Among individuals who get acutely infected with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), a substantial share has been seen to develop prolonged symptoms. As a result, this serious condition has been termed post-acute coronavirus disease 2019 (COVID-19) syndrome (PACS) or long COVID. To develop more efficient treatment options, predictors of PACS are needed.
Study: Immunoglobulin signature predicts risk of post-acute COVID-19 syndrome. Image Credit: Dana.S/ Shutterstock
Background
Symptoms of COVID-19 infection include fever, fatigue, myalgia, weakness, headache, change in smell or taste, etc. In response to the infection, the human body mounts an immune response characterized by increased serum concentrations of chemokines, proinflammatory cytokines, and tumor necrosis factor (TNF). Subsequently, activated monocytes appear, and so do SARS-CoV-2-specific immunoglobulin M (IgM), IgA, and IgG antibodies and interferon-γ-producing T cells.
Most individuals recover within 3 weeks of contracting the infection. However, about one-third of individuals report one or more COVID-19-related symptoms that last for more than 4 weeks - a condition termed post-acute COVID-19 syndrome (PACS) or long COVID. PACS could be subacute COVID-19 (symptoms last 12 weeks or less) or post-COVID-19 syndrome (symptoms persist for more than 84 days).
The most commonly reported symptoms are fatigue, dyspnea, cognitive impairment, pain and aches, cough, change in smell or taste, and diarrhea. PACS is increasingly recognized as a serious consequence of COVID-19 infection, making its early identification crucial.
Key Findings of the New Study
Scientists showed that the development of PACS correlated with a distinct Ig signature. Further, it also closely correlated with patients’ age, history of asthma bronchial, and a number of other symptoms measured during primary infection.
These findings were translated into a model, which was called PACS score. For this study, a cohort of 134 COVID-19 patients was followed up and extensively characterized. When the PACS score was applied to this sample, it performed much better than a symptom-based score.
Further, the results were independent of the timepoint of testing, sex, and only required broadly available Ig measurements. Previous studies have indicated female sex to be a risk factor.
However, researchers claimed that the male sex is associated with a worse outcome in acute COVID-19. A sex-independent prediction score could be applied to different healthcare settings.
The measurement of an Ig signature is superior to symptom-based prediction scores because it enables the identification of patients at risk of developing PACS. This is all the more important for hospitalized patients.
Scientists stated this result hints at a potential pathomechanism, which is different from increased inflammation and immune activation. It must also be noted that inflammatory markers only transiently increase early in the disease course, while unspecific Ig levels are more stable over time. This further increases the utility of Igs as biomarkers. In addition, the irrelevance of sampling point aids in the clinical application, and Ig signatures can be already be determined prior to infection.
Researchers further conducted decision curve analyses and found that the highest clinical benefit of the PACS score lies between threshold probability ranges of 40–60% and above 55% in hospitalized patients. In the future, the PACS score could be applied in settings where false-positive predictions could be of greater harm than false negatives, and this would help target patients at very high risk for developing PACS.
Scientists recommended the use of the PACS score to identify outpatients at risk, high-risk asthmatic patients, and hospitalized patients. Early measurement of Ig titers upon hospitalization of COVID-19 patients could help in appropriate clinical decision-making and devising personalized treatment strategies.
