The coronavirus disease 2019 (COVID-19), caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), shows no sign of abating soon. In fact, the recent surge of cases caused by new and potentially more lethal variants of the virus, with immune evasion capabilities, threatens to overwhelm healthcare systems in many countries, even as vaccines are being rolled out in many.
There is, therefore, a great need to understand how the illness can be diagnosed using more convenient and inexpensive methods. A new study posted to the medRxiv* preprint server reveals the potential role of skin imprints to test for biomarkers that indicate a diagnosis of COVID-19 in a non-invasive way.
Multiple abnormalities in SARS-CoV-2 infection
SARS-CoV-2 infection is known to cause illness through multiple metabolic abnormalities induced by a host of immunological aberrations. These include and are mediated by coagulopathies and hyperintense inflammatory processes.
Many researchers in earlier studies have shown that protein and lipid metabolism undergoes marked changes in this infection. The samples used for these studies mainly included clinical specimens, including plasma, urine, and saliva.
The symptom profile of COVID-19 reflects the distribution of the host cell receptor for the virus. Namely, the angiotensin-converting enzyme 2 (ACE2). Since these are present in the mouth, lungs, skin keratinocytes, and gut, these organs are often involved in disease manifestations.
Skin lesions occur in COVID-19, though rarely. These include reddish-violet lesions, chilblain-like lesions on the toes and fingers, blisters and papules, purpuric spots and urticaria. These may be linked to the high level of inflammation and coagulopathy in this illness.
Given this, metabolites on the skin surface could be a very useful resource for the non-invasive and rapid diagnosis of COVID-19.
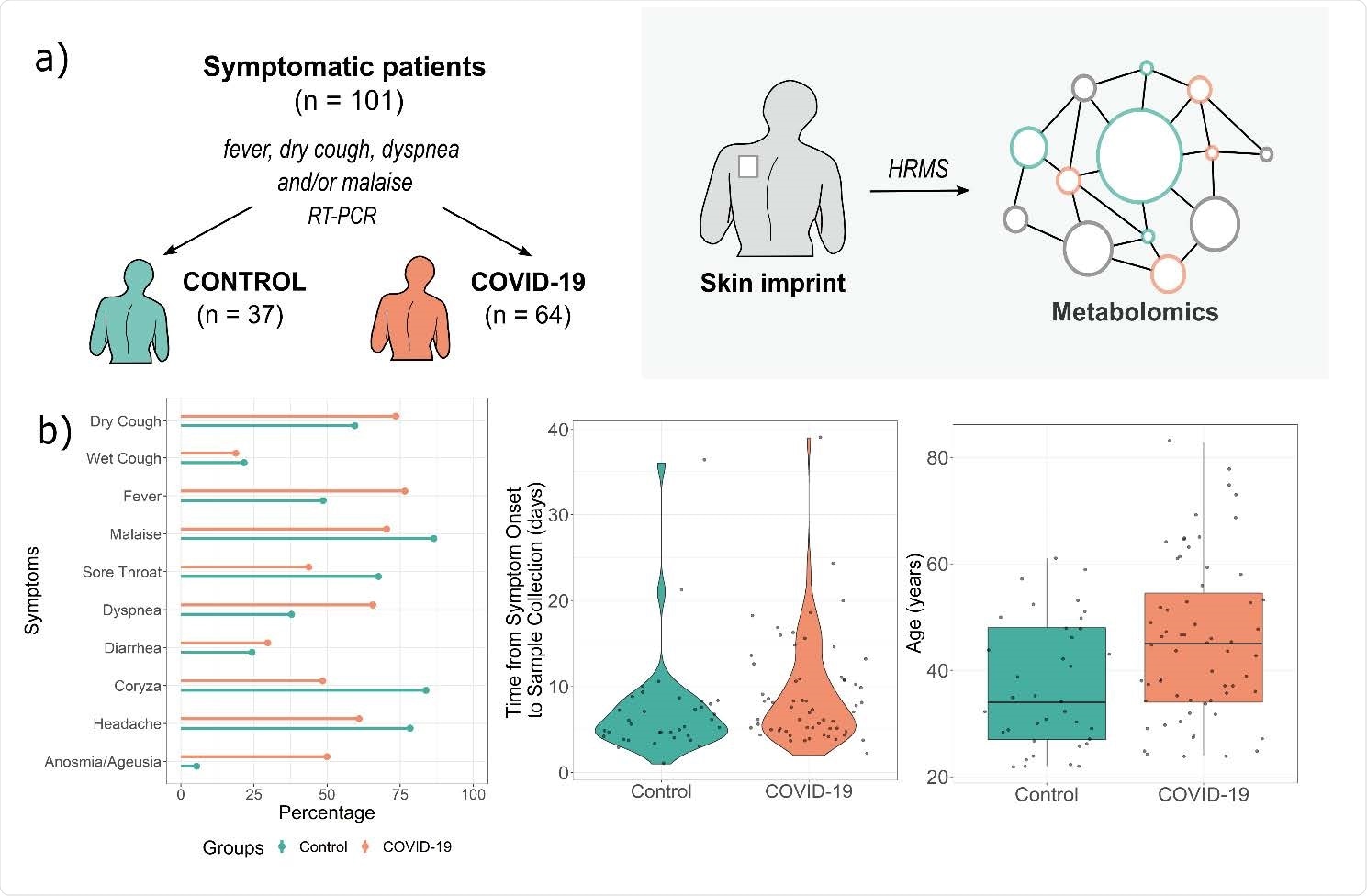
Study design (a) and patient symptoms distributions and age characteristics (b). HRMS – high-resolution mass spectrometry, RT-PCR – reverse transcriptase-polymerase chain reaction.

