A potentially valuable new study discusses the role of lipids in the hyper-inflammatory phenomena that take place within the lungs of patients with severe or critical COVID-19. Involving a large variety of specialized lipids with biological activity, this process includes the generation and release of pro and anti-inflammatory lipids. The study appeared as a preprint on the medRxiv* server in December 2020.
In the large majority of cases, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection is mild or even asymptomatic. However, in a significant percentage, the disease manifests as bilateral pneumonia with exaggerated inflammation, progressive respiratory compromise, and the onset of acute respiratory distress syndrome (ARDS), which may make it necessary to put the patient on mechanical ventilation.
The autopsy findings in such patients show lungs with diffuse damage to the alveoli, small and medium vessel thrombosis, pulmonary embolism with the infiltration of inflammatory cells, and a dysregulated systemic inflammatory scenario called a cytokine storm. This is due to the release of a cascade of pro-inflammatory cytokines such as tumor necrosis factor-α, interleukin (IL)-1β, IL-6, IL-7 granulocyte-colony stimulating factor, and is an important player in the lethal nature of the illness.
The researchers from Canada and Morocco sought to draw attention to the other immunological effector molecules, such as bioactive lipids, that participate in this prolonged state of inflammation. Eicosanoids and their metabolites are lipids known to promote the recruitment and activation of immune cells and platelets. Conversely, specialized pro-resolving mediators (SPMs) are lipids, mostly docosanoids, that reduce inflammation and enhance tissue recovery.
Study Details
The current study aims to measure the levels of these specialized lipid mediators in the lung. The researchers used bronchoalveolar lavage fluid from 33 patients with COVID-19-linked illness who were on mechanical ventilation and from 25 controls. Earlier, the levels of various circulating cytokines have been extensively assessed. Thus, the current analysis focused on bioactive lipids in the lung fluid to discover a possible "lipid storm" in severe COVID-19.
Increase in COX Metabolites
The first assay was for cyclo-oxygenase (COX) pathway metabolites. In healthy people, the only COX compounds found were PGD2, and 12-HHTrE, derived from the enzyme thromboxane synthase. The former was eight times more common. PGD2 is a molecule that enhances the recruitment and activation of eosinophils, basophils, and other cells of the innate lymphoid series, its activity being mediated through the DP2 receptor.
In patients with COVID-19, there was a marked increase in COX metabolites across the board, probably due to the hike in the COX inducer, IL-1β. Of the 33 patients, 27 had measurable levels of PGD2 and 6-keto-PGF1α (derived from PGI2), and all showed the presence of PGE2, PGF2α, thromboxane B2 (TXB2), derived from TXA2, and 12-HHTrE.
The most significant increase was in TXB2, followed by smaller increases in PGE2 and 12-HHTrE. Several of these are associated with bronchoconstriction and with the recruitment and activation of inflammatory cells and of platelets, fitting in with the observed picture in COVID-19.
PGE2 is anti-inflammatory, acting via the EP2 and EP4 receptors to increase cAMP levels in the cells. It thus inhibits the activity of white cells involved in innate immunity. PGE2 and PGI2 may have beneficial effects on the lungs by reducing leukocyte activation and enhancing microvascular dilation, respectively.
