Researchers in the United States have conducted a study suggesting that individuals fully vaccinated against coronavirus disease 2019 (COVID-19) who develop breakthrough infection with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) are less likely to transmit the virus to others than unvaccinated infected individuals.
The team – from Johns Hopkins School of Medicine and The Johns Hopkins Bloomberg School of Public Health in Baltimore, Maryland – found that breakthrough infection among fully vaccinated individuals was associated with a reduced recovery of infectious virus.
Heba Mostafa and colleagues also found that breakthrough infection was associated with the presence of the SARS-CoV-2 lineage B.1.526 and the mutation E484K in the viral spike protein.
A pre-print version of the research paper is available on the medRxiv* server, while the article undergoes peer review.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
More about breakthrough infection
Although the current COVID-19 vaccines have been shown to be highly effective at preventing SARS-CoV-2 infection and severe disease, breakthrough infections have still been reported among fully vaccinated individuals.
As of June 1st, 2021, the Centers for Disease Control and Prevention reported that among more than 135 million fully vaccinated individuals in the US, 3,016 cases of breakthrough infections had occurred that resulted in hospitalization or death.
However, little is known regarding the immune factors at play and the infectivity of viral loads among vaccinees with breakthrough infection.
Concerns surrounding emerging variant
Currently, data support that the vaccines approved for emergency use in the US are effective against most of the SARS-CoV-2 variants currently circulating.
However, “with the general increase in the circulation of variants of concern, it is expected to see a high percentage of breakthrough infections caused by these variants,” writes Mostafa and the team.
What did the researchers do?
Now, the team has provided a comprehensive analysis of samples taken from 133 individuals who tested positive for SARS-CoV-2, despite having received two doses of either the Pfizer-BioNTech BNT162b2 vaccine or the Moderna mRNA-1273 vaccine.
The second vaccine dose had been received between January and April of 2021. Samples were collected between January and May of 2021, with collection times ranging from two to 100 days following the second dose.
The team performed whole-genome sequencing to characterize viral spike protein changes and a polymerase chain reaction (PCR) assay was used to determine the cycle thresholds (Ct; number of PCR cycles required to detect the virus).
Levels of local anti-SARS-CoV-2 immunoglobulin G (IgG) antibodies were examined using an enzyme-linked immunosorbent assay (ELISA) and nasopharyngeal specimens were grown on cell culture to assess the recovery of infectious virus.
The same tests were carried out on samples from a cohort of 124 unvaccinated SARS-CoV-2-positive individuals from a matched time frame that served as a control group.
What did the study find?
Of 133 specimens, 24 failed sequencing and yielded a negative or very low viral load. Of the 109 specimens that underwent further genomic analysis, 68 (62.4%) were from individuals with symptomatic infections, 11 (10.1%) were from hospitalized individuals and two (1.8%) were from people who required ICU admission.
“Our data show that 37.6 % of these breakthrough infections were asymptomatic and in symptomatic patients, the majority were mild cases that did not require hospitalization,” writes the team.
The majority (61%) of infections identified belonged to the B.1.1.7 (alpha) lineage that emerged in England.
However, significant increases in the representation of the New York B.1.526 lineage and the spike (S) amino acid change E484K were observed among the vaccinees with breakthrough infection.
