Rodent brain imaging has led to a deeper insight into brain mechanisms and provides a basis for determining the causes of several neurodegenerative diseases. Complete neural-network diagrams have been mapped, characterizing all input and output of every region in the rodent. This achievement offers a large number of benefits by identifying and locating the specific neuronal circuits involved in emotional, cognitive and motor processes.
Such maps provide better opportunities for studying diseases such as schizophrenia, which is caused by unusual interactions between two distinct cerebral structures. In terms of improving understanding of the causation and related processes in various neurodegenerative diseases such as Huntington’s and Alzheimer’s, imaged models have become invaluable.
Advantages of Models
Models help elucidate how specific diseases are associated with the loss of synapses and neurons in different regions of the brain. They can also help establish potential treatments that can be investigated as a way of blocking the pathways that result in neuronal death or lead to the disappearance of synapses between cells and therefore loss of function. In addition, these models have enabled significant advancements in the sourcing of gene mutations that lead to abnormal protein expression and subsequently motor-neuron degeneration.
Advancements in Rodent Brain Imaging
Rodent brain imaging has been made possible owing to a number of technological advancements improving the ability to produce more precise and high quality brain images.
For instance, neural and axonal pathways in and out of the brain can be indicated by classical or viral fluorescent tracer injections. Ultrasound methods can be used to open the blood/brain barrier, thereby enabling microtubules with immunotherapies inside to be diffused to specific brain regions in a targeted manner.
Microscopic advancements allow for deeper imaging into tissue (depending on sample opacity and the working distance of the objective lens), as well as providing improved optical sections at a range of depths from thick tissue samples.
The benefits offered by optical sectioning methods are:
- Savings on preparation time
- Improved accuracy
- Spatial positioning precision for sections with reduced deformation
- Reduced use of thin sectioning
3-D micrometer scale tomography has been enabled by the use of optical sections and can be used to create high-resolution maps of small, stained rodent brains, including the depiction of associated neurons and corresponding processes. Clear images with improved optical resolution (especially in the depth direction), can also be produced using confocal fluorescence techniques where out-of-focus light from the sample that is outside the focal plane is eliminated via a pinhole.
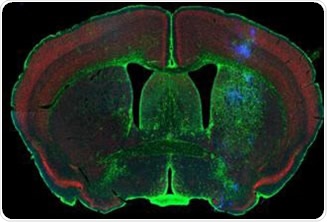
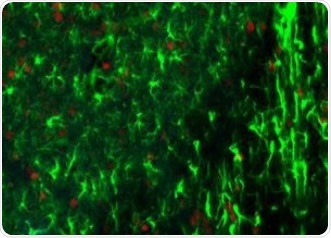
Figure 1. Rodent brain (top) depicting simultaneous acquisition of fluorophores Cy2, Cy3 and Cy5; Brain image (bottom) magnified to 0.5 μm. Image credit: Huron Digital Pathology.
Limitations of Traditional Microscopy in Rodent Brain Imaging
Significant amounts of tissue sections are used in imaging of the rodent brain. There are several limitations associated with using conventional microscopy techniques to image such volumes.
- Reduced Depth Penetration for Optical Sectioning – Higher numbers of optical sections cannot be obtained with existing optical sectioning methods because they do not provide adequate imaging depth into tissue. Most systems available can only image tissue sections that are less than 20μm thickness because these systems are limited by small working distances and increased light dispersion with increased depth. This further restricts the ability to create accurate 3D data sets.
- Light Interference Using Traditional Microscopes – Interference is a problem when using conventional non-confocal microscopes for optical sectioning because they are highly sensitive to light. Other light sources therefore interfere with the images being produced. Also, fluorescent images may have large unfocused backgrounds that overshadow weak features in the regions of interest.
- Increased Processing Time and Photo-Bleaching – Frame-by-frame excitation in fluorescence microscopy over exposes and photo-bleaches combined fluorophores which are exposed to needless repeated illumination from sequential channel acquisition.
TissueScope™ - Huron Digital Pathology’s Rodent Brain Imaging Solution
The TissueScope digital slide scanner offers significant benefits for imaging rodent brain specimens, as described below:
- A large (3mm) proprietary laser-scan lens working distance allows imaging of thicker tissue sections (up to 100μm), as well as facilitating confocal z-stacks and optical sectioning. The thickness of optical sections can be as small as 1μm.
- A greater penetration depth and optical sections enables significant savings on preparation time and improved spatial registration accuracy and precision alignment. When combined with clearing methods such as Scale, it is possible to image thick tissue sections of up to 500μm without reducing the intensity of fluorescent proteins. This is especially useful for observing neural networks of the hippocampus, cerebral cortex and white matter.
- A proprietary laser-scan lens and optical configuration enables a broad (5 to 10mm) field-of-view and therefore the imaging of more rodent brain sections per unit area (up to 48 square inches area).
- Software solutions are available for assembling optical sections into 3D models acquired or stored images. Voluminous image sections obtained from samples are housed and shared using server and storage solutions.
- Improved focusing algorithms enable thicker tissue topography to be imaged, while facilitating z-stacks and optical sectioning.
- This is the only confocal fluorescence digital slide scanner that enables the simultaneous acquisition of up to three fluorophores spanning the whole visible spectrum, from 400nm-850nm. This provides considerable time-savings and less photo bleaching due to the unique flying-spot laser platform.
- Weaker signals that would otherwise have been missed on traditional microscopes, can be acquired from samples by using highly sensitive photomultiplier tubes for image detection.
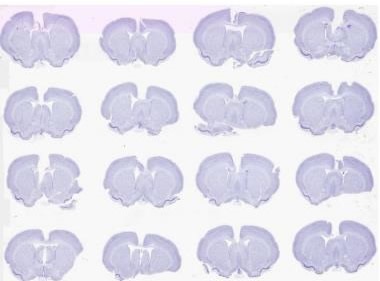
Figure 2. Brightfield scan of 16 Nissil stained rodent brains on a 2” x 3” slide at 0.5 μm. Image credit: Huron Digital Pathology.
About Huron Digital Pathology
 Based in Waterloo, Ontario, Canada, Huron Digital Pathology has a 20 year history designing sophisticated imaging instrumentation. Our end-to-end digital whole slide imaging solutions for digital pathology incorporate our award-winning TissueScope™ digital slide scanners; TissueView™ image viewing, sharing and management platform; and our workflow-enhancing accessories, which include our innovative TissueSnap™ preview scanning station.
Based in Waterloo, Ontario, Canada, Huron Digital Pathology has a 20 year history designing sophisticated imaging instrumentation. Our end-to-end digital whole slide imaging solutions for digital pathology incorporate our award-winning TissueScope™ digital slide scanners; TissueView™ image viewing, sharing and management platform; and our workflow-enhancing accessories, which include our innovative TissueSnap™ preview scanning station.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.Net which is to educate and inform site visitors interested in medical research, science, medical devices and treatments.