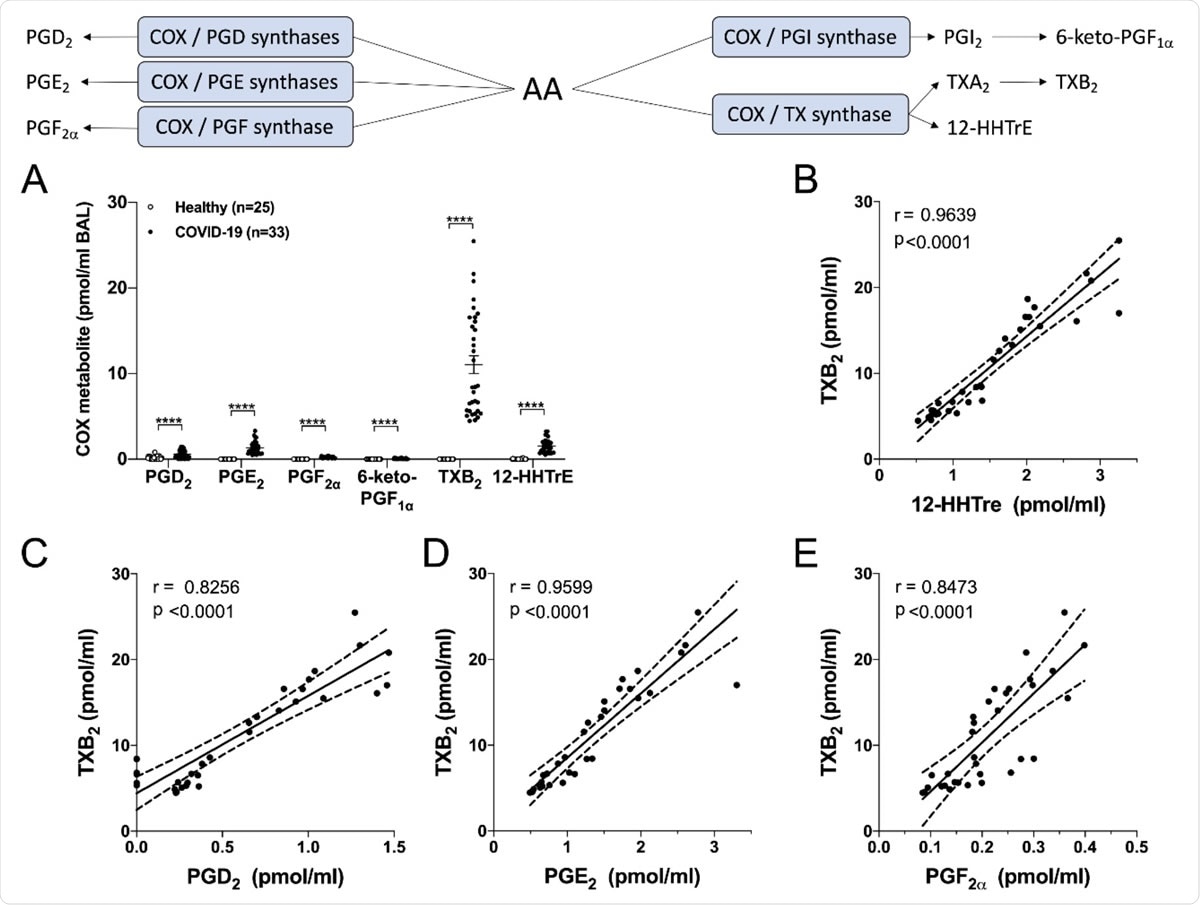
Levels of cyclooxygenase-derived metabolites in the BAL fluids of healthy and severe COVID-19 patients. BALs were obtained and processed as described in Methods. A) Results are from the BALs from 25 healthy subjects and 33 severe COVID-19 patients. P values were obtained by performing a Mann-Whitney test: **** = p < 0.0001. B-E) Correlation tests were performed with COVID-19 patients by using the non-parametric Spearman’s rank. P<0.05 was the threshold of significance. The linear regression is represented with a solid line and the best-fit lines of 95% confidence are represented with dotted lines.
Increased LTB4 Levels
ARDS is also associated with leukotriene (LT) activity, these being very powerful bioactive lipids. The study examined the levels of LTs formed via the 5- and 15-lipooxygenase pathways and found that they were not detectable in healthy individuals. The exception was LTB4, found in about 15%.
In COVID-19 patients, however, both LTB4 and its metabolite were increased to levels similar to those in the BAL of patients with ARDS. The ratio of these compounds favored omega-oxidation of LTB4. Both TXB2 and LTB4 were strongly correlated.
Again, the association between the COX pathway and LT synthesis may also indicate the role played by the latter in causing inflammatory lung damage in severe COVID-19. Though LTB4 is linked to antiviral defense, the omega-oxidation products of LTB4 are also increased, similar to the situation observed in cystic fibrosis and in severe burns, suggestive of poorly controlled infection.
Increased Cysteinyl-LT Metabolites
Most patients with severe COVID-19 also had higher cysteinyl-LTs, LTE4 and EXE4, with the latter being higher in many cases. These are metabolites of the 5- and 15-lipooxygenase (LOX) pathways, respectively. The researchers also found a correlation between LTB4 and EXE4 levels, as with the levels of most of the bioactive lipids measured here. EXE4 levels in blood and BALF was positively correlated with blood eosinophil counts.