The S: E484K mutation has emerged in various SARS-CoV-2 lineages that have previously been shown to exhibit reduced susceptibility to neutralization by sera collected from vaccinated individuals.
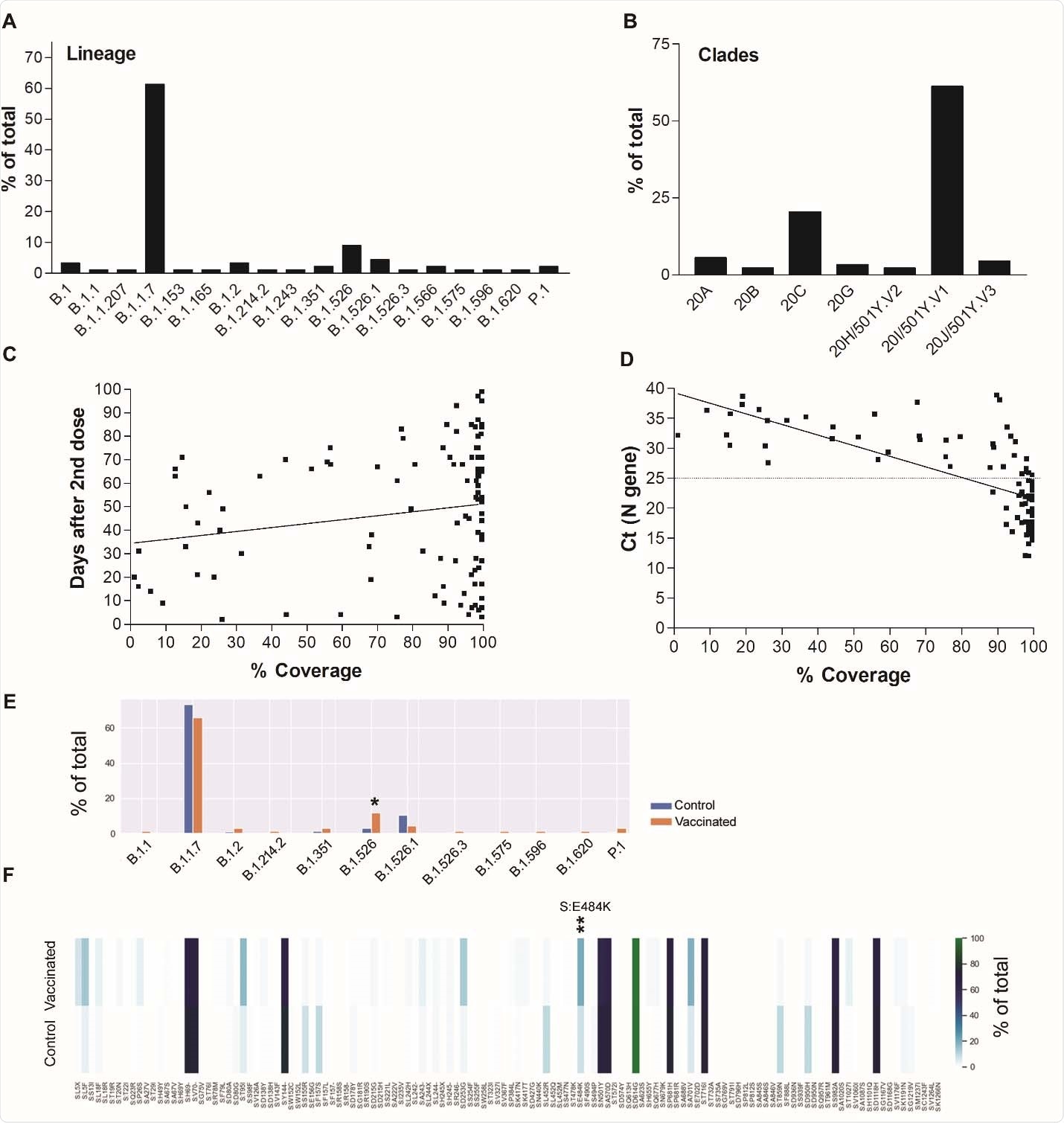
SARS-CoV-2 genomes of positives after full vaccination. A) Lineages and B) Clades of genomes with more than 50% coverage and average depth of 50 (n=88). C) A correlation of SARS-CoV-2 genome coverage with the days of sample collection after the second dose of vaccination. D) A correlation between SARS-CoV-2 genome coverage and cycle thresholds of the N gene using the PerkinElmer SARS-CoV-2 assay. E) A comparison between lineages from fully vaccinated (n = 67) and control (n = 335) genomes with coverage ≥ 90%. F) Spike amino acid changes in vaccinated and control groups. * p < 0.05, *** p < 0.005
Negative recovery of infectious virus correlated with high levels of IgG among vaccinees
Among samples from the vaccinees, only 18.5% exhibited any cytopathic effect on cell culture, compared with 64.5% of control samples.
Furthermore, the recovery of infectious virus from samples was faster for the control group versus the vaccinated group.
Interestingly, a significant increase in anti-SARS-CoV-2 IgG titers was observed in nasal and nasopharyngeal samples from the vaccinated group, compared with those from the control group.
“Negative recovery of infectious virus on cell culture correlated with high levels of IgG in respiratory specimens,” says the team.
To better understand the contribution of Ct values to virus recovery on cell culture, the researchers focused the analysis on samples with Ct values below 25.
This revealed that despite a similar distribution of Ct values between vaccinated and control samples, the recovery of infectious virus was significantly attenuated for the vaccinated samples.
“The reduction in infectious virus in samples with Ct values of less than 25 could be explained if the nasal SARS-CoV-2 IgGs are neutralizing,” writes Mostafa and colleagues.
What are the implications of the findings?
The researchers say the findings indicate that infection among vaccinated individuals results in reduced infectious virus load compared with among unvaccinated individuals and that the Ct value from infected, vaccinated individuals is associated with lower infectious virus loads.
This may reduce the likelihood that infected vaccinated individuals can transmit SARS-CoV-2 to others, they add.
“This study is also the first to show the significant association of S: E484K with positives after full vaccination using a well-controlled analysis and a relatively large sample size,” says Mostafa and colleagues.
“This data highlights the significance of continuous genomic surveillance coupled with metadata and laboratory data collections for providing significant information with the potential to impact decisions related to booster doses or modifying vaccine strains,” they conclude.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Journal references:
- Preliminary scientific report.
Mostafa H, et al. SARS-CoV-2 Infections in mRNA Vaccinated Individuals are Biased for Viruses Encoding Spike E484K and Associated with Reduced Infectious Virus Loads that Correlate with Respiratory Antiviral IgG levels. medRxiv, 2021. doi: https://doi.org/10.1101/2021.07.05.21259105, https://www.medrxiv.org/content/10.1101/2021.07.05.21259105v1
- Peer reviewed and published scientific report.
Mostafa, Heba H., Chun Huai Luo, C. Paul Morris, Maggie Li, Nicholas J. Swanson, Adannaya Amadi, Nicholas Gallagher, and Andrew Pekosz. 2022. “SARS-CoV-2 Infections in MRNA Vaccinated Individuals Are Biased for Viruses Encoding Spike E484K and Associated with Reduced Infectious Virus Loads That Correlate with Respiratory Antiviral IgG Levels.” Journal of Clinical Virology 150-151 (June): 105151. https://doi.org/10.1016/j.jcv.2022.105151. https://www.sciencedirect.com/science/article/pii/S1386653222000841.