 *Important notice: medRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.
*Important notice: medRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.
Study details
Based on earlier research using silica plates to receive skin imprints, with mass spectrometry to analyze the metabolite profile thus imprinted, the researchers carried out the current study to assess the diagnostic utility of this method.
This would help understand the change in the metabolic signature even in normal skin during this infection and add to the sum of knowledge of its pathophysiology.
All study participants were adults with COVID-19, diagnosed by reverse transcriptase-polymerase chain reaction (RT PCR), the gold standard for diagnosis after they reported symptoms of this illness. About 60% of virus-positive individuals were female, compared to ~80% of the controls, who had similar symptoms but tested negative.
The skin imprints were taken from the upper left side of the back of the participants.
What were the results?
The broad metabolomic assay showed changes in 14 markers, of which half showed an increase and half a decrease relative to controls.
The former category belonged to primary fatty acid amides (PFAMs), N-acylethanolamines (NAEs), N-acyl amino acids, and glycerolipids. Those that were decreased included phosphatidylserines, dipeptides and sterols.
The most significant changes are in oleamide, linoleamide, and a diacylglycerol, with the first showing 87% higher values in COVID-19 patients.
For controls, the dipeptide cysteinyl-glutamine and phosphatidylserines showed the most significant measure of change.
Based on the receiver operating characteristic (ROC), oleamide changes could predict the infection with an accuracy of 77%, with 77% sensitivity and 79% specificity.
Overall, the panel of biomarkers had a sensitivity of 74% and a specificity of over 82%, indicating the promise of this method.
What were the implications?
These findings indicate the importance of exploring COVID-19 manifestations outside the respiratory, nervous system, cardiovascular, hematologic and gastrointestinal systems.
“Our study aims to provide an overview on how the skin biochemistry is impacted by COVID-19 infection, demonstrating the main biomarkers present in this scenario, as well as the possible pathways affected by the biochemical disbalance, which may impact the endocannabinoid and immune system.”
Lipid alterations in the skin
The skin lesions peculiar to this condition are being recognized more widely. Though keratinocytes were shown to express ACE2 receptors at high levels, especially compared to basal cells, this study uses a novel and non-invasive method to understand the metabolic alterations in the skin of COVID-19 patients, even those without overt skin lesions.
Sebum and triacylglycerol characteristics have been shown to be altered in COVID-19, and in the current study too, both triglycerides and diacylglycerol were found to be reduced in this condition.
Most viral infections show accompanying disturbances in lipid metabolism, and this is peculiarly pronounced in COVID-19. In fact, glycerolipid aberrations are found to be correlated with the diagnosis as well as with the severity of this disease.
Importance of phosphatidylserine alterations
The reduction in phosphatidylserines in COVID-19, irrespective of disease severity, in both blood samples and upper airway swabs has been reported in earlier studies. Phosphatidylserine is a membrane phospholipid with a negative charge, which may move from the inner layer of the bilayer membrane to the outer layer in incipient viral infection.
It may signal to macrophages to initiate phagocytosis and may also contribute to platelet activation, thus triggering or enhancing the cascade of inflammatory immunological changes that is characteristic of severe COVID-19.
Significance of fatty acid changes
Fatty acids such as oleamide and linoleamide, part of the PFAM group of lipids, seem to be part of the regulatory mechanism for both nervous and immune systems. They are also components of the endocannabinoid system, with the ability to modulate the activity of endocannabinoids with their receptors, CB1 and CB2.
With increased PFAM levels, these, along with NAEs, being inactive at the cannabinoid receptors, may prevent the inactivation of endocannabinoids. This increases the level of these active molecules and prolongs their activity at their receptors.
Future directions
Since CB2 is found in the skin and skin appendages, as well as in immune cells, with an immunosuppressive effect, these receptors inhibit the recruitment and activation of the first antiviral defenses, namely, innate immune cells. They also reduce the activation of T helper cells and the inflammatory response. This action may be carefully exploited in the treatment of COVID-19.
The researchers suggest that the method may usefully distinguish COVID-19 from other similar mild viral illnesses. Still, that risk stratification will be possible only with more extensive studies, including a more varied set of patients.

 *Important notice: medRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.
*Important notice: medRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.