected on days 1, 3, and 5 post-challenge.
Viral RNA was almost wholly ablated in the mild disease model
Compared with placebo-treated animals, viral RNA was almost wholly ablated in most animals, indicating that the therapy can almost completely stop infection becoming established. This effect was seen across all NP and BAL measurements, indicating that viral loads were significantly reduced in both the upper and lower airways.
To assess the treatment effect of REGN-CoV-2, the macaques were given 25mg/kg or 150mg/kg doses one day following challenge with 1x10^6 PFU of the virus.
Compared with placebo treated-animals, those treated with either dose of REGN-CoV-2 showed accelerated clearance of viral RNA in all measurements of both NP and BAL samples.
The antibody cocktail was therefore effective at reducing viral load, even when administered once the infection has already occurred, says the team.
On assessing the lungs of the macaques, the researchers found that the incidence of interstitial pneumonia (including both the number of animals affected and the number of lung lobes involved) and the severity were significantly reduced in both the prophylactic and treatment setting, compared to the placebo setting.
What were the effects on more severe disease?
To assess the prophylactic effect of REGN-CoV-2 in the context of more severe disease, the golden hamster was given 50, 5, or 0.5mg/kg of the drug two days prior to challenge with 2.3x10^4 PFU of virus.
At all of the doses given, the animals were protected from weight loss, and viral loads in the lungs were significantly reduced seven days following viral challenge.
The team also observed a significant therapeutic benefit among the hamsters, with viral replication and lung pathology reduced by treatment with 50mg/kg and 5mg/kg doses just one day following viral challenge.
The researchers say that to their knowledge, this is the first study to show the ability of any treatment to minimize weight loss in the hamster model of SARS-CoV-2 infection, thereby suggesting the potential benefit of antibody treatment for severe infection.
Potential for both protecting against and treating disease
The authors say the findings point to the therapeutic potential of REGN-COV2 in both protecting against SARS-COV-2 infection and treating COVID-19.
They also say the effect the antibody cocktail had on viral RNA levels in NP and BAL swabs suggests not only the potential to prevent disease following exposure but also to limit further transmission.
“Our data provide evidence that REGN-COV2 based therapy may offer clinical benefit in both prevention and treatment settings of COVID-19 disease, where it is currently being evaluated (clinicaltrials.gov NCT04426695, NCT04425629 and NCT 04452318),” concludes the team.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Journal references:
- Preliminary scientific report.
Kyratsous C, et al. REGN-COV2 antibody cocktail prevents and treats SARS-CoV-2 infection in rhesus macaques and hamsters. bioRxiv 2020. doi: https://www.biorxiv.org/content/10.1101/2020.08.02.233320v1
- Peer reviewed and published scientific report.
Baum, Alina, Dharani Ajithdoss, Richard Copin, Anbo Zhou, Kathryn Lanza, Nicole Negron, Min Ni, et al. 2020. “REGN-COV2 Antibodies Prevent and Treat SARS-CoV-2 Infection in Rhesus Macaques and Hamsters.” Science, October, eabe2402. https://doi.org/10.1126/science.abe2402. https://www.science.org/doi/10.1126/science.abe2402.