Various measures around the world have been implemented to contain the spread of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) since its discovery in late December 2019. However, even with precautions and protective measures applied at many levels, the risk of infection via airborne and respiratory droplet spread remains high when many people are confined together in a closed space.
With a combination of positively charged and negatively charged fatty acids, natural oils can make a preventative barrier against viral infections.
A new preprint research paper posted to the bioRxiv* server offers a potential candidate for disinfecting the respiratory tract and mouth during and after exposure to the virus, in the form of proprietary herbal oil named Viruxal.

 *Important notice: bioRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.
*Important notice: bioRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.
Background
SARS-CoV-2 enters the body through the mucosal membranes of the nose, mouth and eyes. This happens mostly through direct contact with aerosolized or droplet viruses, or through indirect contact between the hands after touching a virus-contaminated surface and the mucosae.
The primary recommendations to prevent such transmission have been social distancing, regular hand washing, and the use of face masks, whenever one leaves one’s home. Despite this, infection risk is significant during public transport or when traveling across national borders, eating at indoor restaurants, in crowded areas, and some sports.
In such circumstances, other protective measures are clearly needed, but none have so far been discovered.
Viruxal has high virucidal activity
Viruxal is formulated as oil, because natural oils contain both anionic and cationic fatty acids that coat the mucosal surface to create a barrier to viruses. This plant-based oil is sprayed onto the nasal or oral mucosa, where it makes it difficult for microbes to adhere to the epithelium, while inactivating virus particles.
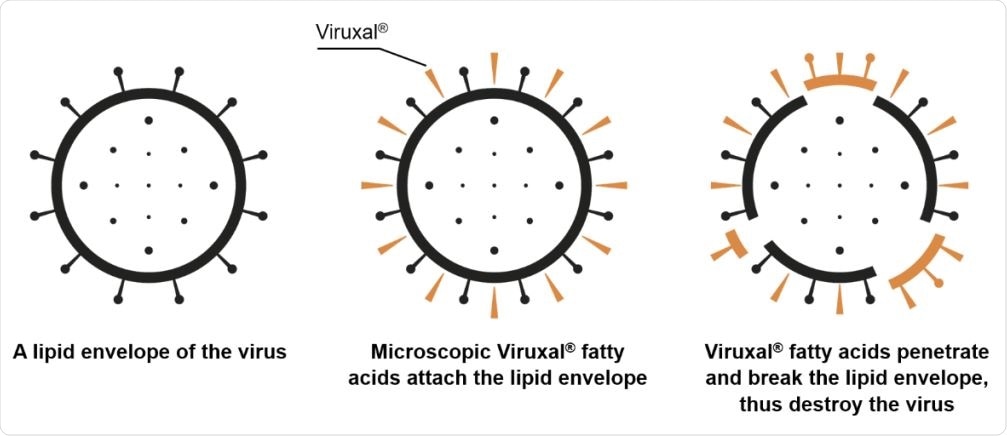
A proposed antiviral mechanism of fatty acids in Viruxal against enveloped viruses.
It has already passed in vitro tests for safety and effectiveness, and clinical trials are underway. The current paper presents the results of testing Viruxal effects on the SARS-CoV-2 and Influenza A(H1N1) viruses.
Different doses of Viruxal were used in spray form (ViruxOral and ViruxNasal) on the two viruses. Both have the same active ingredients, namely, oleic, palmitic, linoleic, stearic and linolenic acids (all of which are natural fatty acids), triglycerides and free fatty acids. The spray was used in the undiluted form at 100% strength, as well as at 50%, 25% and 12.5%.
99% inactivation of SARS-CoV-2
At all concentrations, Viruxal reduced SARS-CoV-2 viral titers by over 1 log in just one minute. In other words, it inactivated >90% of the virus. At 30 minutes, the same degree of success was observed at 100% strength, but at 50, 25 and 12.5%, >99% of the virus was inactivated.
However, it did not reduce the viral load to undetectable levels at any concentration at any time point.
99.99% inactivation of influenza
With Influenza A(H1N1) virus, Viruxal reduced viral loads by >99.99% (>3 logs) at 100% strength, with 1 minute contact time. At 50% strength, >99% of the virus particles were inactivated, for a reduction of >2 logs.
After 30 minutes of contact, Viruxal reduced the viral load >2 logs at all concentrations. 100% strength oil reduced it > 3 logs. Again, the virus remained detectable at all concentrations.
Thus, Viruxal is shown to be a potent virucidal oil in vitro, with potential inactivation efficacy against SARS-CoV-2 and influenza A(H1N1).
How Viruxal works
It is supposed that the fat molecules degrade the lipid covering of these enveloped viruses and thus inactivate them before they can enter the host cells to establish infection.
Natural fats, both vegetable and animal, have long been shown to possess antibacterial, antifungal and antiviral (to enveloped viruses) activity.
Both gram-positive and gram-negative bacteria are inhibited, though not killed, by linoleic acid, linolenic acid, and oleic acid (the last only in part). Enveloped viruses like the herpes virus and the flu virus are also inactivated rapidly by unsaturated fatty acids like oleic and linoleic acid.
Fresh milk is rich in fatty acids, which may account for the protection it confers against viral infections in infancy, especially when stored at 4°C.
Viruxal is rich in fatty acids with medium to long side chains, the latter class being unsaturated while the former are saturated. These have high antiviral activity while other saturated fatty acids lack it. The ability to inactivate viruses is due to the damage they cause to the fatty viral envelope, allowing the contents to leak and even the complete breakup of the whole virus.
What are the implications?
Viruxal promises to be useful if these results are confirmed because of its ability to suppress SARS-CoV-2 shedding from the nose and the mouth. In addition, these are the entrance points for viruses, and the oil reduces their activity here dramatically.
By blocking viral infection, Viruxal protects the exposed person. Again, by inactivating the virus on the oral and nasal mucosa in already infected individuals, it protects others by reducing the transmission risks.
Thirdly, the broad spectrum of action of Viruxal reduces the chances of influenza, either alone, as during the annual winter flu outbreaks in the temperate zone, or along with COVID-19, since both viruses are inactivated by the oil.
This in vitro study offers a highly promising drug candidate that could inactivate enveloped viruses, including the novel coronavirus SARS-CoV-2.
Research is needed to validate these findings, as well as to develop the treatment for COVID-19 and other enveloped respiratory virus infections.
Competing Interest Statement
Corresponding author Jón Magnús Kristjánsson is an employee of the company Viruxal that is a subsidiary of Kerecis.

 *Important notice: bioRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.
*Important notice: bioRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.