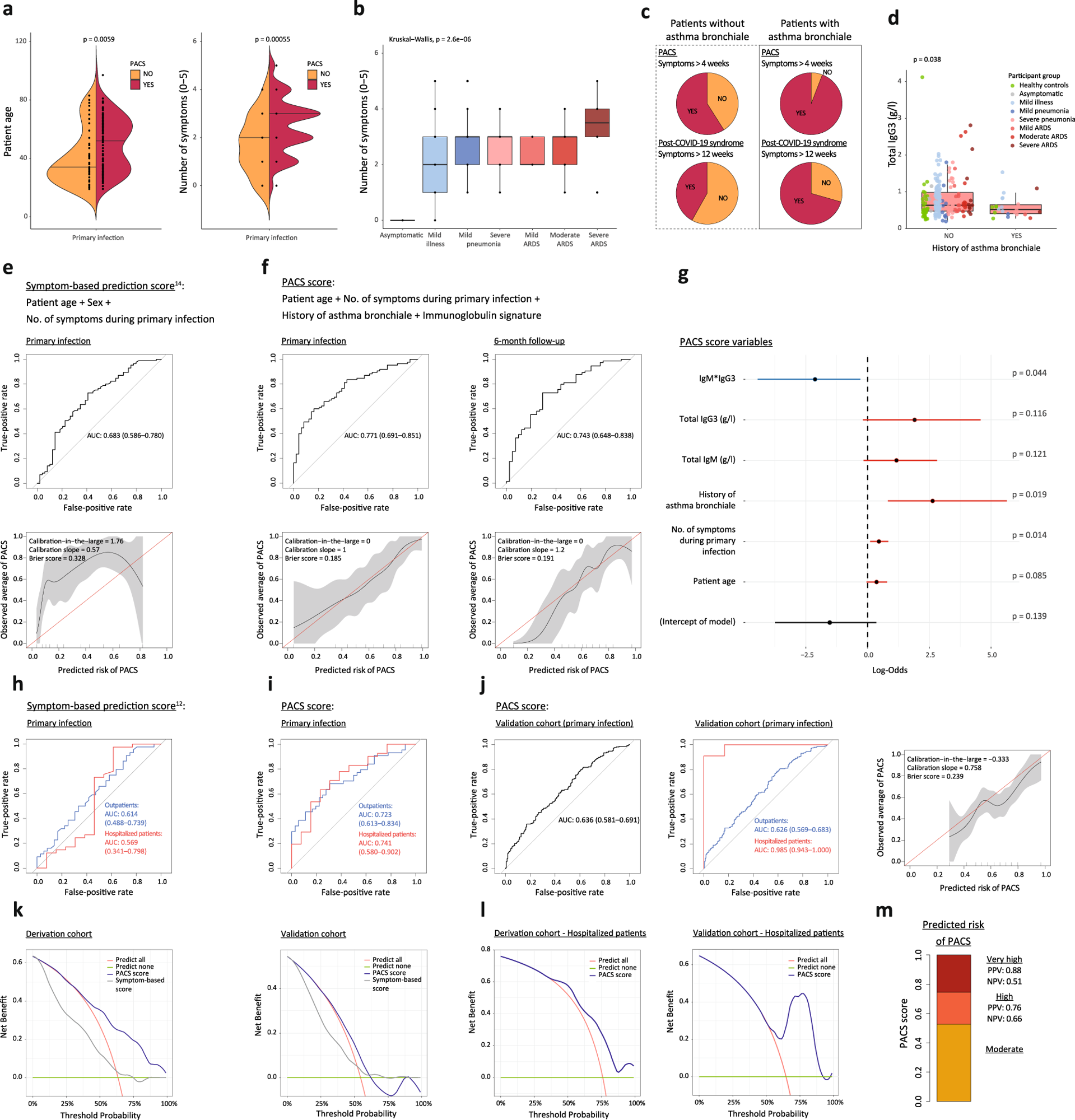 a Age and number of symptoms during primary infection (0–5; fever, fatigue, cough, dyspnea, gastrointestinal symptoms) in patients without or with PACS. b Number of symptoms during primary infection in COVID-19 patients with different disease severities (n = 134, with 85 having PACS). c PACS and post-COVID-19 syndrome in patients without and with history of asthma bronchiale. d IgG3 titers in healthy controls (green symbols) and all COVID-19 patients (n = 215; disease severity indicated by colors) at primary infection, without or with history of asthma bronchiale. e and f Receiver operating characteristic (ROC) curves (top) and calibration plots (bottom) reporting the area under the curve (AUC) with 95% confidence intervals (CI) or calibration-in-the-large, calibration slopes, and Brier scores of logistic regression models for predicting PACS. Use of (e) a symptom-based model14 and (f) the PACS score on data of our patient cohort at primary infection (e and f, left; n = 134, with 85 having PACS) and after shrinkage of coefficients on 6-month follow-up-data (f, right; n = 115, with 74 having PACS). g Regression coefficients of PACS score with 95% CI and p values. h and i ROC curves reporting AUC with CI of PACS score in outpatients (blue) and hospitalized patients (red) of derivation cohort (n = 80 and 54, with 44 and 41 having PACS, respectively). j Validation of PACS score in independent cohort at primary infection (n = 389, with 212 having PACS) and subgroup analysis in outpatients (blue; n = 372, with 201 having PACS) and hospitalized patients (red; n = 17, with 11 having PACS). k Decision curve analysis of PACS score in derivation (left) and validation cohort (right) comparing the PACS score to a symptom-based score14 and clinical strategies of predicting none or all subjects with COVID-19 develop PACS. l Decision curve analysis of PACS score in hospitalized patients. m Estimated risk groups based on two probability thresholds (0.523 and 0.746) with corresponding positive (PPV) and negative (NPV) predictive values in the derivation cohort. Boxplots represent median (middle line) with upper and lower quartiles (box limits), and 1.5*interquartile ranges (whiskers). Variables were compared using a two-sided Wilcoxon’s test if not specified otherwise.