Increased 15-LOX Activity
They next turned their attention to other fatty acid metabolites processed via the 12- and 15-LOX pathways. They found a significant but smaller increase in 12- and 15-HETE, suggesting increased AA metabolism as well as increased activity of the 15-LOX pathway in severe COVID-19. These were correlated with TXB2 levels. They also found evidence of increased non-enzymatic oxidation of AA and LA, indicating a burst of oxidative activity in these patients' lungs.
They postulate that severe COVID-19 patients have increased arachidonic acid (AA) production and metabolism. In support, they cite their findings of markedly higher precursor molecules, namely, linoleic acid (LA), AA, docosapentaenoic acid n-3 (DPAn-3), and docosahexaenoic acid (DHA), in the BALF of these patients, but without a corresponding change in eicosapentaenoic acid (EPA) levels. This suggests that multiple phospholipases A2 contribute to this lipidomic profile.
All the fatty acids were inversely related to the liver enzymes AST and ALT and positively to the ratio of platelets to lymphocytes. However, parameters such as blood counts, AST and ALT, or CRP, showed poor correlation with any of these metabolites.
Anti-Inflammatory Lipid Pathways
These samples also contain inflammation resolving molecules, SPMs, like 14- and 17-HDHA, at increased levels. While undetectable in all healthy individuals, multiple SPMs were found in the severe COVID-19 patients, with LXA4 being at the highest level. This signifies that both the 5- and 15-LOX pathways are involved in severe COVID-19.
The levels of the SMPs correlated with each other but not with the level of DHA. This suggests that the rise in D-series pro-resolving molecules is due primarily to high 15-LOX activity, and not merely the greater availability of DHA. LXA4 levels correlated well with other compounds from the 5- and 15-LOX pathways since it is a product of these pathways, and also with TXB4 and other prostaglandin molecules.
The study also shows the co-existence of prostaglandins, leukotrienes and SPMs in a situation of acute lung inflammation. They may mediate the enhancement of host defenses to limit the infection. The levels of SPMs are probably proportional to the activity of biosynthetic enzymes rather than those in the LOX pathway, acting on EPA and DHA, both of which are omega-3 fatty acids.
Most of the lipids found in the current study showed a high degree of correlation. The rise in the levels of the bioactive lipids did not correlate with the levels of fatty acids in the blood, suggesting the greater role played by the activation of enzymes such as COX and LOX in this process.
Drug Targets in Lipid Pathways
There are many drugs directed at inhibition of the prostaglandin pathway, such as the corticosteroid dexamethasone. This drug reduces symptom severity and mortality in severe COVID-19, reduces COX-2 expression by inhibiting the production of prostaglandins and also, perhaps, TXA2. Some suggest that it also increases the production of SPMs, limiting the infection. Further study is required.
Aspirin, a non-steroidal anti-inflammatory drug (NSAID), is also a COX inhibitor and has been suggested to reduce COVID-19 severity and adverse outcomes. The 5-LOX inhibitor Zileuton could also play a role in reducing LT production since it is already approved for the treatment of asthma and has a good safety profile.
The drug Ramotraban, which inhibits both DP2/TP pathways, could be a good choice for testing in this disease. Another candidate is the phosphodiesterase 4 inhibitor Roflumilast, which might increase cAMP levels, thus inhibiting AA release and thereby reducing the output of both prostaglandins and leukotrienes. Adenosine and PGE2 are known for their anti-inflammatory effects mediated via their inhibition of many white cell functions associated with inflammation. Of course, the action of phosphodiesterase inhibitors reduces PGE2 levels, but the residual PGE2 is amplified in its anti-inflammatory activity.
What are the Implications?
The authors conclude, "Our data unmask that in addition to the recognized cytokine storm previously documented, lung fluids of SARS-CoV-2-infected patients indicate that a lipid storm also occurs. The rich library of governmental health agencies-approved lipid modulators might provide beneficial and usually low-cost add-ons to the current therapeutic arsenal utilized to diminish the severity of COVID-19 and possibly additional coronavirus-mediated infections."

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources