Researchers from Chongqing Medical University have tried to answer one of the burning questions regarding the longevity of immune response to coronavirus disease 2019 (COVID-19): How long does immunity last after infection? Their study looked at the lasting effects and continued immunity against severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) over eight months after recovery from the infection.
Their study titled, “Changes of Humoral Immunity Response in SARS-CoV-2 Convalescent Patients over eight months,” has been released on pre-print server medRxiv*.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Background
There has been a steep rise in the number of cases of COVID-19, with many nations experiencing a second wave of infections as businesses and educational institutions reopen. At present, over 5.9 million people around the world have been infected with the virus and over 1.26 million have died. The second wave of infections had started in October in several parts of the world.
Some studies have shown that after a bout of COVID-19, there has been a decline in the antibody response within three months after recovery from the infection. In early months this was reported frequently, with many experts speculating this to raise the risk of reinfection among convalescent patients.
This study was conducted to investigate further the level of antibodies over time and the continued immune response to the infection over months after recovery from the infection.
Study design
For this study, the team of researchers included twenty patients who had earlier recovered from COVID-19. Blood samples were obtained from the patients between February and October 2020. This corresponds to a median of 25 days (ranging between 5 and 33 days) and 230 days (ranging between 221 and 248 days) after the onset of symptoms.
The antibody titers were assessed using Enzyme-linked immunosorbent assay (ELISA) to check for the anti-SARS-CoV-2 spike (S) receptor-binding domain (RBD) IgG over eight months. The neutralizing antibodies (NAbs) were measured using SARS-CoV-2 wild-type (S-D614) and S-G614 mutant pseudovirus-based assays in 293T-ACE2 cells. SARS-CoV-2 S-G614 has been the dominant strain of the virus around the world. Levels of antibodies 3 Standard Deviation over those of negative control patients was considered to be positive. The inhibitory dose (ID50) was assessed for titers of NAbs.
Findings
The study findings show the following:
- Antibodies of all the participants against the spike RBD proteins fell from a mean OD450 value of 1.78 (range 0.55 to 2.72) at the start of the study in February to 0.38 (range 0.15 to 1.01) in October over eight months
- Values less than OD450 value 0.26 were considered to be negative
- From the samples in October, the IgG level of five participants (25 percent) fell below 0.26 (considered seronegative)
- This fall was seen in the pseudovirus neutralization assay.
- The NAb titers fell from the mean ID50 value 836.55 (range 263-1160) to 170.30 (range 33 to 365) over the study period
- NAb titers of three participants (15 percent) fell below the threshold considered to be positive eight months after the onset of the symptoms
- NAb titers correlated with IgG levels in the study
- SARS-CoV-2 S-G614 has been the dominant strain of the virus around the world.
- NAb titers of five participants (25 percent) fell below the threshold against S-G614 mutant pseudovirus.
- The neutralizing capacity of the convalescent plasma for SARS-CoV-2 S-D614 and S-G614 was significantly different
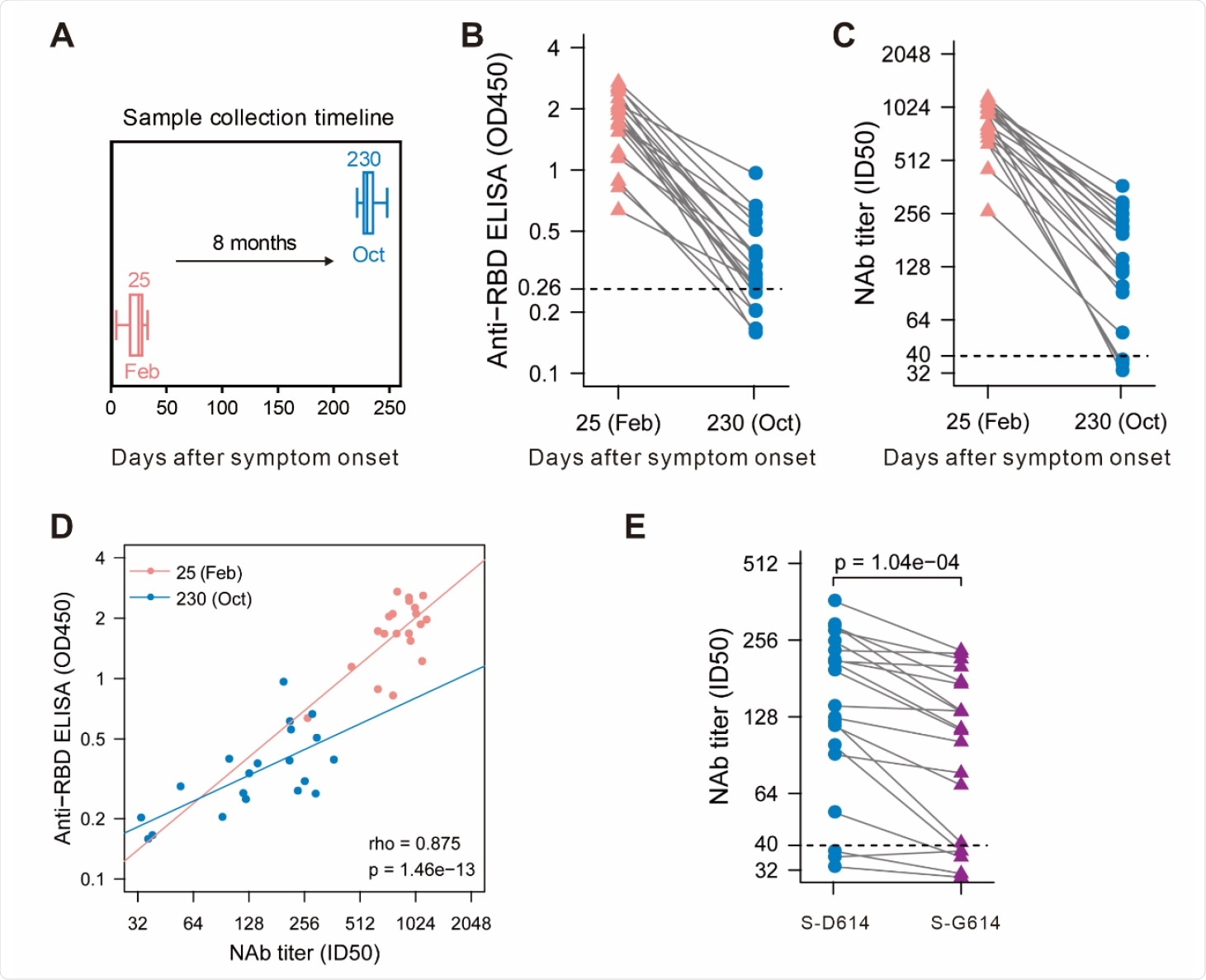
Maintenance of humoral response to SARS-CoV-2 in convalescent patients over 8 months. (A) Blood samples were collected in February and October. Enzyme-linked immunosorbent assay (ELISA) (B) and pseudovirus-based neutralization assay (C) were performed to detect IgG levels and neutralizing antibody (NAb) titers against SARS-CoV-2. The thresholds of detection were 0.26 of OD450 value and 1:40 of ID50, separately. (D) Correlation of IgG and NAb level. (E) Different neutralizing activity of convalescent plasma between SARS-CoV-2 S-D614 and S-G614 mutant at 8 months after symptom onset.
Conclusions and implications
This study revealed the changes in humoral immunity response in the plasma samples of those recovered from COVID-19 over eight months. It shows that there is a decline in the IgG and NAb levels over time. There is also a significant correlation between IgG and NAb levels seen at the start of the study in February. This was not seen in October. This meant that protective immunity levels may not be the same in terms of neutralizing antibodies eight months after recovery.
The authors write, “Facing the challenge of the second wave of SARS-CoV-2, the risk of reinfection by the currently dominant SARS-CoV-2 S-G614 variant is worthy to be considered, especially for Chinese convalescent patients infected by wild-type SARS-CoV-2.” With fall in NAb it can be speculated from this study that there is a “possible loss of the protective ability of convalescent plasma with lower titers to SARS-CoV-2 S-G614 variant, like the reinfection case found in Hongkong.”
The team calls for more research on the longevity of humoral immune response against SARS CoV-2 to evaluate the efficacy of the upcoming vaccines as well as the effectiveness of the purported herd immunity. This was a small study with only twenty patients and thus larger studies in the future could help in understanding the immune mechanisms against the infection better, state the researchers.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Journal references:
- Preliminary scientific report.
Peng, Pai et al. (2020) Changes of Humoral Immunity Response in SARS-CoV-2 Convalescent Patients over eight months. medRxiv, doi: https://doi.org/10.1101/2020.11.06.20227439, https://www.medrxiv.org/content/10.1101/2020.11.06.20227439v1
- Peer reviewed and published scientific report.
Peng, Pai, Jie Hu, Hai-jun Deng, Bei-zhong Liu, Liang Fang, Kai Wang, Ni Tang, and Ai-long Huang. 2021. “Changes in the Humoral Immunity Response in SARS-CoV-2 Convalescent Patients over 8 Months.” Cellular & Molecular Immunology 18 (2): 490–91. https://doi.org/10.1038/s41423-020-00605-4. https://www.nature.com/articles/s41423-020-00605-4.