a Age and number of symptoms during primary infection (0–5; fever, fatigue, cough, dyspnea, gastrointestinal symptoms) in patients without or with PACS. b Number of symptoms during primary infection in COVID-19 patients with different disease severities (n = 134, with 85 having PACS). c PACS and post-COVID-19 syndrome in patients without and with history of asthma bronchiale. d IgG3 titers in healthy controls (green symbols) and all COVID-19 patients (n = 215; disease severity indicated by colors) at primary infection, without or with history of asthma bronchiale. e and f Receiver operating characteristic (ROC) curves (top) and calibration plots (bottom) reporting the area under the curve (AUC) with 95% confidence intervals (CI) or calibration-in-the-large, calibration slopes, and Brier scores of logistic regression models for predicting PACS. Use of (e) a symptom-based model14 and (f) the PACS score on data of our patient cohort at primary infection (e and f, left; n = 134, with 85 having PACS) and after shrinkage of coefficients on 6-month follow-up-data (f, right; n = 115, with 74 having PACS). g Regression coefficients of PACS score with 95% CI and p values. h and i ROC curves reporting AUC with CI of PACS score in outpatients (blue) and hospitalized patients (red) of derivation cohort (n = 80 and 54, with 44 and 41 having PACS, respectively). j Validation of PACS score in independent cohort at primary infection (n = 389, with 212 having PACS) and subgroup analysis in outpatients (blue; n = 372, with 201 having PACS) and hospitalized patients (red; n = 17, with 11 having PACS). k Decision curve analysis of PACS score in derivation (left) and validation cohort (right) comparing the PACS score to a symptom-based score14 and clinical strategies of predicting none or all subjects with COVID-19 develop PACS. l Decision curve analysis of PACS score in hospitalized patients. m Estimated risk groups based on two probability thresholds (0.523 and 0.746) with corresponding positive (PPV) and negative (NPV) predictive values in the derivation cohort. Boxplots represent median (middle line) with upper and lower quartiles (box limits), and 1.5*interquartile ranges (whiskers). Variables were compared using a two-sided Wilcoxon’s test if not specified otherwise.
Concluding Remarks
The study has some limitations, including a selection bias of patients enrolled in the study; thereby affecting the transferability of the findings to all COVID-19 patients.
Further, the study included only a small number of non-white participants due to Central European demographics. In summary, PACS could result from tissue damage due to high viral loads, excessive inflammation, or thrombotic events.
The results presented in this study highlight the benefits of measuring Igs for the early identification of patients at high risk for PACS. This is extremely important for understanding the pathomechanisms of PACS and identifying preventive measures for treatment and care.